Abstract
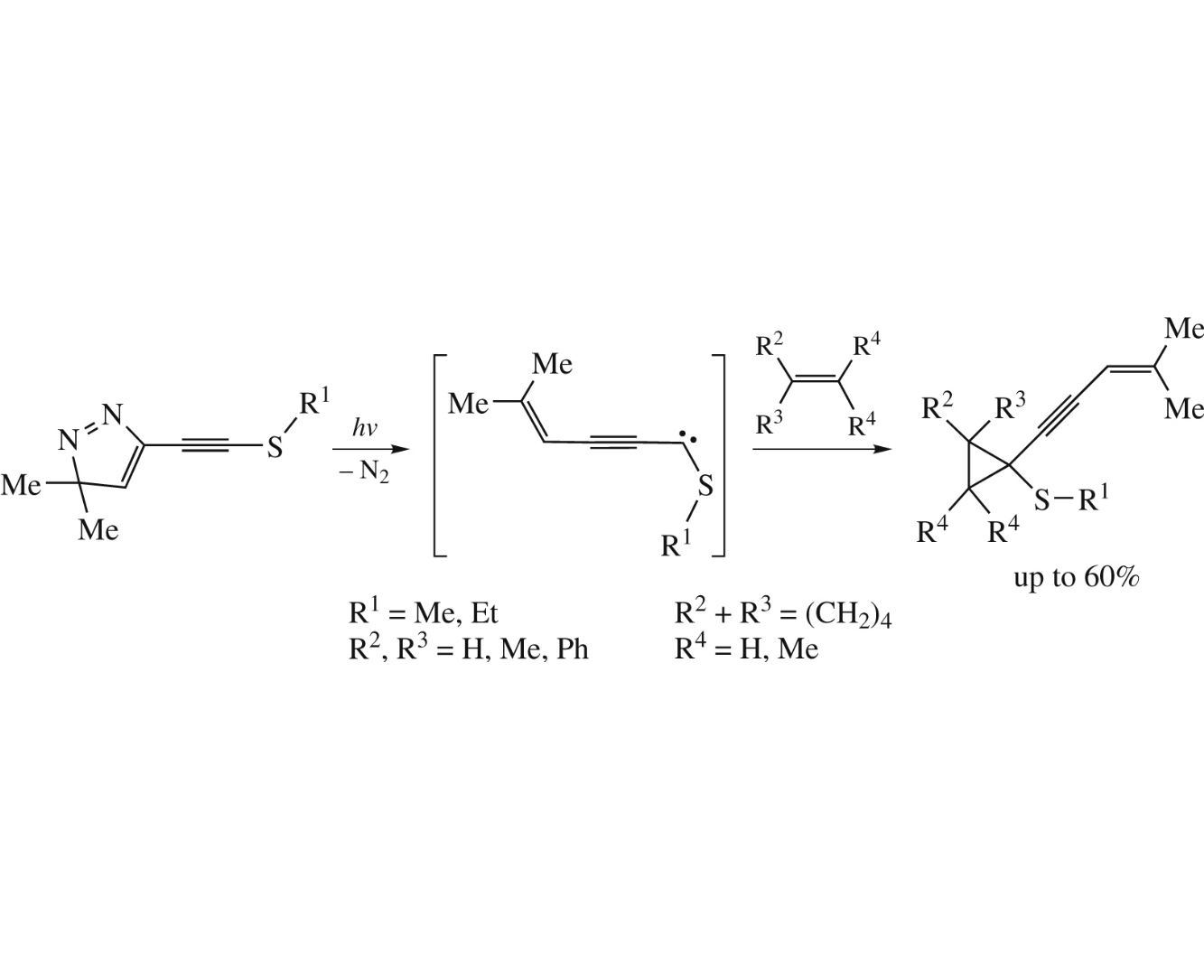
New singlet alkylthio(4-methylpent-3-en-1-ynyl)carbenes were generated by photolysis of corresponding 5-alkylthioethynyl-3,3-dimethyl-3H-pyrazoles and subsequently trapped by alkenes giving rise to 1-alkynyl-1-(alkylthio)cyclopropanes in yields up to 60%.
References
1.

Qin Y., Tang P.
Synthesis,
2012
2.

Chen D.Y., Pouwer R.H., Richard J.
Chemical Society Reviews,
2012
3.

Ebner C., Carreira E.M.
Chemical Reviews,
2017
4.
Ananikov V.P., Eremin D.B., Yakukhnov S.A., Dilman A.D., Levin V.V., Egorov M.P., Karlov S.S., Kustov L.M., Tarasov A.L., Greish A.A., Shesterkina A.A., Sakharov A.M., Nysenko Z.N., Sheremetev A.B., Stakheev A.Y., et. al.
Mendeleev Communications,
2017
5.

Novakov I.A., Babushkin A.S., Yablokov A.S., Nawrozkij M.B., Vostrikova O.V., Shejkin D.S., Mkrtchyan A.S., Balakin K.V.
Russian Chemical Bulletin,
2018
6.

Franck- Neumann M., Geoffroy P.
Tetrahedron Letters,
1983
7.

Davies H.M., Boebel T.A.
Tetrahedron Letters,
2000
8.

Shavrin K.N., Gvozdev V.D., Nefedov O.M.
Russian Chemical Bulletin,
2002
9.
Shavrin K.N., Gvozdev V.D., Nefedov O.M.
Mendeleev Communications,
2003
10.

Shavrin K.N., Gvozdev V.D., Pinus I.Y., Dotsenko I.P., Nefedov O.M.
Russian Chemical Bulletin,
2004
11.

Barluenga J., Fernández-Rodríguez M.A., García-García P., Aguilar E., Merino I.
Chemistry - A European Journal,
2006
12.

Gvozdev V.D., Shavrin K.N., Ageshina A.A., Nefedov O.M.
Russian Chemical Bulletin,
2017
13.

Fernández-García J.M., Garro H.A., Fernández-García L., García-García P., Fernández-Rodríguez M.A., Merino I., Aguilar E.
Advanced Synthesis and Catalysis,
2017
14.

Yi F., Huang B., Nie Q., Cai M.
Tetrahedron Letters,
2016
15.
![Sc(OTf)3-Catalyzed Diastereoselective Formal [3+2] Cycloaddition Reactions of Alkynylcyclopropane Ketones with Electron-Rich Aromatic Aldehydes To Yield 2,5-trans-Tetrahydrofurans](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Zhang C., Xu M., Ren J., Wang Z.
European Journal of Organic Chemistry,
2016
16.
![Lewis Acid Catalyzed Intramolecular [4+2] and [3+2] Cross-Cycloaddition of Alkynylcyclopropane Ketones with Carbonyl Compounds and Imines](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Bai Y., Tao W., Ren J., Wang Z.
Angewandte Chemie - International Edition,
2012
17.

Ye S., Yu Z.
Chemical Communications,
2011
18.

Shavrin K.N., Gvozdev V.D., Nefedov O.M.
Russian Chemical Bulletin,
2010
19.
![Synthesis of 5-methylidenehexahydropyrrolo[l,2-a]imidazoles and 6-methylideneoctahydropyrrolo[l,2-a]pyrimidines by the reaction of 1-alkynyl-1-chlorocyclopropanes with lithium derivatives of 1,2- and 1,3-diaminoalkanes](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Shavrin K.N., Gvozdev V.D., Nefedov O.M.
Russian Chemical Bulletin,
2010
20.
Shavrin K.N., Gvozdev V.D., Nefedov O.M.
Mendeleev Communications,
2008
21.

Trost B.M., Xie J., Maulide N.
Journal of the American Chemical Society,
2008
22.

Barluenga J., Tudela E., Vicente R., Ballesteros A., Tomás M.
Angewandte Chemie - International Edition,
2011
23.

Markham J.P., Staben S.T., Toste F.D.
Journal of the American Chemical Society,
2005
24.

Zampella A., D'Auria M.V., Minale L., Debitus C., Roussakis C.
Journal of the American Chemical Society,
1996
25.
10.1016/j.mencom.2019.03.006_bib0125
Tedford
J. Pharmacol. Exp. Ther.,
1999
26.

Corbett J.W., Ko S.S., Rodgers J.D., Gearhart L.A., Magnus N.A., Bacheler L.T., Diamond S., Jeffrey S., Klabe R.M., Cordova B.C., Garber S., Logue K., Trainor G.L., Anderson P.S., Erickson-Viitanen S.K., et. al.
Journal of Medicinal Chemistry,
2000
27.

Maier G., Preiss T., Reisenauer H.P., Hess B.A., Schaad L.J.
Journal of the American Chemical Society,
1994
28.

Boganov S.E., Faustov V.I., Shavrin K.N., Gvozdev V.D., Promyslov V.M., Egorov M.P., Nefedov O.M.
Journal of the American Chemical Society,
2009
29.

Misochko E.Y., Akimov A.V., Korchagin D.V., Masitov A.A., Shavrin K.N.
Physical Chemistry Chemical Physics,
2012
30.

Melzig L., Metzger A., Knochel P.
Chemistry - A European Journal,
2011
31.

Zheng W., Zheng F., Hong Y., Hu L.
Heteroatom Chemistry,
2011
32.

Shavrin K.N., Krylova I.V., Shvedova I.B., Okonnishnikova G.P., Dolgy I.E., Nefedov O.M.
Journal of the Chemical Society Perkin Transactions 2,
1991