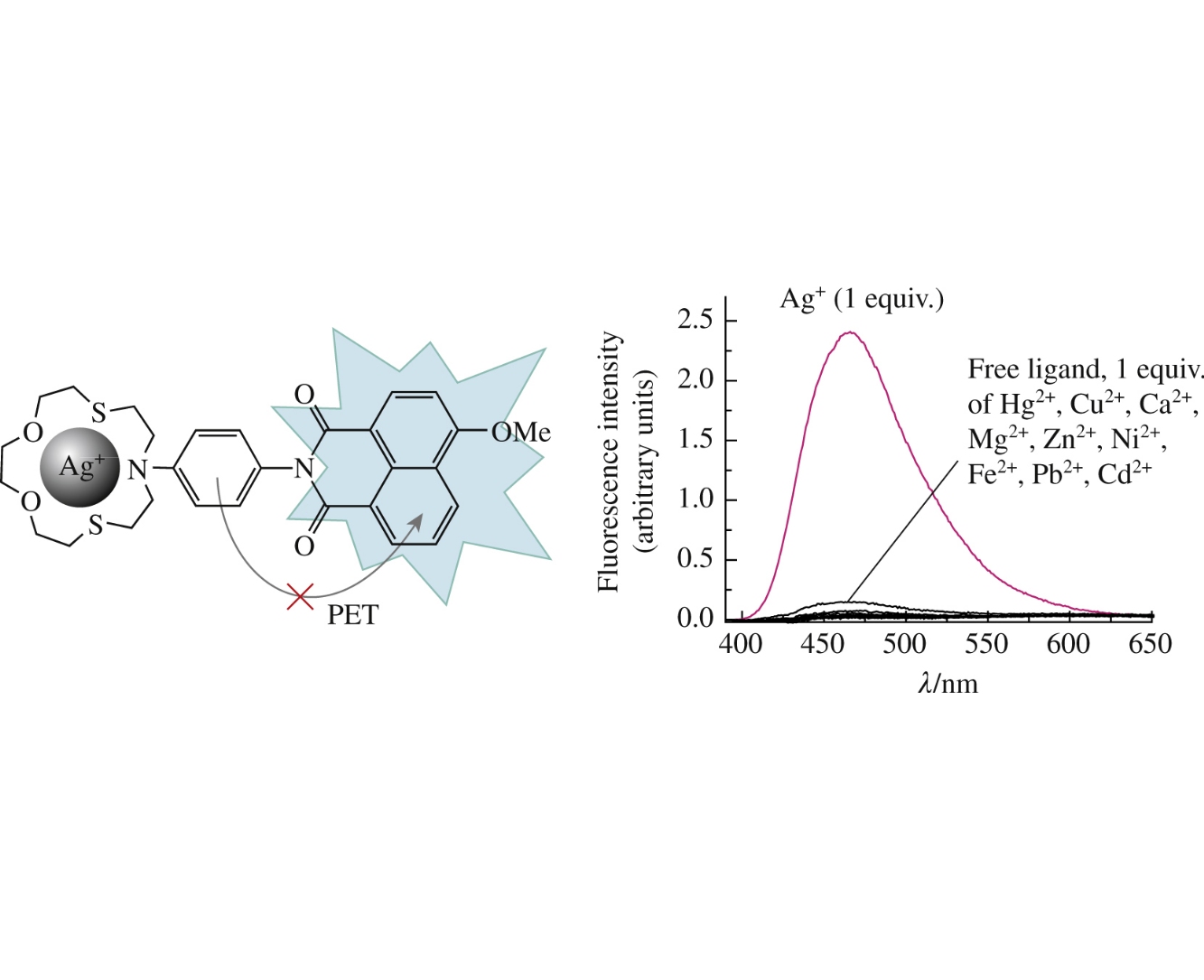
Abstract
A novel derivative of 4-methoxy-1,8-naphthalimide bearing N-phenylazadithia-15-crown-5 ether receptor has been demonstrated as the selective and sensitive fluorescent probe for the detection of silver(I) ions in purely aqueous solution at neutral pH.
References
1.

Chen C., Liu H., Zhang B., Wang Y., Cai K., Tan Y., Gao C., Liu H., Tan C., Jiang Y.
Tetrahedron,
2016
2.

Lee S.Y., Bok K.H., Kim C.
RSC Advances,
2017
3.

Velmurugan K., Thamilselvan A., Antony R., Kannan V.R., Tang L., Nandhakumar R.
Journal of Photochemistry and Photobiology A: Chemistry,
2017
4.

Affrose A., Parveen S.D., Kumar B.S., Pitchumani K.
Sensors and Actuators, B: Chemical,
2015
5.

Feng Q.L., Wu J., Chen G.Q., Cui F.Z., Kim T.N., Kim J.O.
Journal of Biomedical Materials Research,
2000
6.

Formica M., Fusi V., Giorgi L., Micheloni M.
Coordination Chemistry Reviews,
2012
7.

Lee M.H., Kim J.S., Sessler J.L.
Chemical Society Reviews,
2015
8.
Gorbatov S.A., Kozlov M.A., Zlobin I.E., Kartashov A.V., Zavarzin I.V., Volkova Y.A.
Mendeleev Communications,
2018
9.

Panchenko P.A., Fedorova O.A., Fedorov Y.V.
Russian Chemical Reviews,
2014
10.
10.1016/j.mencom.2019.03.012_sbref0030a
Arulraj
Sens. Biosens. Res.,
2015
11.

Chen T., Zhu W., Xu Y., Zhang S., Zhang X., Qian X.
Dalton Transactions,
2010
12.

Park C.S., Lee J.Y., Kang E., Lee J., Lee S.S.
Tetrahedron Letters,
2009
13.

Panchenko P.A., Fedorov Y.V., Perevalov V.P., Jonusauskas G., Fedorova O.A.
Journal of Physical Chemistry A,
2010
14.
Panchenko P.A., Fedorov Y.V., Fedorova O.A., Izmailov B.A., Vasnev V.A., Istratov V.V., Makeeva E.A., Rumyantseva M.N., Gaskov A.M.
Mendeleev Communications,
2011
15.

Sergeeva A.N., Panchenko P.A., Fedorov Y.V., Fedorova O.A.
Protection of Metals and Physical Chemistry of Surfaces,
2012
16.

Panchenko P.A., Fedorov Y.V., Fedorova O.A., Jonusauskas G.
Dyes and Pigments,
2013
17.

Panchenko P.A., Park V.V., Fedorova O.A., Fedorov Y.V., Kataev E.A.
Russian Chemical Bulletin,
2015
18.

Panchenko P.A., Fedorov Y.V., Fedorova O.A., Jonusauskas G.
Physical Chemistry Chemical Physics,
2015
19.

Selector S.L., Bogdanova L.B., Shokurov A.V., Panchenko P.A., Fedorova O.A., Arslanov V.V.
Macroheterocycles,
2014
20.
![Synthesis of Potential Anticancer Agents. I. Nitrogen Mustards Derived from p-[N,N-Bis(2-chloroethyl)amino]benzaldehyde1](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
ELDERFIELD R.C., COVEY I.S., GEIDUSCHEK J.B., MEYER W.L., ROSS A.B., ROSS J.H.
Journal of Organic Chemistry,
1958
21.

Descalzo A.B., Martínez-Máñez R., Radeglia R., Rurack K., Soto J.
Journal of the American Chemical Society,
2003
22.

Jiménez D., Martínez‐Máñez R., Sancenón F., Ros‐Lis J.V., Soto J., Benito Á., García‐Breijo E.
European Journal of Inorganic Chemistry,
2005
23.
![Selective Colorimetric Detection of Hg2+ and Mg2+ with Crown Ether Substituted N-Aryl-9-aminobenzo[b]quinolizinium Derivatives](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Tian M., Ihmels H.
European Journal of Organic Chemistry,
2011
24.

Pardo A., Poyato J.M., Martin E., Camacho J.J., Reyman D., Braña M.F., Castellano J.M.
Journal of Photochemistry and Photobiology A: Chemistry,
1989
25.

Panchenko P.A., Fedorov Y.V., Fedorova O.A., Perevalov V.P., Jonusauskas G.
Russian Chemical Bulletin,
2009
26.

Szakács Z., Rousseva S., Bojtár M., Hessz D., Bitter I., Kállay M., Hilbers M., Zhang H., Kubinyi M.
Physical Chemistry Chemical Physics,
2018
27.

Panchenko P.A., Arkhipova A.N., Fedorova O.A., Fedorov Y.V., Zakharko M.A., Arkhipov D.E., Jonusauskas G.
Physical Chemistry Chemical Physics,
2017
28.
Arkhipova A.N., Panchenko P.A., Fedorov Y.V., Fedorova O.A.
Mendeleev Communications,
2017
29.

Martin E., Weigand R., Pardo A.
Journal of Luminescence,
1996
30.

Demets G.J., Triboni E.R., Alvarez E.B., Arantes G.M., Filho P.B., Politi M.J.
Spectrochimica Acta - Part A: Molecular and Biomolecular Spectroscopy,
2006
31.

Loock H., Wentzell P.D.
Sensors and Actuators, B: Chemical,
2012
32.

Park K.S., Lee J.Y., Park H.G.
Chemical Communications,
2012
33.

Ábalos T., Jiménez D., Martínez-Máñez R., Ros-Lis J.V., Royo S., Sancenón F., Soto J., Costero A.M., Gil S., Parra M.
Tetrahedron Letters,
2009
34.
![Self-assembly of a ternary architecture driven by cooperative Hg2+ ion binding between cucurbit[7]uril and crown ether macrocyclic hosts](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Chernikova E., Berdnikova D., Fedorov Y., Fedorova O., Peregudov A., Isaacs L.
Chemical Communications,
2012
35.

Jeon C.H., Lee J., Ahn S.J., Ha T.H.
Tetrahedron Letters,
2013
36.

Atilgan S., Kutuk I., Ozdemir T.
Tetrahedron Letters,
2010
37.

Panchenko P.A., Fedorov Y.V., Fedorova O.A.
Journal of Photochemistry and Photobiology A: Chemistry,
2018
38.

Walcarius A., Etienne M., Delacote C.
Analytica Chimica Acta,
2004