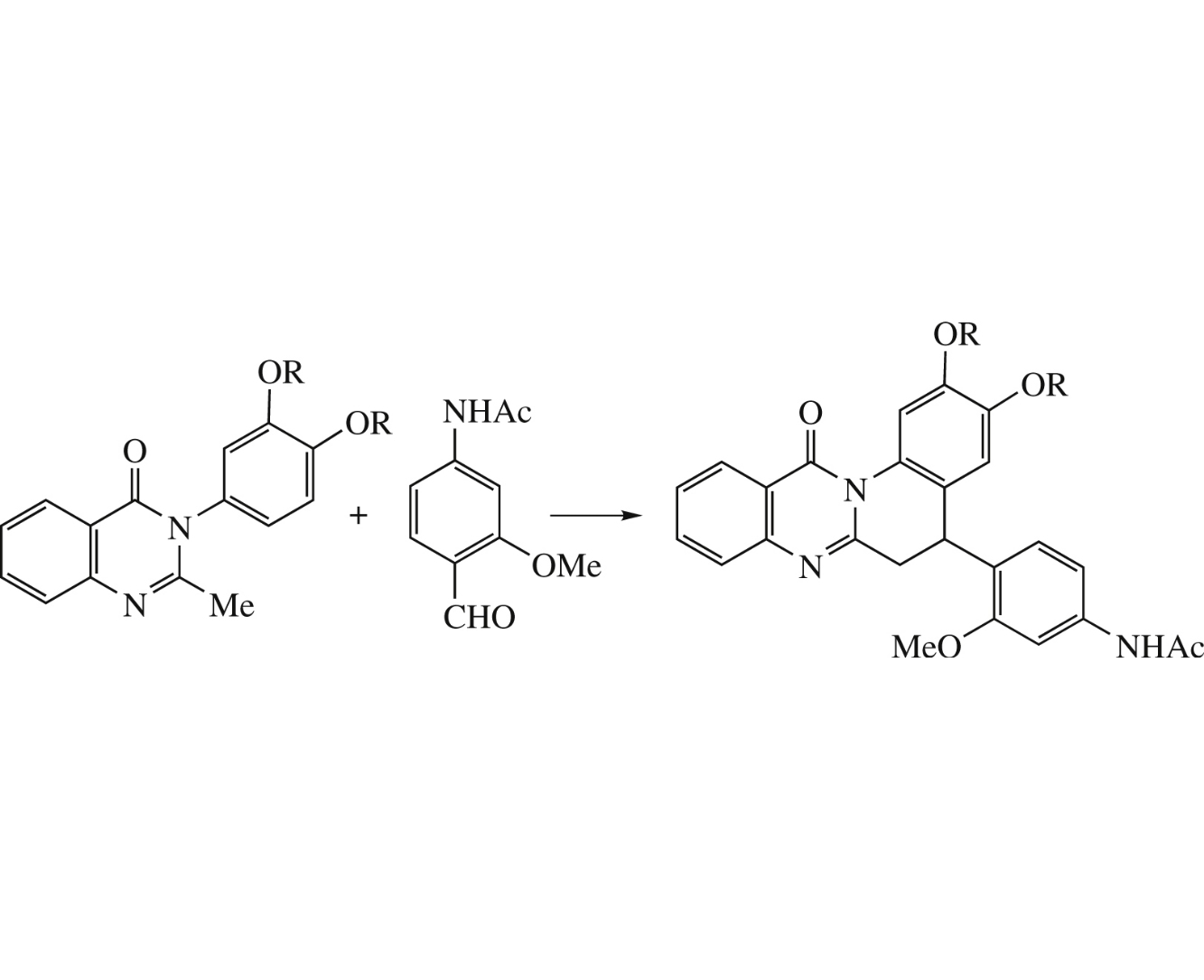
Abstract
Novel tetracyclic 5-aryl-5,6-dihydroquinolino[2,1-b]quinazolin-12-ones bearing benzo-15-crown-5 ether and dimethoxyphenyl moieties were prepared by one-pot cascade synthesis involving the thermally allowed pericyclic transformations of (E)-2-(2-methoxystyryl)quinazolinones. The pseudocyclic transition state of six-electron electrocyclization has been located and the activation barrier has been estimated by RHF/3-21G and 6-311G* calculation methods. The crystal structure of N-[4-(2,3-dimethoxy-12-oxo-5,6-dihydroquinolino[2,1-b]quinazolin-5-yl)-3-methoxyphenyl]acetamide was determined by X-ray diffraction analysis.
References
1.

Woodward R.B., Hoffmann R.
Angewandte Chemie International Edition in English,
1969
2.
10.1016/j.mencom.2019.03.004_sbref0010a
Gilbert
CRC Handbook of Organic Photochemistry and Photobiology,
2004
3.
10.1016/j.mencom.2019.03.004_sbref0010b
Hoffmann
Handbook of Organic Photochemistry and Photobiology,
2004
4.

Zaki M.A., Balachandran P., Khan S., Wang M., Mohammed R., Hetta M.H., Pasco D.S., Muhammad I.
Journal of Natural Products,
2013
5.
10.1016/j.mencom.2019.03.004_bib0020
Meier
Angew. Chem., Int. Ed.,
1992
6.
![An Alternative Procedure for the Synthesis of [5]- and [7]Carbohelicenes](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
El Abed R., Ben Hassine B., Genêt J., Gorsane M., Marinetti A.
European Journal of Organic Chemistry,
2004
7.

Padwa A., Mazzu A.
Tetrahedron Letters,
1974
8.

Lewis F.D., Kalgutkar R.S., Yang J.
Journal of the American Chemical Society,
2001
9.
![Synthesis and Characterization of Some Aza[5]helicenes](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Bazzini C., Brovelli S., Caronna T., Gambarotti C., Giannone M., Macchi P., Meinardi F., Mele A., Panzeri W., Recupero F., Sironi A., Tubino R.
European Journal of Organic Chemistry,
2005
10.

Fedorova O.A., Gulakova E.N., Fedorov Y.V., Lobazova I.E., Alfimov M.V., Jonusauskas G.
Journal of Photochemistry and Photobiology A: Chemistry,
2008
11.

Beaudry C.M., Malerich J.P., Trauner D.
Chemical Reviews,
2005
12.

Tandem pericyclic reactions in a new FeCl3-promoted synthesis of catechol analogues of restrytisol C
Thomas N.F., Lee K.C., Paraidathathu T., Weber J.F., Awang K., Rondeau D., Richomme P.
Tetrahedron,
2002
13.
![Benzo[c]quinolizinium Salts via Intramolecular Cylization1,2](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Fozard A., Bradsher C.K.
Journal of Organic Chemistry,
1966
14.

Lvov A.G., Shirinian V.Z., Kachala V.V., Kavun A.M., Zavarzin I.V., Krayushkin M.M.
Organic Letters,
2014
15.

Ovchinnikova I.G., Kim G.A., Matochkina E.G., Kodess M.I., Slepukhin P.A., Kovalev I.S., Nosova E.V., Rusinov G.L., Charushin V.N.
Journal of Photochemistry and Photobiology A: Chemistry,
2018
16.

Evanseck J.D., IV B.E., Spellmeyer D.C., Houk K.N.
Journal of Organic Chemistry,
1995
17.

Neese F.
Wiley Interdisciplinary Reviews: Computational Molecular Science,
2011
18.

Krishnan R., Binkley J.S., Seeger R., Pople J.A.
Journal of Chemical Physics,
1980
19.

Clark T., Chandrasekhar J., Spitznagel G.W., Schleyer P.V.
Journal of Computational Chemistry,
1983
20.

Frisch M.J., Pople J.A., Binkley J.S.
Journal of Chemical Physics,
1984
21.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007