Abstract
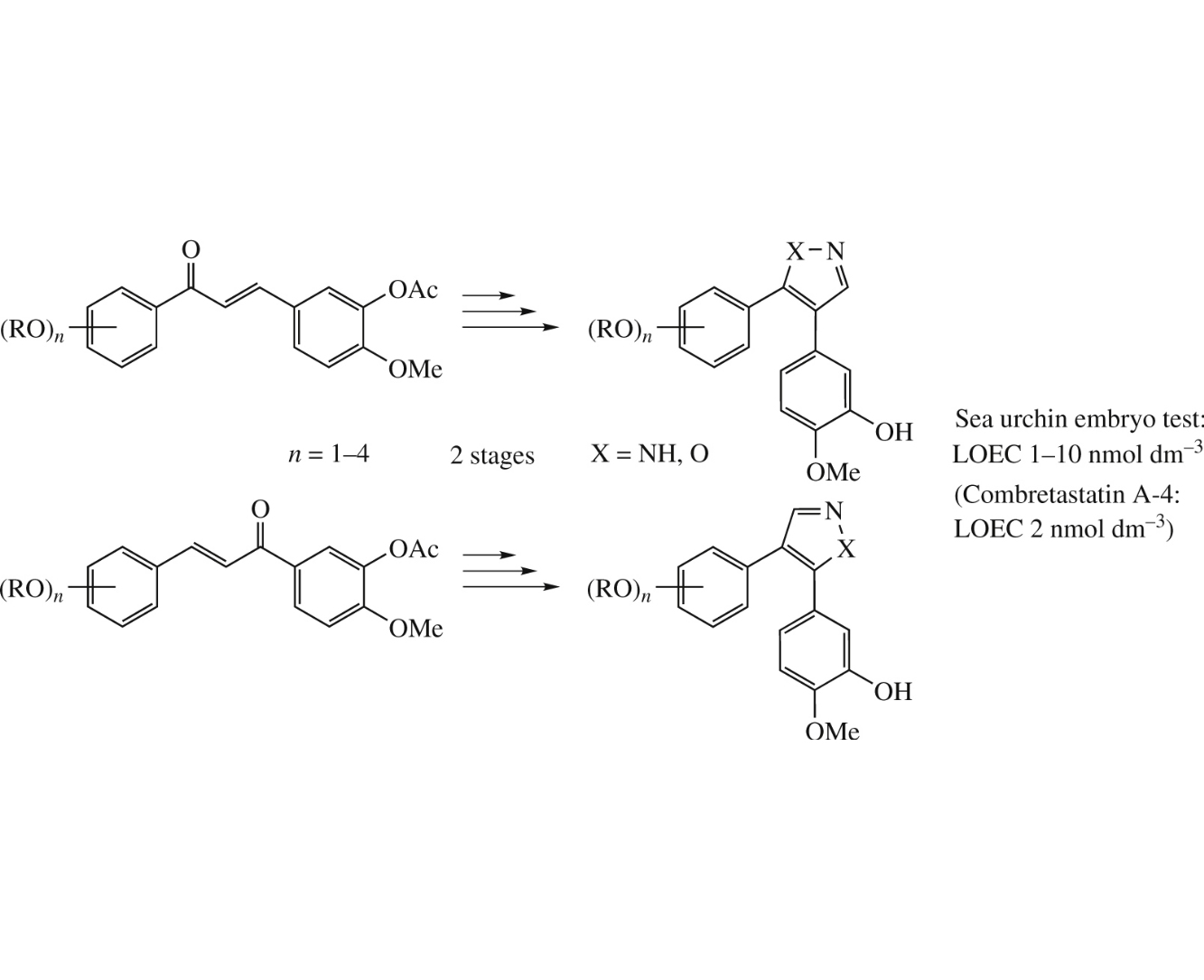
A series of combretastatin analogues, diarylpyrazoles and diarylisoxazoles, have been synthesized and evaluated for their antimitotic tubulin-binding activity using the phenotypic sea urchin (Paracentrotus lividus) embryo assay. One pyrazole analogue and four isoxazole analogues have been identified as potent antimitotic agents comparable with combretastatins A-2 and A-4, with the lowest observable effective concentration of 1–10nmoldm−3 for cleavage alteration of the test embryos.
References
1.

Cushman M., Nagarathnam D., Gopal D., Chakraborti A.K., Lin C.M., Hamel E.
Journal of Medicinal Chemistry,
1991
2.

Pettit G.R., Rhodes M.R., Herald D.L., Hamel E., Schmidt J.M., Pettit R.K.
Journal of Medicinal Chemistry,
2005
3.

Semenova M.N., Demchuk D.V., Tsyganov D.V., Chernysheva N.B., Samet A.V., Silyanova E.A., Kislyi V.P., Maksimenko A.S., Varakutin A.E., Konyushkin L.D., Raihstat M.M., Kiselyov A.S., Semenov V.V.
ACS Combinatorial Science,
2018
4.

Brown A.W., Fisher M., Tozer G.M., Kanthou C., Harrity J.P.
Journal of Medicinal Chemistry,
2016
5.

Sun C., Lin L., Yu H., Cheng C., Tsai Y., Chu C., Din Y., Chau Y., Don M.
Bioorganic and Medicinal Chemistry Letters,
2007
6.

Kaffy J., Pontikis R., Carrez D., Croisy A., Monneret C., Florent J.
Bioorganic and Medicinal Chemistry,
2006
7.

Mikstacka R., Stefański T., Różański J.
Cellular and Molecular Biology Letters,
2013
8.

Rajak H., Kumar Dewangan P., Patel V., Kumar Jain D., Singh A., Veerasamy R., Chander Sharma P., Dixit A.
Current Pharmaceutical Design,
2013
9.

Demchuk D.V., Samet A.V., Chernysheva N.B., Ushkarov V.I., Stashina G.A., Konyushkin L.D., Raihstat M.M., Firgang S.I., Philchenkov A.A., Zavelevich M.P., Kuiava L.M., Chekhun V.F., Blokhin D.Y., Kiselyov A.S., Semenova M.N., et. al.
Bioorganic and Medicinal Chemistry,
2014
10.

Tsyganov D.V., Khrustalev V.N., Konyushkin L.D., Raihstat M.M., Firgang S.I., Semenov R.V., Kiselyov A.S., Semenova M.N., Semenov V.V.
European Journal of Medicinal Chemistry,
2014
11.

Tron G.C., Pirali T., Sorba G., Pagliai F., Busacca S., Genazzani A.A.
Journal of Medicinal Chemistry,
2006
12.

Jaroch K., Karolak M., Górski P., Jaroch A., Krajewski A., Ilnicka A., Sloderbach A., Stefański T., Sobiak S.
Pharmacological Reports,
2016
13.

Chernysheva N.B., Maksimenko A.S., Andreyanov F.A., Kislyi V.P., Strelenko Y.A., Khrustalev V.N., Semenova M.N., Semenov V.V.
European Journal of Medicinal Chemistry,
2018
14.

Nam N.
Current Medicinal Chemistry,
2003
15.

Schneider W., Kraft E.
Berichte der deutschen chemischen Gesellschaft (A and B Series),
1922
16.
Chernyshova N.B., Tsyganov D.V., Khrustalev V.N., Raihstat M.M., Konyushkin L.D., Semenov R.V., Semenova M.N., Semenov V.V.
Arkivoc,
2017
17.

Domínguez E., Ibeas E., Martínez de Marigorta E., Palacios J.K., SanMartín R.
Journal of Organic Chemistry,
1996