Abstract
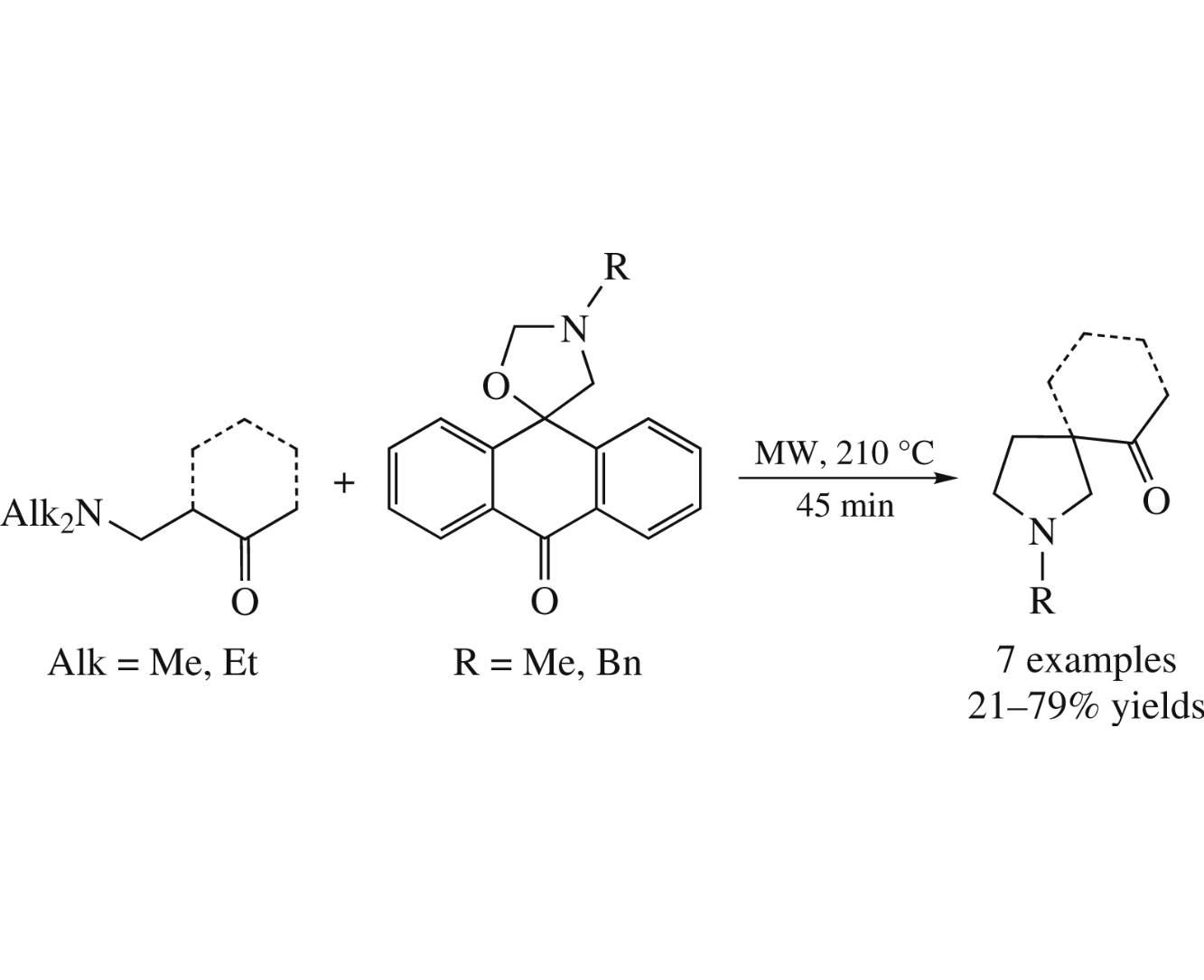
Mannich bases obtained from cycloalkanones and methyl-ketones decompose on heating to give α,β-enones, which react in situ with nonstabilized azomethine ylides formed from spiro[anthracene-oxazolidines]. The final products, 3-acylpyrrolidines, were obtained in yields of 21–79% by heating the starting compounds in a microwave reactor in o-xylene at 210°C for 45min
References
1.

Plunkett A.O.
Natural Product Reports,
1994
2.

Dubuffet T., Muller O., Simonet S.S., Descombes J., Laubie M., Verbeuren T.J., Lavielle G.
Bioorganic and Medicinal Chemistry Letters,
1996
3.
![Novel benzopyrano[3,4-c]pyrrole derivatives as potent and selective dopamine D3 receptor antagonist.](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Novel benzopyrano[3,4-c]pyrrole derivatives as potent and selective dopamine D3 receptor antagonist.
Dubuffet T., Newman-Tancredi A., Cussac D., Audinot V., Loutz A., Millan M.J., Lavielle G.
Bioorganic and Medicinal Chemistry Letters,
1999
4.

Grigg R., Thianpatanagul S.
Journal of the Chemical Society Chemical Communications,
1984
5.

Tsuge O., Kanemasa S.
Advances in Heterocyclic Chemistry,
1989
6.
10.1016/j.mencom.2019.03.008_sbref0010c
Harwood
2002
7.

Najera C., Sansano J.
Current Organic Chemistry,
2003
8.

Coldham I., Hufton R.
Chemical Reviews,
2005
9.
Moshkin V.S., Sosnovskikh V.Y., Slepukhin P.A., Röschenthaler G.
Mendeleev Communications,
2012
10.
11.

Buev E.M., Moshkin V.S., Sosnovskikh V.Y.
Journal of Organic Chemistry,
2017
12.

Buev E.M., Moshkin V.S., Sosnovskikh V.Y.
Organic Letters,
2016
13.

Mannich C., Hönig P.
Archiv der Pharmazie,
1927
14.

Blicke F.F., Burckhalter J.H.
Journal of the American Chemical Society,
1942
15.

Nguyet Anh Le, Jones M., Bickelhaupt F., De Wolf W.H.
Journal of the American Chemical Society,
1989
16.

Helsley G.C., Richman J.A., Lunsford C.D., Jenkins H., Mays R.P., Funderburk W.H., Johnson D.N.
Journal of Medicinal Chemistry,
1968
17.

Liu G., Abraham S., Tran L., Vickers T.D., Xu S., Hadd M.J., Quiambao S., Holladay M.W., Hua H., Ford Pulido J.M., Gunawardane R.N., Davis M.I., Eichelberger S.R., Apuy J.L., Gitnick D., et. al.
Journal of Medicinal Chemistry,
2012
18.
S. Liming, S. Jian, Y. Xicheng, Y. Mingcheng, X. Li, L. Wei and X. Qiong, CN Patent 106278994, 2017.