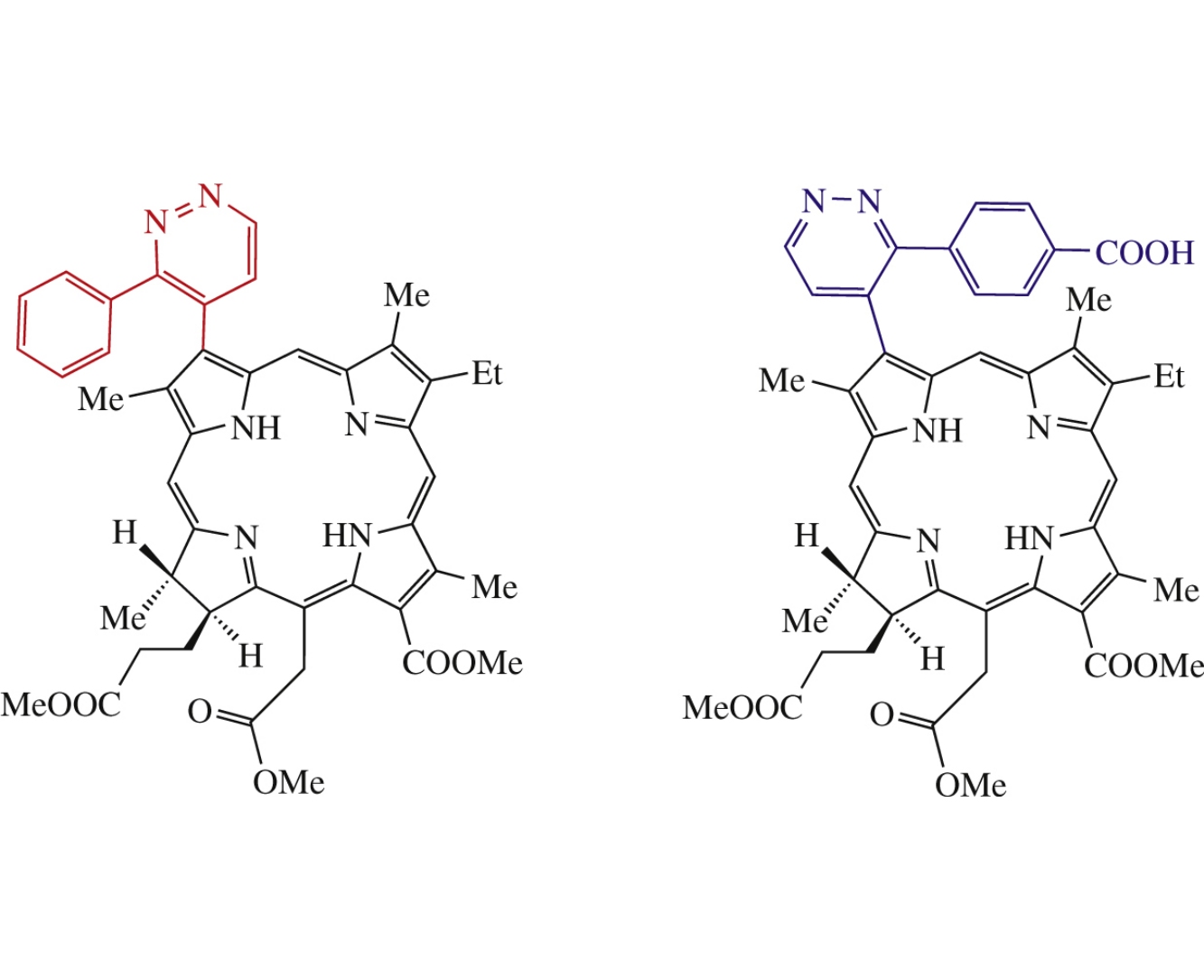
Abstract
The addition of 3-aryl-1,2,4,5-tetrazines at the vinyl group of chlorin e6 trimethyl ester affords the corresponding pyridazine–porphyrin conjugates. The study of spectral properties of the obtained compounds revealed insignificant changes in the absorbance and fluorescence spectra as compared to those of chlorin.
References
1.
Gushchina O.I., Gramma V.A., Larkina E.A., Mironov A.F.
Mendeleev Communications,
2017
2.

Oliveira K., Momo P., Assis F., Ferreira M., Brocksom T.
Current Organic Synthesis,
2014
3.

Tetrapyrroles: A chemical class of potent photosensitizers for the photodynamic treatment of tumours
Roeder B.
Lasers in Medical Science,
1990
4.
10.1016/j.mencom.2019.03.031_bib0020
Gorshkova
Tonkie Khimicheskie Tekhnologii,
2018
5.

Jinadasa R.G., Hu X., Vicente M.G., Smith K.M.
Journal of Medicinal Chemistry,
2011
6.

Chen H., Humble S.W., Jinadasa R.G., Zhou Z., Nguyen A.L., Vicente M.G., Smith K.M.
Journal of Porphyrins and Phthalocyanines,
2017
7.

Suvorov N.V., Machulkin A.E., Ivanova A.V., Popkov A.M., Bondareva E.A., Plotnikova E.A., Yakubovskaya R.I., Majouga A.G., Mironov A.F., Grin M.A.
Journal of Porphyrins and Phthalocyanines,
2018
8.

Pavlov V.Y., Ponomarev G.V.
Chemistry of Heterocyclic Compounds,
2004
9.

Gerlach B., Brantley S.E., Smith K.M.
Journal of Organic Chemistry,
1998
10.
Lebedeva V.S., Karmova F.M., Toukach F.V., Mironov A.F.
Mendeleev Communications,
2015
11.
Lonin I.S., Grin M.A., Lakhina A.A., Mironov A.F.
Mendeleev Communications,
2012
12.

Jiang X., Pandey R.K., Smith K.M.
Tetrahedron Letters,
1995
13.

Kolb H.C., Sharpless K.B.
Drug Discovery Today,
2003
14.

Carboni R.A., Lindsey R.V.
Journal of the American Chemical Society,
1959
15.

Oliveira B.L., Guo Z., Bernardes G.J.
Chemical Society Reviews,
2017
16.

Karver M.R., Weissleder R., Hilderbrand S.A.
Bioconjugate Chemistry,
2011