Abstract
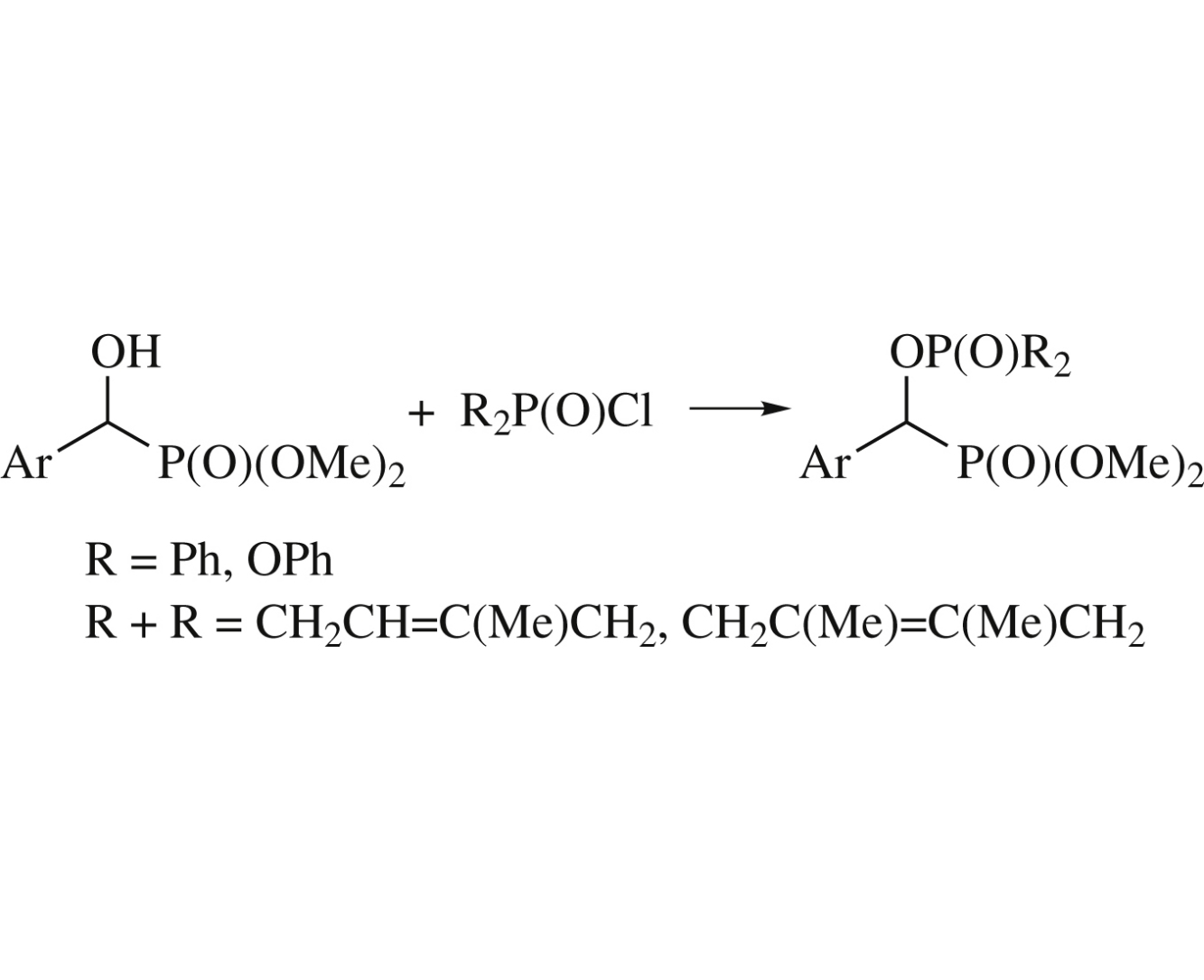
The reaction of dimethyl (1-aryl-1-hydroxymethyl)phosphonates with 1-chloro-3-phospholene 1-oxides, diphenylphosphinic chloride or diphenyl chloridophosphonate affords the corresponding (1-phosphoryloxymethyl)phosphonates. The products with two different >P(O)– moieties exhibit characteristic δP shifts and 3JP,P couplings in the 31P NMR spectra.
References
1.
10.1016/j.mencom.2019.03.011_bib0005
Tajti
Organophosphorus Chemistry: Novel Developments,,
2018
2.

Rádai Z., Szeles P., Kiss N.Z., Hegedűs L., Windt T., Nagy V., Keglevich G.
Heteroatom Chemistry,
2018
3.

Green D., Elgendy S., Patel G., Skordalakes E., Goodwin C.A., Scully M.F., Kakkar V.V., Deadman J.J.
Phosphorus, Sulfur and Silicon and the Related Elements,
2000
4.

He H., Yuan J., Peng H., Chen T., Shen P., Wan S., Li Y., Tan H., He Y., He J., Li Y.
Journal of Agricultural and Food Chemistry,
2011
5.

Synthesis and herbicidal activity of O,O-dialkyl phenoxyacetoxyalkylphosphonates containing fluorine
Chen T., Shen P., Li Y., He H.
Journal of Fluorine Chemistry,
2006
6.

Long Q., Deng X., Gao Y., Xie H., Peng H., He H.
Phosphorus, Sulfur and Silicon and the Related Elements,
2013
7.
He H., Li M., Joshi R., Meng L.
1970
8.

Peng H., Long Q., Deng X., He H.
Phosphorus, Sulfur and Silicon and the Related Elements,
2013
9.

Wang W., He H., Zuo N., Zhang X., Lin J., Chen W., Peng H.
Journal of Fluorine Chemistry,
2012
10.

Wang W., He H., Zuo N., He H., Peng H., Tan X.
Journal of Agricultural and Food Chemistry,
2012
11.

Wang T., Wang W., Peng H., He H.
Chemical Research in Chinese Universities,
2013
12.
![Synthesis and herbicidal activity of α-[(substituted phenoxybutyryloxy or valeryoxy)]alkylphosphonates and 2-(substituted phenoxybutyryloxy)alkyl-5,5-dimethyl-1,3,2-dioxaphosphinan-2-one containing fluorine](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Wang W., Zhou Y., Peng H., He H., Lu X.
Journal of Fluorine Chemistry,
2017
13.

Chen X., Shi D.
Phosphorus, Sulfur and Silicon and the Related Elements,
2008
14.
![Synthesis and insecticidal activities of O,O-dialkyl-2-[3-bromo-1-(3-chloropyridin-2-yl)-1H-pyrazole-5-carbonyloxy] (aryl) methylphosphonates](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Wang W., Wang L., Ning B., Mao M., Xue C., Wang H.
Phosphorus, Sulfur and Silicon and the Related Elements,
2016
15.

Yang J., Ma J., Che W., Li M., Li G., Song B.
Chinese Journal of Organic Chemistry,
2015
16.

Wang T., He H.W.
Synthetic Communications,
2004
17.

Iranpoor N., Firouzabadi H., Sobhani S., Amoozgar Z.
Synthesis,
2004
18.

Firouzabadi H., Iranpoor N., Farahi S.
Journal of Molecular Catalysis A Chemical,
2008
19.

Iranpoor N., Firouzabadi H., Sobhani S., Amoozgar Z.
Synthesis,
2004
20.

Jin C., He H.
Phosphorus, Sulfur and Silicon and the Related Elements,
2011
21.

The First Mitsunobu Protocol for Efficient Synthesis of α-Acyloxyphosphonates Using 4,4′-Azopyridine
Iranpoor N., Firouzabadi H., Khalili D.
Phosphorus, Sulfur and Silicon and the Related Elements,
2011
22.

Xu L., You G., Peng H., He H.
Phosphorus, Sulfur and Silicon and the Related Elements,
2014
23.

Kaboudin B., Emadi S., Faghihi M.R., Fallahi M., Sheikh-Hasani V.
Journal of Enzyme Inhibition and Medicinal Chemistry,
2012
24.
10.1016/j.mencom.2019.03.011_bib0120
Li
S. Afr. J. Chem.,
2008
25.

Creary X., Geiger C.C., Hilton K.
Journal of the American Chemical Society,
1983
26.

Li Z., Sun H., Wang Q., Huang R.
Heteroatom Chemistry,
2003
27.

Hu L., Lu S., Yang F., Feng J., Liu Z., Xu H., He H.
Phosphorus, Sulfur and Silicon and the Related Elements,
2002
28.

Kong D., Li G., Liu R.
Asian Journal of Chemistry,
2014
29.

Davidson R.S., Sheldon R.A., Trippett S.
Journal of the Chemical Society C Organic,
1967
30.

Ruel R., Bouvier J., Young R.N.
Journal of Organic Chemistry,
1995
31.

Grün A., Molnár I.G., Bertók B., Greiner I., Keglevich G.
Heteroatom Chemistry,
2009
32.

Keglevich G., Grün A., Molnár I.G., Greiner I.
Heteroatom Chemistry,
2011
33.

Årstad E., Skattebøl L.
Tetrahedron Letters,
2002
34.

Zhou J., Chen R., Yang X.
Heteroatom Chemistry,
1998
35.
10.1016/j.mencom.2019.03.011_bib0175
Kiss
Synthesis,
2013
36.

Keglevich G., Rádai Z., Kiss N.Z.
Green Processing and Synthesis,
2017
37.

Keglevich G., Petnehazy I., Miklos P., Almasy A., Toth G., Toke L., Quin L.D.
Journal of Organic Chemistry,
1987