Abstract
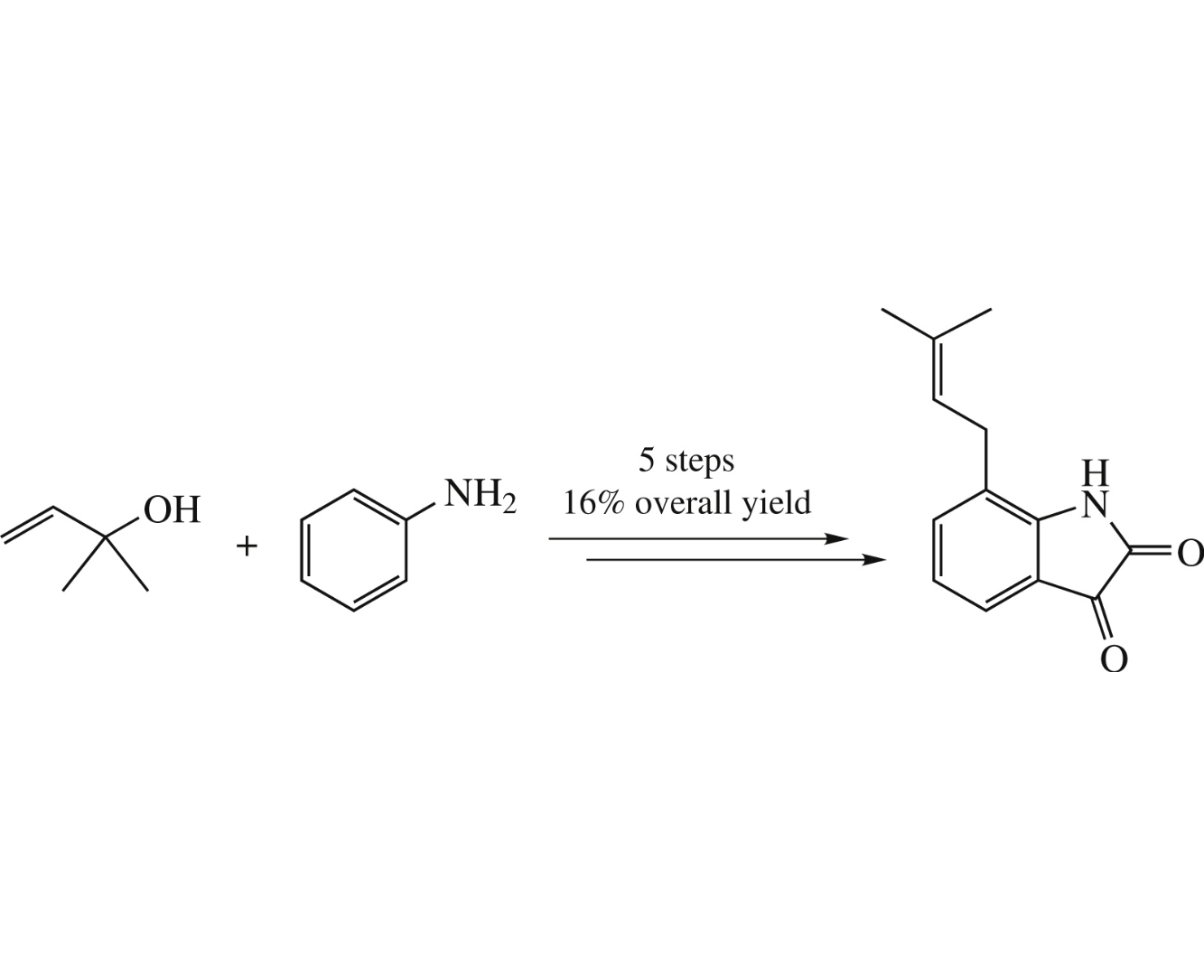
The first synthesis of 7-prenylisatin, an isatin-type antibiotic with prominent activity against Bacillus subtilis, has been accomplished in 16% overall yield from 2-methylbut-3-en-2-ol and aniline via a five-step procedure. The protocol includes the aromatic aza-Claisen rearrangement and the Sandmeyer isonitrosoacetanilide isatin synthesis as key steps.
References
1.

Newman D.J., Cragg G.M., Snader K.M.
Journal of Natural Products,
2003
2.
D’yakonov V.A., Makarov A.A., Dzhemileva L.U., Andreev E.N., Dzhemilev U.M.
Mendeleev Communications,
2017
3.
Vostrikov N.S., Lobko I.F., Spirikhin L.V., Vakhitova Y.V., Pivnitsky K.K., Miftakhov M.S.
Mendeleev Communications,
2017
4.
Gruzdev D.A., Chulakov E.N., Levit G.L., Kravchenko M.A., Krasnov V.P., Charushin V.N.
Mendeleev Communications,
2017
5.
Pazynina G.V., Tsygankova S.V., Ryzhov I.M., Paramonov A.S., Nicolai, Bovin V.
Mendeleev Communications,
2018
6.

Newman D.J., Cragg G.M.
Journal of Natural Products,
2012
7.

Wu C., Du C., Gubbens J., Choi Y.H., van Wezel G.P.
Journal of Natural Products,
2015
8.
10.1016/j.mencom.2019.03.018_bib0040
Vine
Chem.,
2009
9.
Breinholt J., Demuth H., Heide M., Jensen G.W., Møller I.L., Nielsen R.I., Olsen C.E., Rosendahl C.N., Haugg M., Trabesinger-Rüf N., Weinhold E.G.
Acta Chemica Scandinavica,
1996
10.

GRÄFE U., RADICS L.
Journal of Antibiotics,
1986
11.

Ruchti J., Carreira E.M.
Journal of the American Chemical Society,
2014
12.

Cooper M.A., Lucas M.A., Taylor J.M., Ward A.D., Williamson N.M.
Synthesis,
2001
13.

Beare K.D., McErlean C.S.
Organic and Biomolecular Chemistry,
2013
14.

Alt I.T., Guttroff C., Plietker B.
Angewandte Chemie - International Edition,
2017
15.

Xu H., He J., Shi J., Tan L., Qiu D., Luo X., Li Y.
Journal of the American Chemical Society,
2018
16.

Sandmeyer T.
Helvetica Chimica Acta,
1919
17.

Lisowski V., Robba M., Rault S.
Journal of Organic Chemistry,
2000
18.

Lin S., Yang Z., Kwok B.H., Koldobskiy M., Crews C.M., Danishefsky S.J.
Journal of the American Chemical Society,
2004
19.

Ferandin Y., Bettayeb K., Kritsanida M., Lozach O., Polychronopoulos P., Magiatis P., Skaltsounis A., Meijer L.
Journal of Medicinal Chemistry,
2006