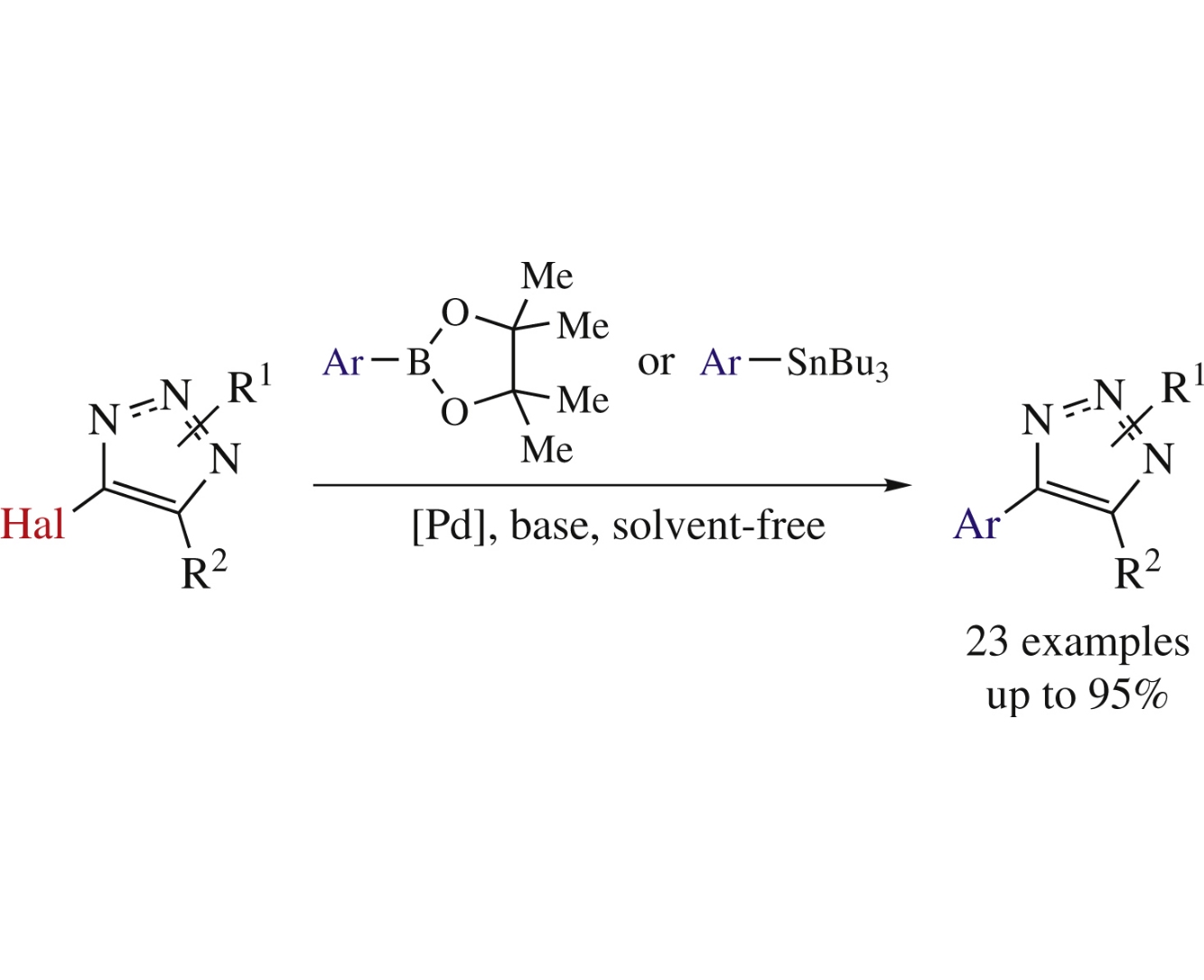
Abstract
An environmentally friendly and efficient synthesis of fully substituted 1,2,3-triazoles comprises solvent-free palladium-catalyzed Suzuki cross-coupling of halo-1,2,3-triazoles with pinacol arylboronates. The efficiencies of Stille and Suzuki reactions with halotriazoles under solvent-free conditions were compared.
References
1.

Agalave S.G., Maujan S.R., Pore V.S.
Chemistry - An Asian Journal,
2011
2.

Kolb H.C., Sharpless K.B.
Drug Discovery Today,
2003
3.

Kantheti S., Narayan R., Raju K.V.
RSC Advances,
2015
4.

Crowley J.D., Bandeen P.H.
Dalton Transactions,
2010
5.

Schulze B., Schubert U.S.
Chemical Society Reviews,
2014
6.

Wang X., Huang B., Liu X., Zhan P.
Drug Discovery Today,
2016
7.

Lauria A., Delisi R., Mingoia F., Terenzi A., Martorana A., Barone G., Almerico A.M.
European Journal of Organic Chemistry,
2014
8.

9.

Huang D., Zhao P., Astruc D.
Coordination Chemistry Reviews,
2014
10.

Lau Y.H., Rutledge P.J., Watkinson M., Todd M.H.
Chemical Society Reviews,
2011
11.
10.1016/j.mencom.2019.03.009_bib0035
Deng
Synthesis,
2005
12.

Maurya S.K., Gollapalli D.R., Kirubakaran S., Zhang M., Johnson C.R., Benjamin N.N., Hedstrom L., Cuny G.D.
Journal of Medicinal Chemistry,
2009
13.

Duan H., Sengupta S., Petersen J.L., Akhmedov N.G., Shi X.
Journal of the American Chemical Society,
2009
14.

Hein J.E., Fokin V.V.
Chemical Society Reviews,
2010
15.

Chattopadhyay B., Gevorgyan V.
Angewandte Chemie - International Edition,
2011
16.

Thirumurugan P., Matosiuk D., Jozwiak K.
Chemical Reviews,
2013
17.

Azagarsamy M.A., Anseth K.S.
ACS Macro Letters,
2012
18.

Gulevich A.V., Gevorgyan V.
Angewandte Chemie - International Edition,
2013
19.

Gu Q., Al Mamari H.H., Graczyk K., Diers E., Ackermann L.
Angewandte Chemie - International Edition,
2014
20.

Zhang L., Chen X., Xue P., Sun H.H., Williams I.D., Sharpless K.B., Fokin V.V., Jia G.
Journal of the American Chemical Society,
2005
21.

Hein J., Tripp J., Krasnova L., Sharpless K. ., Fokin V.
Angewandte Chemie - International Edition,
2009
22.

Worrell B.T., Ellery S.P., Fokin V.V.
Angewandte Chemie - International Edition,
2013
23.

Ding S., Jia G., Sun J.
Angewandte Chemie - International Edition,
2014
24.

Gribanov P.S., Topchiy M.A., Karsakova I.V., Chesnokov G.A., Smirnov A.Y., Minaeva L.I., Asachenko A.F., Nechaev M.S.
European Journal of Organic Chemistry,
2017
25.

Huang J., Macdonald S.J., Harrity J.P.
Chemical Communications,
2009
26.

Wang X., Sidhu K., Zhang L., Campbell S., Haddad N., Reeves D.C., Krishnamurthy D., Senanayake C.H.
Organic Letters,
2009
27.

Wang X., Zhang L., Krishnamurthy D., Senanayake C.H., Wipf P.
Organic Letters,
2010
28.
![Discovery of potent dipeptidyl peptidase IV inhibitors derived from β-aminoamides bearing substituted [1,2,3]-triazolopiperidines for the treatment of type 2 diabetes](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Shan Z., Peng M., Fan H., Lu Q., Lu P., Zhao C., Chen Y.
Bioorganic and Medicinal Chemistry Letters,
2011
29.

De Simone R., Chini M.G., Bruno I., Riccio R., Mueller D., Werz O., Bifulco G.
Journal of Medicinal Chemistry,
2011
30.

Zhang L., Li Z., Wang X., Yee N., Senanayake C.
Synlett,
2012
31.

Kavitha M., Mahipal B., Mainkar P., Chandrasekhar S.
Combinatorial Chemistry and High Throughput Screening,
2013
32.

Cao X., Sun Z., Cao Y., Wang R., Cai T., Chu W., Hu W., Yang Y.
Journal of Medicinal Chemistry,
2014
33.

Goyard D., Chajistamatiou A.S., Sotiropoulou A.I., Chrysina E.D., Praly J., Vidal S.
Chemistry - A European Journal,
2014
34.

Oakdale J.S., Sit R.K., Fokin V.V.
Chemistry - A European Journal,
2014
35.

Ahmed N., Konduru N.K., Ahmad S., Owais M.
European Journal of Medicinal Chemistry,
2014
36.

Wang D., Chen S., Chen B.
Tetrahedron Letters,
2014
37.

Fehér K., Gömöry Á., Skoda-Földes R.
Monatshefte fur Chemie,
2015
38.
10.1016/j.mencom.2019.03.009_sbref0060a
Malnuit
Synlett,
2009
39.

Thiery E., You V., Mora A., Abarbri M.
European Journal of Organic Chemistry,
2015
40.

Wei F., Zhou T., Ma Y., Tung C., Xu Z.
Organic Letters,
2017
41.

Amdouni H., Robert G., Driowya M., Furstoss N., Métier C., Dubois A., Dufies M., Zerhouni M., Orange F., Lacas-Gervais S., Bougrin K., Martin A.R., Auberger P., Benhida R.
Journal of Medicinal Chemistry,
2017
42.

Yamada M., Matsumura M., Kawahata M., Murata Y., Kakusawa N., Yamaguchi K., Yasuike S.
Journal of Organometallic Chemistry,
2017
43.

Gribanov P.S., Chesnokov G.A., Topchiy M.A., Asachenko A.F., Nechaev M.S.
Organic and Biomolecular Chemistry,
2017
44.

Chesnokov G.A., Topchiy M.A., Dzhevakov P.B., Gribanov P.S., Tukov A.A., Khrustalev V.N., Asachenko A.F., Nechaev M.S.
Dalton Transactions,
2017
45.

Morozov O.S., Gribanov P.S., Asachenko A.F., Dorovatovskii P.V., Khrustalev V.N., Rybakov V.B., Nechaev M.S.
Advanced Synthesis and Catalysis,
2016
46.

Asachenko A.F., Sorochkina K.R., Dzhevakov P.B., Topchiy M.A., Nechaev M.S.
Advanced Synthesis and Catalysis,
2013
47.

Topchiy M.A., Asachenko A.F., Nechaev M.S.
European Journal of Organic Chemistry,
2014
48.

Dzhevakov P.B., Topchiy M.A., Zharkova D.A., Morozov O.S., Asachenko A.F., Nechaev M.S.
Advanced Synthesis and Catalysis,
2016
49.

Topchiy M.A., Dzhevakov P.B., Rubina M.S., Morozov O.S., Asachenko A.F., Nechaev M.S.
European Journal of Organic Chemistry,
2016
50.
Chesnokov G.A., Gribanov P.S., Topchiy M.A., Minaeva L.I., Asachenko A.F., Nechaev M.S., Bermesheva E.V., Bermeshev M.V.
Mendeleev Communications,
2017
51.

Gribanov P.S., Golenko Y.D., Topchiy M.A., Minaeva L.I., Asachenko A.F., Nechaev M.S.
European Journal of Organic Chemistry,
2018
52.
Gribanov P.S., Golenko Y.D., Topchiy M.A., Philippova A.N., Kirilenko N.Y., Krivoshchapov N.V., Sterligov G.K., Asachenko A.F., Bermeshev M.V., Nechaev M.S.
Mendeleev Communications,
2018
53.

Ji H., Cai J., Gan N., Wang Z., Wu L., Li G., Yi T.
Chemistry Central Journal,
2018
54.

Dong J., Guo H., Hu Q.
European Journal of Organic Chemistry,
2017
55.

Yan G., Huang D., Wu X.
Advanced Synthesis and Catalysis,
2017
56.

Qi X., Jiang L., Zhou C., Peng J., Wu X.
ChemistryOpen,
2017
57.

Cheng Y., Mück‐Lichtenfeld C., Studer A.
Angewandte Chemie - International Edition,
2018
58.

Xu Y., Yang X., Fang H.
Journal of Organic Chemistry,
2018
59.

Qi X., Li H., Peng J., Wu X.
Tetrahedron Letters,
2017
60.
10.1016/j.mencom.2019.03.009_bib0095
Boronic Acids,
2005
61.

Catalysts for Suzuki−Miyaura Coupling Processes: Scope and Studies of the Effect of Ligand Structure
Barder T.E., Walker S.D., Martinelli J.R., Buchwald S.L.
Journal of the American Chemical Society,
2005
62.
10.1016/j.mencom.2019.03.009_bib0105
Palladium in Heterocyclic Chemistry: A Guide for the Synthetic Chemist,
2007