Abstract
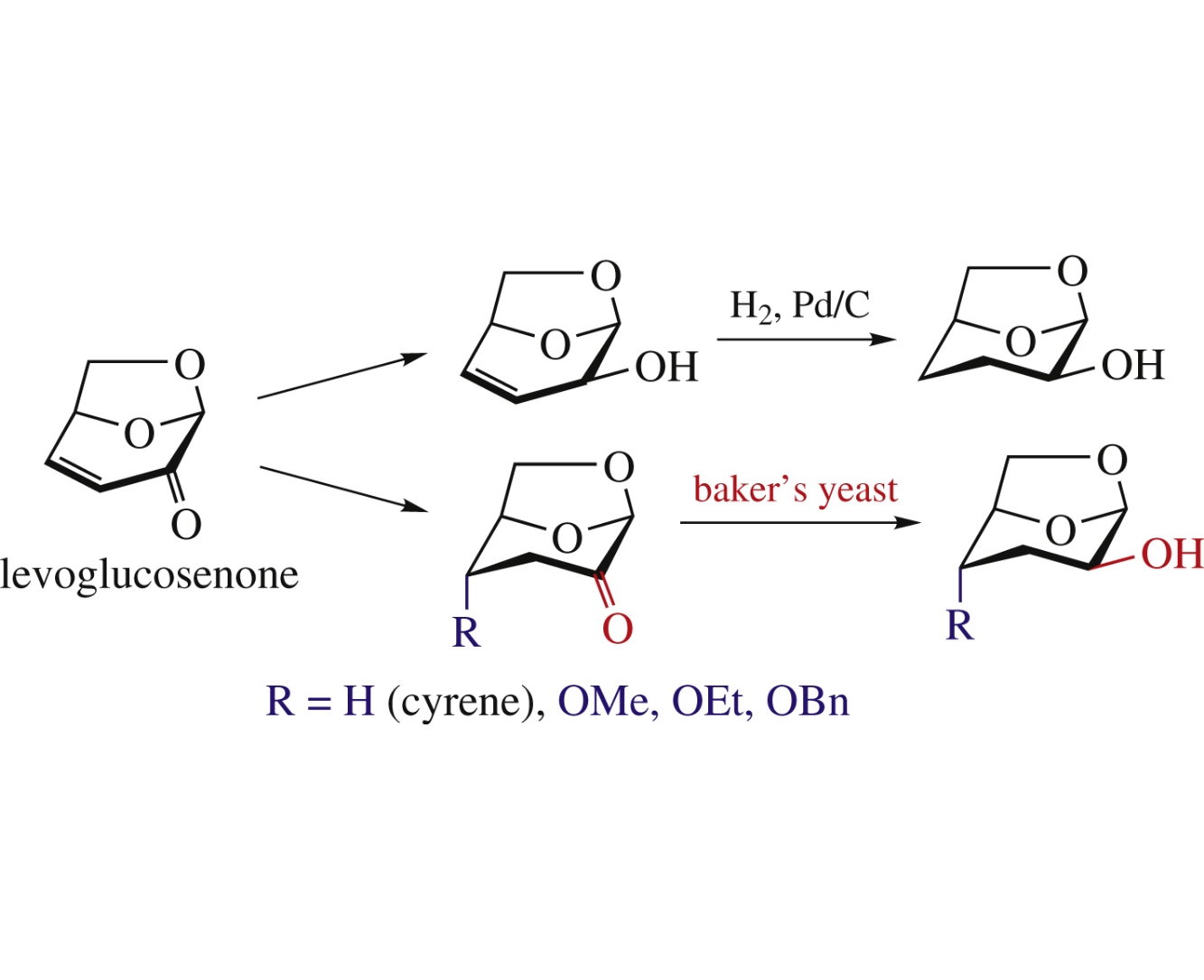
Diastereomerically pure levoglucosenone alcohol, synthesized from levoglucosenone, upon hydrogenation on Raney Ni or Pd/BaSO4 undergoes epimerization at C2 atom caused by formation of cyrene by-product and its subsequent non-specific reduction. A microbiological stereospecific technique using baker's yeast (Saccharomyces cerevisiae) has been developed levoglucosenone for cyrene reduction into cyrene alcohol and also applied to reduce 4-alkoxy and 4-benzyloxy derivatives of cyrene.
References
1.

Miftakhov M.S., Valeev F.A., Gaisina I.N.
Russian Chemical Reviews,
1994
2.

M. Sarotti A., M. Zanardi M., A. Spanevello R.
Current Organic Synthesis,
2012
3.

Comba M.B., Tsai Y., Sarotti A.M., Mangione M.I., Suárez A.G., Spanevello R.A.
European Journal of Organic Chemistry,
2017
4.
Sharipov B.T., Pershin A.A., Valeev F.A.
Mendeleev Communications,
2017
5.

Stockton K.P., Greatrex B.W.
Organic and Biomolecular Chemistry,
2016
6.

Zanardi M.M., Suárez A.G.
Tetrahedron Letters,
2009
7.

Highly Diastereoselective Diels−Alder Reaction Using a Chiral Auxiliary Derived from Levoglucosenone
Sarotti A.M., Spanevello R.A., Suárez A.G.
Organic Letters,
2006
8.

Sarotti A.M., Fernández I., Spanevello R.A., Sierra M.Á., Suárez A.G.
Organic Letters,
2008
9.

Sarotti A.M., Spanevello R.A., Suárez A.G.
Tetrahedron,
2009
10.

Greatrex B., Klepp J., Sumby C.
Synlett,
2018
11.

Zanardi M.M., Botta M.C., Suárez A.G.
Tetrahedron Letters,
2014
12.

Sherwood J., De bruyn M., Constantinou A., Moity L., McElroy C.R., Farmer T.J., Duncan T., Raverty W., Hunt A.J., Clark J.H.
Chemical Communications,
2014
13.

Alves Costa Pacheco A., Sherwood J., Zhenova A., McElroy C.R., Hunt A.J., Parker H.L., Farmer T.J., Constantinou A., De bruyn M., Whitwood A.C., Raverty W., Clark J.H.
ChemSusChem,
2016
14.

Zhang J., White G.B., Ryan M.D., Hunt A.J., Katz M.J.
ACS Sustainable Chemistry and Engineering,
2016
15.

Wilson K.L., Kennedy A.R., Murray J., Greatrex B., Jamieson C., Watson A.J.
Beilstein Journal of Organic Chemistry,
2016
16.

Watson A., Wilson K., Murray J., Jamieson C.
Synlett,
2017
17.
Halbych J., Trnka T., Černý M.
Collection of Czechoslovak Chemical Communications,
2012
18.
Pecka J., Staněk J., Černý M.
Collection of Czechoslovak Chemical Communications,
2012
19.
Pecka J., Černý M.
Collection of Czechoslovak Chemical Communications,
2012
20.

Giri G.F., Viarengo G., Furlán R.L., Suárez A.G., Garcia Véscovi E., Spanevello R.A.
Industrial Crops and Products,
2017
21.

Shafizadeh F., Chin P.P.
Carbohydrate Research,
1977
22.

Brimacombe J.S., Hunedy F., Tucker L.C.
Carbohydrate Research,
1978
23.

Jung M.E., Kiankarimi M.
Journal of Organic Chemistry,
1998
24.

Krishna S.H., McClelland D.J., Rashke Q.A., Dumesic J.A., Huber G.W.
Green Chemistry,
2017
25.

Molander G.A.
Chemical Reviews,
1992
26.

Shafizadeh F., Furneaux R.H., Stevenson T.T.
Carbohydrate Research,
1979
27.

KLEIDERER E.C., KORNFELD E.C.
Journal of Organic Chemistry,
1948
28.

29.

Odinokov V.N., Shafikov R.V., Savchenko R.G., Afon’kina S.R., Galyautdinov I.V., Khalilov L.M., Shashkov A.S.
Russian Journal of Organic Chemistry,
2008
30.

Fantin G., Fogagnolo M., Guerzoni M.E., Medici A., Pedrini P., Poli S.
Journal of Organic Chemistry,
1994
31.

Wipf B., Kupfer E., Bertazzi R., Leuenberger H.G.
Helvetica Chimica Acta,
1983
32.
![Enantioselective reduction of σ-Symmetric bicyclo[3.3.0]octane-2,8-diones with baker's yeast](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Inoue T., Hosomi K., Araki M., Nishide K., Node M.
Tetrahedron Asymmetry,
1995
33.

Kawai T., Isobe M., Peters S.
Australian Journal of Chemistry,
1995
34.

Furneaux R.H., Gainsford G.J., Shafizadeh F., Stevenson T.T.
Carbohydrate Research,
1986