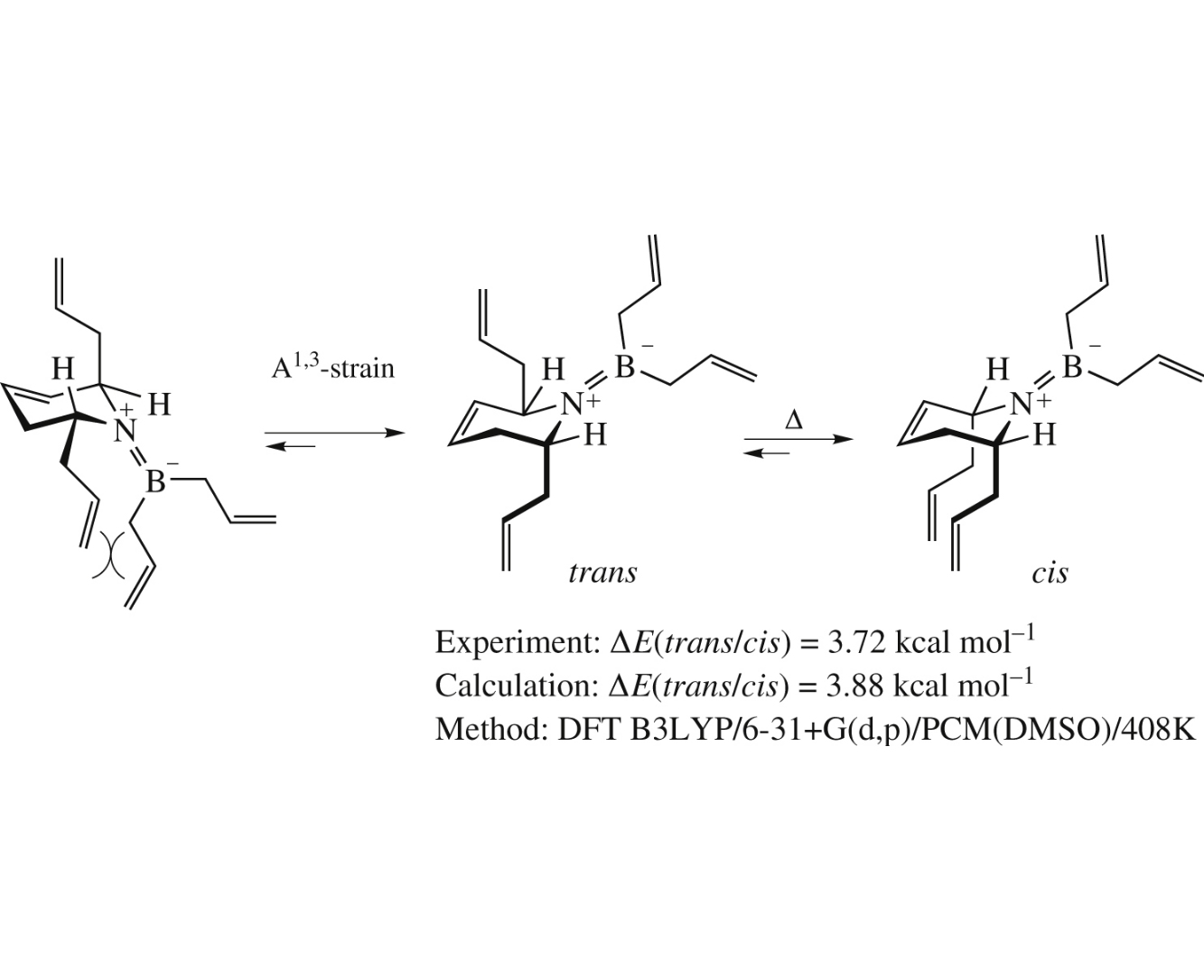
Abstract
Triallylborane-mediated thermal trans/cis-isomerization of α-allylated azaheterocycles is a unique stereoselective transformation providing straightforward access to important heterocycles. The main experimental features of this process, namely, thermodynamically controlled isomer ratio, 1,3-allylic strain as a driving force and regioselectivity are quantitatively described by quantum chemical calculations at B3LYP/6-31+G(d,p)/PCM(DMSO) level of theory.
References
1.
10.1016/j.mencom.2019.03.025_bib0005
Bubnov
Sci. Synth.,
2004
2.
10.1016/j.mencom.2019.03.025_bib0010
Hall
Allylboration of Carbonyl Compounds,
2012
3.

Diner C., Szabó K.J.
Journal of the American Chemical Society,
2016
4.

Huo H., Duvall J.R., Huang M., Hong R.
Organic Chemistry Frontiers,
2014
5.
10.1016/j.mencom.2019.03.025_bib0025
Chiral Amine Synthesis: Methods, Developments and Applications,
2010
6.

Vitaku E., Smith D.T., Njardarson J.T.
Journal of Medicinal Chemistry,
2014
7.

Kuznetsov N.Y., Bubnov Y.N.
Russian Chemical Reviews,
2015
8.

Bubnov Y.N., Spiridonov Y.Y., Kuznetsov N.Y.
Russian Chemical Bulletin,
2018
9.

Bubnov Y.N.
Pure and Applied Chemistry,
1994
10.

Lichtenberg C., Spaniol T.P., Okuda J.
Organometallics,
2011
11.

Bubnov Y.N., Demina E.E., Ignatenko A.V.
Russian Chemical Bulletin,
1997
12.

Ramachandran P.V., Burghardt T.E.
Chemistry - A European Journal,
2005
13.

Bubnov Y.N., Klimkina E.V., Ignatenko A.V.
Russian Chemical Bulletin,
1998
14.

Kuznetsov N.Y., Khrustalev V.N., Godovikov I.A., Bubnov Y.N.
European Journal of Organic Chemistry,
2006
15.

Hoffmann R.W.
Chemical Reviews,
1989
16.

Deiters A., Martin S.F.
Chemical Reviews,
2004
17.
10.1016/j.mencom.2019.03.025_bib0085
Schrödinger: Maestro,
2016
18.
10.1016/j.mencom.2019.03.025_bib0090
Frisch
Gaussian 09, Revision D.01,
2009
19.

Stephens P.J., Devlin F.J., Chabalowski C.F., Frisch M.J.
The Journal of Physical Chemistry,
1994
20.

Becke A.D.
Journal of Chemical Physics,
1993
21.

Hehre W.J., Ditchfield R., Pople J.A.
Journal of Chemical Physics,
1972
22.

Mennucci B.
Wiley Interdisciplinary Reviews: Computational Molecular Science,
2012
23.

Mardirossian N., Head-Gordon M.
Molecular Physics,
2017
24.

Medvedev M.G., Bushmarinov I.S., Sun J., Perdew J.P., Lyssenko K.A.
Science,
2017
25.
Marjewski A.A., Medvedev M.G., Gerasimov I.S., Panova M.V., Perdew J.P., Lyssenko K.A., Dmitrienko A.O.
Mendeleev Communications,
2018
26.

Grimme S., Antony J., Ehrlich S., Krieg H.
Journal of Chemical Physics,
2010
27.

Jensen F.
Journal of Chemical Theory and Computation,
2018
28.

Plumley J.A., Dannenberg J.J.
Journal of Computational Chemistry,
2011
29.

Kuznetsov N.Y., Lyssenko K.A., Peregudov A.S., Bubnov Y.N.
Russian Chemical Bulletin,
2007
30.
Medvedev M.G., Panova M.V., Chilov G.G., Bushmarinov I.S., Novikov F.N., Stroganov O.V., Zeifman A.A., Svitanko I.V.
Mendeleev Communications,
2017
31.

32.

Medvedev M.G., Zeifman A.A., Novikov F.N., Bushmarinov I.S., Stroganov O.V., Titov I.Y., Chilov G.G., Svitanko I.V.
Journal of the American Chemical Society,
2017
33.

Cruchter T., Medvedev M.G., Shen X., Mietke T., Harms K., Marsch M., Meggers E.
ACS Catalysis,
2017
34.

Bubnov Y.N., Demina E.E., Ignatenko A.V.
Russian Chemical Bulletin,
1997