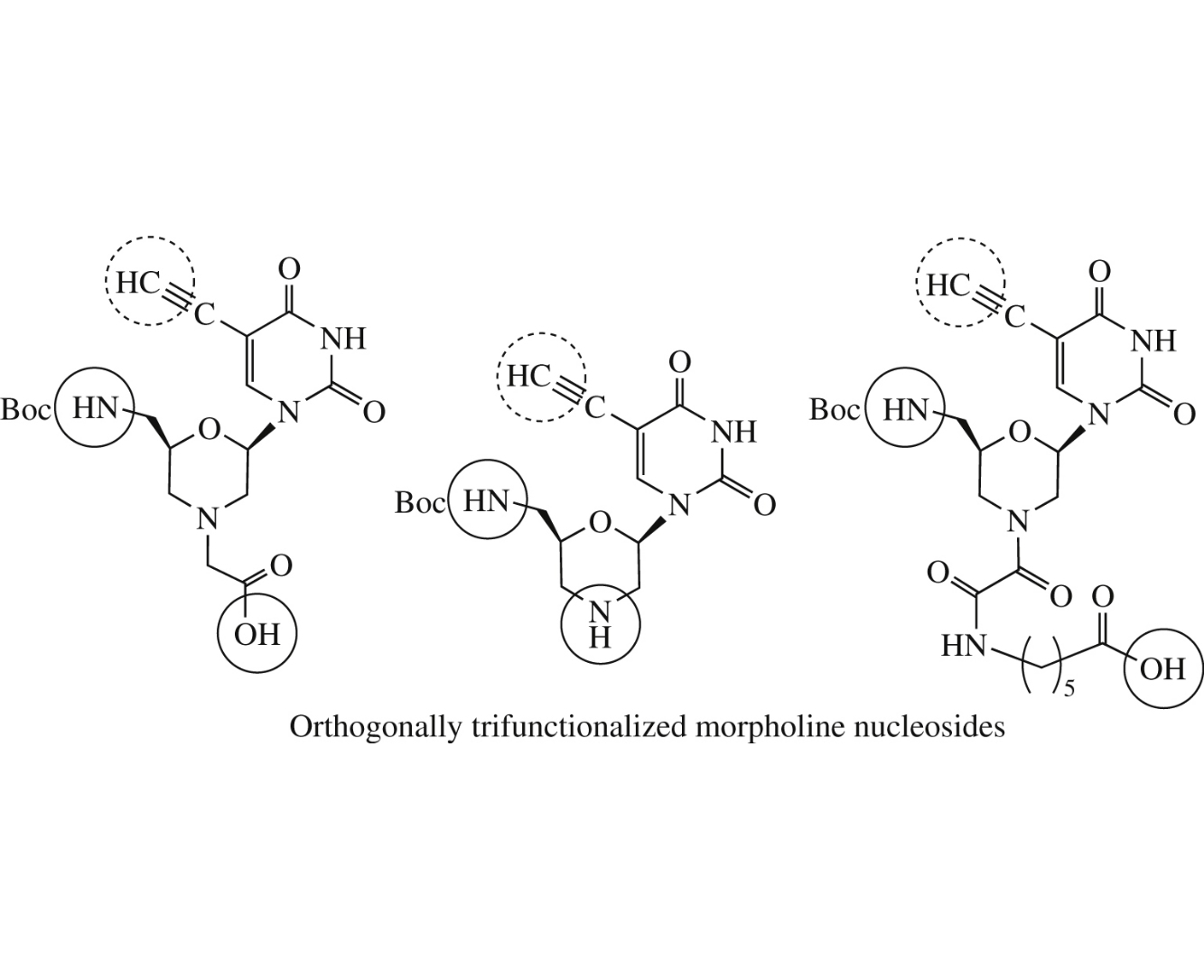
Abstract
Three novel uracil-based morpholine nucleosides each containing three different orthogonally reactive functional groups, namely, amino, carboxy and acetylene ones, were synthesized. The obtained monomers are intended for the synthesis of labeled nucleotide, nucleic acid or peptide mimics.
References
1.

Kharitonova M.I., Konstantinova I.D., Miroshnikov A.I.
Russian Chemical Reviews,
2018
2.
10.1016/j.mencom.2019.03.017_bib0010
Summerton
2017
3.

Aartsma-Rus A., Krieg A.M.
Nucleic Acid Therapeutics,
2016
4.

Aartsma-Rus A.
Nucleic Acid Therapeutics,
2017
5.

Abramova T.V., Belov S.S., Tarasenko Y.V., Silnikov V.N.
Beilstein Journal of Organic Chemistry,
2014
6.

Golyshev V.M., Abramova T.V., Pyshnyi D.V., Lomzov A.A.
Biophysical Chemistry,
2018
7.

Sherstyuk Y.V., Zakharenko A.L., Kutuzov M.M., Sukhanova M.V., Lavrik O.I., Silnikov V.N., Abramova T.V.
Russian Journal of Bioorganic Chemistry,
2017
8.

Schneider A.F., Hackenberger C.P.
Current Opinion in Biotechnology,
2017
9.

Jones C.E., Berliner L.J.
Cell Biochemistry and Biophysics,
2016
10.

Calce E., De Luca S.
Chemistry - A European Journal,
2016
11.

Liu Y., Hou W., Sun H., Cui C., Zhang L., Jiang Y., Wu Y., Wang Y., Li J., Sumerlin B.S., Liu Q., Tan W.
Chemical Science,
2017
12.

Kölmel D.K., Kool E.T.
Chemical Reviews,
2017
13.
10.1016/j.mencom.2019.03.017_bib0065
Obermaier
2015
14.
10.1016/j.mencom.2019.03.017_bib0070
Lang
2015
15.

Ingale S.A., Leonard P., Yang H., Seela F.
Organic and Biomolecular Chemistry,
2014
16.

Cavanagh B.L., Walker T., Norazit A., Meedeniya A.C.
Molecules,
2011
17.

Knall A., Hollauf M., Saf R., Slugovc C.
Organic and Biomolecular Chemistry,
2016
18.

McKay C., Finn M.G.
Chemistry & Biology,
2014
19.

20.

Lovitt C.J., Hilko D.H., Avery V.M., Poulsen S.
Bioorganic and Medicinal Chemistry,
2016
21.

Paul S., Nandi B., Pattanayak S., Sinha S.
Tetrahedron Letters,
2012
22.

Nandi B., Pattanayak S., Paul S., Sinha S.
European Journal of Organic Chemistry,
2013
23.

Vohtancev I.P., Sherstyuk Y.V., Silnikov V.N., Abramova T.V.
Organic Preparations and Procedures International,
2018
24.

Robins M.J., Barr P.J., Giziewicz J.
Canadian Journal of Chemistry,
1982
25.

Kasakin M.F., Abramova T.V., Silnikov V.N.
Russian Journal of Bioorganic Chemistry,
2011
26.

Mungall W.S., Greene G.L., Heavner G.A., Letsinger R.L.
Journal of Organic Chemistry,
1975
27.

Mondal S., Manna D., Mugesh G.
Angewandte Chemie - International Edition,
2015
28.

Schinazi R.F., Chen M.S., Prusoff W.H.
Journal of Medicinal Chemistry,
1979
29.

Lin T., Neenan J.P., Cheng Y., Prusoff W.H., Ward D.C.
Journal of Medicinal Chemistry,
1976
30.

McGuigan C., Pathirana R.N., Snoeck R., Andrei G., De Clercq E., Balzarini J.
Journal of Medicinal Chemistry,
2004
31.

Garg S., Shakya N., Srivastav N.C., Agrawal B., Kunimoto D.Y., Kumar R.
Bioorganic and Medicinal Chemistry,
2016
32.

Gritsenko O.M., Gromova E.S.
Russian Chemical Reviews,
1999
33.

Tarasenko Y.V., Abramova T.V., Mamatuk V.I., Silnikov V.N.
Nucleosides, Nucleotides and Nucleic Acids,
2016
34.
10.1016/j.mencom.2019.03.017_bib0170
Talhaoui
Nucleic Acids Res.,
2016
35.

Langelier M., Zandarashvili L., Aguiar P.M., Black B.E., Pascal J.M.
Nature Communications,
2018