Abstract
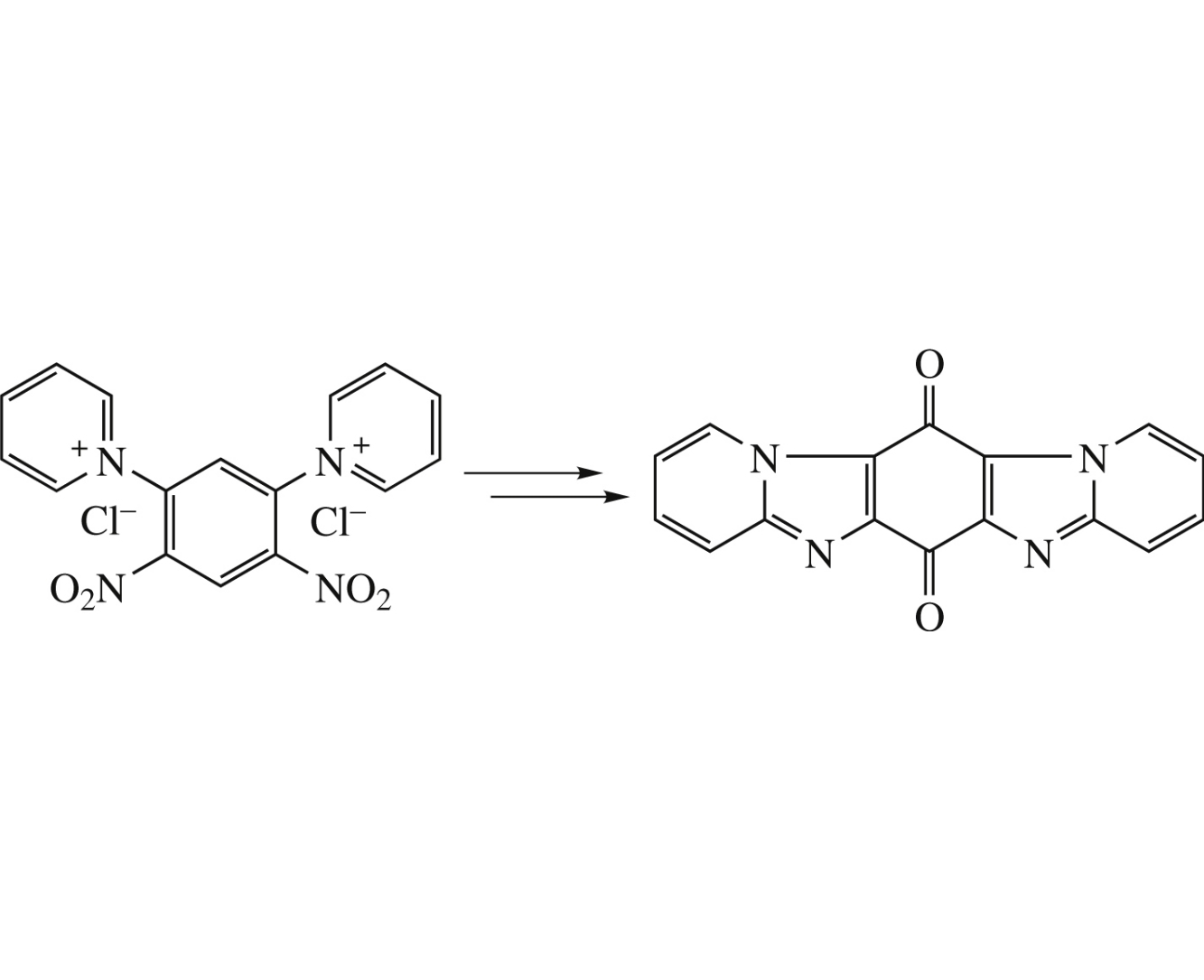
Reduction of 1,1′-(4,6-dinitro-1,3-phenylene)dipyridinium dichloride with SnCl2 afforded dipyrido[1,2-a:1′,2′-a′]benzo[1,2-d:5,4-d]diimidazole which was converted into the corresponding quinone-type 6,13-dione in three simple steps.
References
1.
![The phosphorescent and magnetic properties of a novel radical and its salt derived from 2,3′-biimidazo[1,2-a]pyridin-2′-one radical](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Zhao Y., Yong G., Feng Y., Shen C.
Synthetic Metals,
2014
2.

Zhang Y., Hanifi D., Lim E., Chourou S., Alvarez S., Pun A., Hexemer A., Ma B., Liu Y.
Advanced Materials,
2013
3.

Schönamsgruber J., Hirsch A.
European Journal of Organic Chemistry,
2015
4.

Xie J., Chen W., Wang Z., Jie K.C., Liu M., Zhang Q.
Chemistry - An Asian Journal,
2017
5.

6.

Seminario J.M., De La Cruz C., Derosa P.A., Yan L.
Journal of Physical Chemistry B,
2004
7.

Zhang Q., Bu X., Lin Z., Biasini M., Beyermann W.P., Feng P.
Inorganic Chemistry,
2007
8.

Meng X., Song Y., Hou H., Fan Y., Li G., Zhu Y.
Inorganic Chemistry,
2003
9.

Kitagawa S., Kitaura R., Noro S.
Angewandte Chemie - International Edition,
2004
10.

Lan A., Li K., Wu H., Olson D., Emge T., Ki W., Hong M., Li J.
Angewandte Chemie - International Edition,
2009
11.

Wang H., Cheng J., Ma J., Dong Y., Huang R.
Inorganic Chemistry,
2010
12.

Liu Q., Ma J., Dong Y.
Journal of the American Chemical Society,
2010
13.
10.1016/j.mencom.2019.03.023_bib0065
Soni
Indian J. Chem.,
1979
14.

Zincke T.
Journal für praktische Chemie,
1910
15.
![Synthesis of pyrido[1,2-a]benzimidazoles and other fused imidazole derivatives with a bridgehead nitrogen atom](/storage/images/resized/9Mus3KG1Tkd7Bwaurt8H3RwWh0CxRlGoO6ng9UK1_small_thumb.webp)
Begunov R.S., Ryzvanovich G.A.
Russian Chemical Reviews,
2013
16.
Sokolov A.A., Syroeshkin M.A., Begunov R.S., Rusakova N.N., Gultyai V.P.
Mendeleev Communications,
2012
17.

Begunov R.S., Sokolov A.A., Shebunina T.V.
Russian Journal of Organic Chemistry,
2013
18.
![Efficient electrochemical synthesis of pyrido[1,2-a]benzimidazoles](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Sokolov A.A., Syroeshkin M.A., Solkan V.N., Shebunina T.V., Begunov R.S., Mikhal’chenko L.V., Leonova M.Y., Gultyai V.P.
Russian Chemical Bulletin,
2014
19.
10.1016/j.mencom.2019.03.023_bib0095
Frolov
J. Gen. Chem. USSR (Engl. Transl.),
1992
20.
10.1016/j.mencom.2019.03.023_bib0100
Prostakov
J. Org. Chem. USSR (Engl. Transl.),
1990
21.
10.1016/j.mencom.2019.03.023_bib0105
Ezema
Chem. Process Eng. Res.,
2013
22.

Parrick J., Ragunathan R.
Journal of the Chemical Society Perkin Transactions 1,
1993
23.

Potts K.T., Bhattacharjee D., Walsh E.B.
Journal of Organic Chemistry,
1986
24.

Joseph P.K., Joullié M.M.
Journal of Medicinal Chemistry,
1964
25.
10.1016/j.mencom.2019.03.023_bib0125
Chowdhury
Indian J. Chem. Technol.,
2005
26.

Ayad M.M., Shenashin M.A.
European Polymer Journal,
2004
27.

Kumar S., Verma R., Gangadharan S.
The Analyst,
1993
28.

Sayyah S.M., Abd El-Khalek A.A., Bahgat A.A., Abd El-Salam H.M.
International Journal of Polymeric Materials and Polymeric Biomaterials,
2001
29.

Cihaner A., Önal A.M.
European Polymer Journal,
2001
30.
10.1016/j.mencom.2019.03.023_bib0150
Olah
Nitration. Methods and Mechanisms,
1989
31.
![Reaction of substituted pyrido[1,2-a]benzimidazoles with electrophilic agents](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Begunov R.S., Sokolov A.A., Belova V.O., Fakhrutdinov A.N., Shashkov A.S., Fedyanin I.V.
Tetrahedron Letters,
2015