Abstract
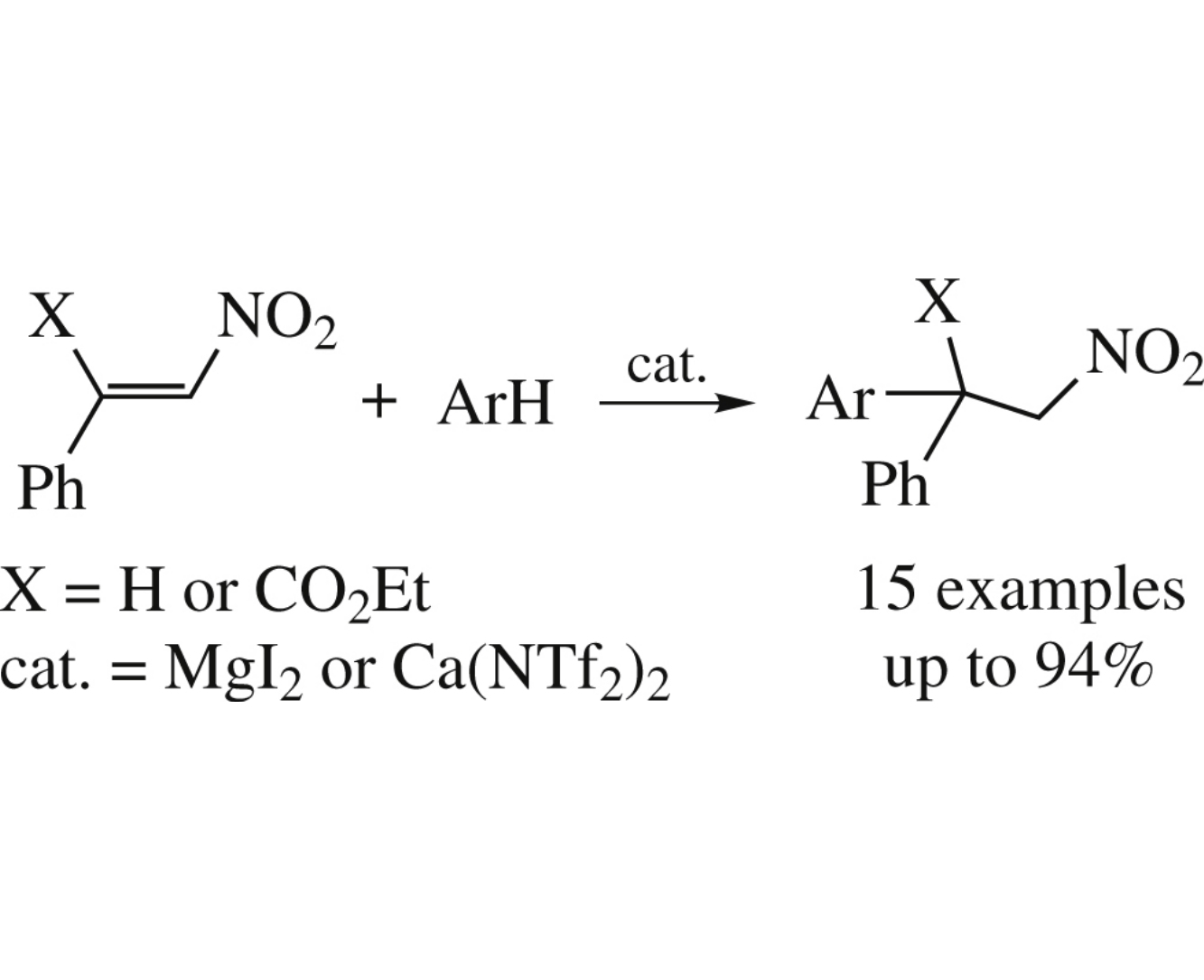
The Friedel–Crafts reaction between electron-rich (het)arenes and β-nitrostyrenes under MgI2 or Ca(NTf2)2 catalysis affords 1-(het)aryl-2-nitro-1-phenylethanes in yields up to 94%.
References
1.
10.1016/j.mencom.2019.03.005_bib0005
Ono
The Nitro Group in Organic Synthesis,
2001
2.

Meshram H., Kumar D., Reddy B.
Helvetica Chimica Acta,
2009
3.

4.

Mane A., Lohar T., Salunkhe R.
Tetrahedron Letters,
2016
5.

Izaga A., Herrera R.P., Gimeno M.C.
ChemCatChem,
2017
6.

da Silva R.C., da Silva G.P., Sangi D.P., Pontes J.G., Ferreira A.G., Corrêa A.G., Paixão M.W.
Tetrahedron,
2013
7.

McGuirk C.M., Katz M.J., Stern C.L., Sarjeant A.A., Hupp J.T., Farha O.K., Mirkin C.A.
Journal of the American Chemical Society,
2015
8.

Rao P.C., Mandal S.
ChemCatChem,
2017
9.

Moriyama K., Sugiue T., Saito Y., Katsuta S., Togo H.
Advanced Synthesis and Catalysis,
2015
10.

So S.S., Burkett J.A., Mattson A.E.
Organic Letters,
2011
11.

Fan Y., Kass S.R.
Journal of Organic Chemistry,
2017
12.

Weng J., Deng Q., Wu L., Xu K., Wu H., Liu R., Gao J., Jia Y.
Organic Letters,
2014
13.

Huang K., Ma Q., Shen X., Gong L., Meggers E.
Asian Journal of Organic Chemistry,
2016
14.

Despotopoulou C., McKeon S.C., Connon R., Coeffard V., Müller-Bunz H., Guiry P.J.
European Journal of Organic Chemistry,
2017
15.

O’Reilly S., Aylward M., Keogh-Hansen C., Fitzpatrick B., McManus H.A., Müller-Bunz H., Guiry P.J.
Journal of Organic Chemistry,
2015
16.

Scherer A., Mukherjee T., Hampel F., Gladysz J.A.
Organometallics,
2014
17.

Liu J., Gong L., Meggers E.
Tetrahedron Letters,
2015
18.

Turks M., Rolava E., Stepanovs D., Mishnev A., Marković D.
Tetrahedron Asymmetry,
2015
19.

Li N., Qiu R., Zhang X., Chen Y., Yin S., Xu X.
Tetrahedron,
2015
20.

More G.V., Bhanage B.M.
Catalysis Science and Technology,
2015
21.

Le T., Diter P., Pégot B., Bournaud C., Toffano M., Guillot R., Vo-Thanh G., Yagupolskii Y., Magnier E.
Journal of Fluorine Chemistry,
2015
22.

Ma J., Kass S.R.
Organic Letters,
2016
23.

Arai T., Awata A., Wasai M., Yokoyama N., Masu H.
Journal of Organic Chemistry,
2011
24.

Guo F., Chang D., Lai G., Zhu T., Xiong S., Wang S., Wang Z.
Chemistry - A European Journal,
2011
25.

Chittoory A.K., Kumari G., Mohapatra S., Kundu P.P., Maji T.K., Narayana C., Rajaram S.
Tetrahedron,
2014
26.

Zhang G.
Organic and Biomolecular Chemistry,
2012
27.

Halimehjani A.Z., Farvardin M.V., Zanussi H.P., Ranjbari M.A., Fattahi M.
Journal of Molecular Catalysis A Chemical,
2014
28.

Stoermer M.J., Richter H., Kaufmann D.E.
Tetrahedron Letters,
2013
29.

Han X., Ye C., Chen F., Chen Q., Wang Y., Zeng X.
Organic and Biomolecular Chemistry,
2017
30.

Carmona D., Lamata M.P., Sánchez A., Viguri F., Oro L.A.
Tetrahedron Asymmetry,
2011
31.

Tran N.T., Wilson S.O., Franz A.K.
Organic Letters,
2011
32.

Jarava-Barrera C., Esteban F., Navarro-Ranninger C., Parra A., Alemán J.
Chemical Communications,
2013
33.

Aldoshin A.S., Tabolin A.A., Ioffe S.L., Nenajdenko V.G.
European Journal of Organic Chemistry,
2018
34.

Beletskaya I., Feofanov M., Anokhin M., Averin A.
Synthesis,
2017
35.

Anokhin M.V., Feofanov M.N., Averin A.D., Beletskaya I.P.
ChemistrySelect,
2018
36.
Feofanov M.N., Lozhkin B.A., Anokhin M.V., Averin A.D., Beletskaya I.P.
Mendeleev Communications,
2018