Abstract
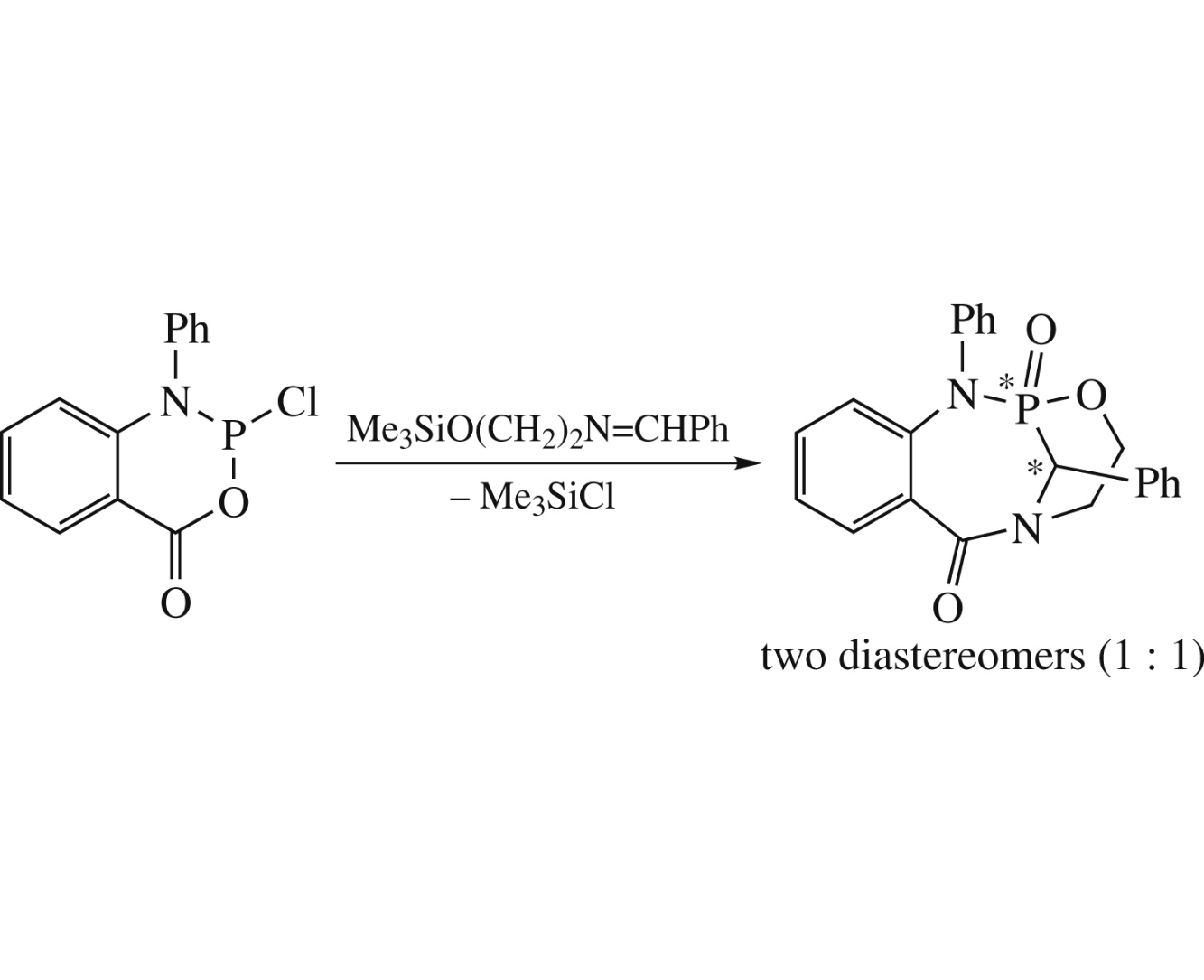
A mild approach to the synthesis of the title cage aminophosphonate has been developed based on the intramolecular cyclization of 2-(2-benzylideneaminoethoxy)-1-phenylbenzo[e]-1,3,2-azaoxaphosphorin-4-one obtained from 1-phenyl-2-chlorobenzo[e]-1,3,2-azaoxaphosphorin-4-one and (2-benzylideneaminoethoxy)trimethylsilane. The structure of isolated diastereomers of the product was determined by NMR spectroscopy and single crystal X-ray diffraction analysis.
References
1.

Orsini F., Sello G., Sisti M.
Current Medicinal Chemistry,
2009
2.

H. Kudzin Z., H. Kudzin M., Drabowicz J., V. Stevens C.
Current Organic Chemistry,
2011
3.

Turcheniuk K.V., Kukhar V.P., Röschenthaler G., Aceña J.L., Soloshonok V.A., Sorochinsky A.E.
RSC Advances,
2013
4.
10.1016/j.mencom.2019.03.010_sbref0005d
Prasad
J. Mod. Med. Chem.,
2013
5.

Makarov M.V., Leonova E.S., Matveeva E.V., Rybalkina E.Y., Röschenthaler G., Timofeeva T.V., Odinets I.L.
Phosphorus, Sulfur and Silicon and the Related Elements,
2011
6.

Tusek-Bozic L.
Current Medicinal Chemistry,
2013
7.

Huang K., Chen Z., Liu Y., Li Z., Wei J., Wang M., Zhang G., Liang H.
European Journal of Medicinal Chemistry,
2013
8.

Cherkasov R.A., Galkin V.I.
Russian Chemical Reviews,
1998
9.
Zefirov N.S., Matveeva E.D.
Arkivoc,
2008
10.

Keglevich G., Bálint E.
Molecules,
2012
11.
10.1016/j.mencom.2019.03.010_sbref0015d
Ali
ARKIVOC,
2014
12.

Abell J.P., Yamamoto H.
Journal of the American Chemical Society,
2008
13.

Prishchenko A.A., Livantsov M.V., Novikova O.P., Livantsova L.I., Milaeva E.R.
Heteroatom Chemistry,
2009
14.

Prishchenko A.A., Livantsov M.V., Novikova O.P., Livantsova L.I.
Russian Journal of General Chemistry,
2010
15.
10.1016/j.mencom.2019.03.010_sbref0020a
Litvinov
Russ. J. Gen. Chem.,
1999
16.

Mironov V.F., Gubaidullin A.T., Ivkova G.A., Litvinov I.A., Buzykin B.I., Burnaeva L.M., Konovalova I.V.
Russian Journal of General Chemistry,
2001
17.

Mironov V.F., Gubaidullin A.T., Burnaeva L.M., Litvinov I.A., Ivkova G.A., Romanov S.V., Zyablikova† T.A., Konovalov A.I., Konovalova I.V.
Russian Journal of General Chemistry,
2004
18.
![Reaction of 2-chloro-1,3,2-benzodioxaphosphinin-4-one with 2-(arylmethylideneamino)phenols. Stereoselective formation of 10-aryl-3,4: 8,9-dibenzo-5,7-dioxa-1-aza-6-phosphabicyclo[4.3.1]decane-2,6-diones](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Dimukhametov M.N., Ivkova G.A., Mironov V.F., Onys’ko P.P., Rassukanaya Y.V., Musin R.Z.
Russian Journal of Organic Chemistry,
2014
19.
![TMSCl-promoted addition of diethyl phosphite to an imine for the synthesis of bis [1-diethoxyphosphorylalkyl] amines](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
Kaboudin B., Moradi K.
Synthesis,
2006
20.

Pokalwar R.U., Hangarge R.V., Madje B.R., Ware M.N., Shingare M.S.
Phosphorus, Sulfur and Silicon and the Related Elements,
2008
21.

Pokalwar R.U., Sadaphal S.A., Kategaonkar A.H., Shingate B.B., Shingare M.S.
Green Chemistry Letters and Reviews,
2010
22.
![Synthesis, in vitro antibacterial and antifungal evaluations of new alpha-hydroxyphosphonate and new alpha-acetoxyphosphonate derivatives of tetrazolo [1, 5-a] quinoline.](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Kategaonkar A.H., Pokalwar R.U., Sonar S.S., Gawali V.U., Shingate B.B., Shingare M.S.
European Journal of Medicinal Chemistry,
2010
23.
10.1016/j.mencom.2019.03.010_sbref0035d
Nagargoje
Pharma Chem.,
2011
24.

Baccari Z., Sanhoury M.A., Crousse B., Barhoumi-Slimi T.
Synthetic Communications,
2018
25.

Anderson J.C., Chapman H.A.
Organic and Biomolecular Chemistry,
2007
26.

Sheldrick G.M.
Acta Crystallographica Section A: Foundations and Advances,
2015
27.

Sheldrick G.M.
Acta crystallographica. Section C, Structural chemistry,
2015
28.

Spek A.L.
Acta Crystallographica Section D Biological Crystallography,
2009