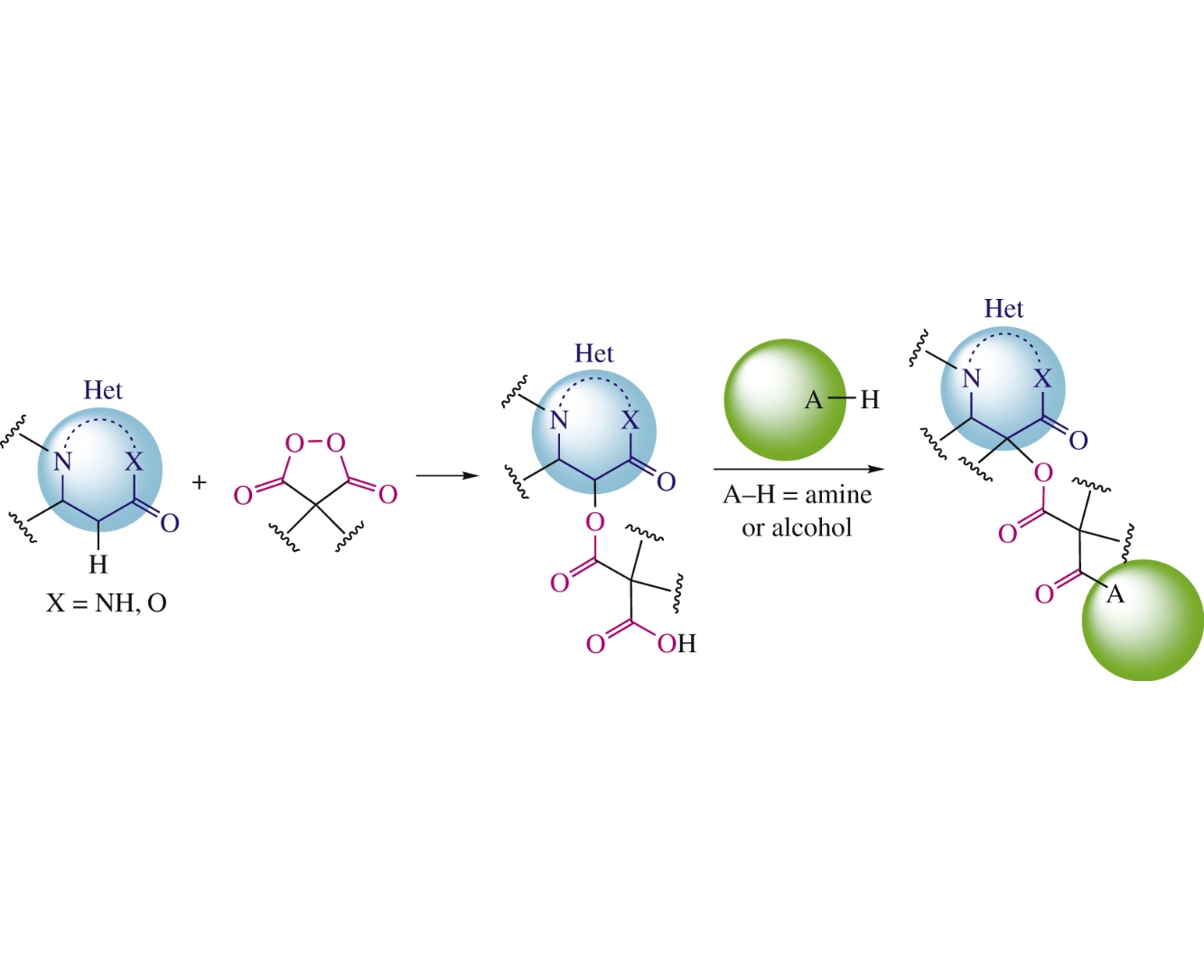
Abstract
Malonyl peroxides were used for the one-pot construction of hybrid structures by conjugation of heterocycle (3H-pyrazol-3-one, isoxazol-5(2H)-one, pyrazolidine-3,5-dione, or barbituric acid) and amine or alcohol. The first stage represents oxidative C–O coupling with liberation of the carboxyl group which is functionalized at the second stage thus affording ‘clicklike’ products with malonic linker.
References
1.

Posner G.H., Ploypradith P., Hapangama W., Wang D., Cumming J.N., Dolan P., Kensler T.W., Klinedinst D., Shapiro T.A., Qun Yi Zheng, Murray C.K., Pilkington L.G., Jayasinghe L.R., Bray J.F., Daughenbaugh R., et. al.
Bioorganic and Medicinal Chemistry,
1997
2.

Posner G.H., Ploypradith P., Parker M.H., O'Dowd H., Woo S., Northrop J., Krasavin M., Dolan P., Kensler T.W., Xie S., Shapiro T.A.
Journal of Medicinal Chemistry,
1999
3.

Posner G.H., Paik I., Sur S., McRiner A.J., Borstnik K., Xie S., Shapiro T.A.
Journal of Medicinal Chemistry,
2003
4.

Jeyadevan J.P., Bray P.G., Chadwick J., Mercer A.E., Byrne A., Ward S.A., Park B.K., Williams D.P., Cosstick R., Davies J., Higson A.P., Irving E., Posner G.H., O'Neill P.M.
Journal of Medicinal Chemistry,
2004
5.

Chadwick J., Mercer A.E., Park B.K., Cosstick R., O’Neill P.M.
Bioorganic and Medicinal Chemistry,
2009
6.

Galal A.M., Gul W., Slade D., Ross S.A., Feng S., Hollingshead M.G., Alley M.C., Kaur G., ElSohly M.A.
Bioorganic and Medicinal Chemistry,
2009
7.

Chaturvedi D., Goswami A., Pratim Saikia P., Barua N.C., Rao P.G.
Chemical Society Reviews,
2010
8.

Fröhlich T., Çapcı Karagöz A., Reiter C., Tsogoeva S.B.
Journal of Medicinal Chemistry,
2016
9.

Paik I., Xie S., Shapiro T.A., Labonte T., Narducci Sarjeant A.A., Baege A.C., Posner G.H.
Journal of Medicinal Chemistry,
2006
10.

Alagbala A.A., McRiner A.J., Borstnik K., Labonte T., Chang W., D'Angelo J.G., Posner G.H., Foster B.A.
Journal of Medicinal Chemistry,
2006
11.

Horwedel C., Tsogoeva S.B., Wei S., Efferth T.
Journal of Medicinal Chemistry,
2010
12.

Reiter C., Herrmann A., Çapci A., Efferth T., Tsogoeva S.B.
Bioorganic and Medicinal Chemistry,
2012
13.

Wang S., Sasaki T.
Bioorganic and Medicinal Chemistry Letters,
2013
14.

Fröhlich T., Hahn F., Belmudes L., Leidenberger M., Friedrich O., Kappes B., Couté Y., Marschall M., Tsogoeva S.B.
Chemistry - A European Journal,
2018
15.
Ananikov V.P., Eremin D.B., Yakukhnov S.A., Dilman A.D., Levin V.V., Egorov M.P., Karlov S.S., Kustov L.M., Tarasov A.L., Greish A.A., Shesterkina A.A., Sakharov A.M., Nysenko Z.N., Sheremetev A.B., Stakheev A.Y., et. al.
Mendeleev Communications,
2017
16.

Kolb H.C., Finn M.G., Sharpless K.B.
Angewandte Chemie - International Edition,
2001
17.

Sletten E., Bertozzi C.
Angewandte Chemie - International Edition,
2009
18.

Jewett J.C., Bertozzi C.R.
Chemical Society Reviews,
2010
19.

20.

Lallana E., Riguera R., Fernandez-Megia E.
Angewandte Chemie - International Edition,
2011
21.

Grammel M., Hang H.C.
Nature Chemical Biology,
2013
22.

Xie R., Hong S., Chen X.
Current Opinion in Chemical Biology,
2013
23.

Su Y., Ge J., Zhu B., Zheng Y., Zhu Q., Yao S.Q.
Current Opinion in Chemical Biology,
2013
24.

Zeng D., Zeglis B.M., Lewis J.S., Anderson C.J.
Journal of Nuclear Medicine,
2013
25.

Evans R.A.
Australian Journal of Chemistry,
2007
26.

Hou J., Liu X., Shen J., Zhao G., Wang P.G.
Expert Opinion on Drug Discovery,
2012
27.

Fröhlich T., Ndreshkjana B., Muenzner J.K., Reiter C., Hofmeister E., Mederer S., Fatfat M., El-Baba C., Gali-Muhtasib H., Schneider-Stock R., Tsogoeva S.B.
ChemMedChem,
2017
28.

Fröhlich T., Reiter C., Ibrahim M.M., Beutel J., Hutterer C., Zeitträger I., Bahsi H., Leidenberger M., Friedrich O., Kappes B., Efferth T., Marschall M., Tsogoeva S.B.
ACS Omega,
2017
29.

Held F.E., Guryev A.A., Fröhlich T., Hampel F., Kahnt A., Hutterer C., Steingruber M., Bahsi H., von Bojničić-Kninski C., Mattes D.S., Foertsch T.C., Nesterov-Mueller A., Marschall M., Tsogoeva S.B.
Nature Communications,
2017
30.

Gangaprasad D., Paul Raj J., Kiranmye T., Karthikeyan K., Elangovan J.
European Journal of Organic Chemistry,
2016
31.

Lin F.L., Hoyt H.M., van Halbeek H., Bergman R.G., Bertozzi C.R.
Journal of the American Chemical Society,
2005
32.

Xiong J., Wei X., Liu Z., Ding M.
Journal of Organic Chemistry,
2017
33.

Davies H.M., Morton D.
Journal of Organic Chemistry,
2016
34.

Yamaguchi J., Yamaguchi A.D., Itami K.
Angewandte Chemie - International Edition,
2012
35.

Li C.
Accounts of Chemical Research,
2008
36.

Yeung C.S., Dong V.M.
Chemical Reviews,
2011
37.
10.1016/j.mencom.2019.03.003_sbref0060e
Girard
Angew. Chem. Int. Ed.,
2013
38.

Krylov I.B., Vil’ V.A., Terent’ev A.O.
Beilstein Journal of Organic Chemistry,
2015
39.

Zhang C., Tang C., Jiao N.
Chemical Society Reviews,
2012
40.

Samanta R., Matcha K., Antonchick A.P.
European Journal of Organic Chemistry,
2013
41.

Terent'ev A.O., Vil' V.A., Gorlov E.S., Rusina O.N., Korlyukov A.A., Nikishin G.I., Adam W.
ChemistrySelect,
2017
42.

Kucera L.S., Morris‐Natschke S.L., Ishaq K.S., Hes J., Iyer N., Furman P.A., Fleming R.A.
Nucleosides, Nucleotides and Nucleic Acids,
2004
43.

Jung M., Tak J., Chung W., Park K.
Bioorganic and Medicinal Chemistry Letters,
2006
44.

Sherman D., Xiong L., Mankin A.S., Melman A.
Bioorganic and Medicinal Chemistry Letters,
2006
45.
![Synthesis of decacationic [60]fullerene decaiodides giving photoinduced production of superoxide radicals and effective PDT-mediation on antimicrobial photoinactivation.](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Wang M., Maragani S., Huang L., Jeon S., Canteenwala T., Hamblin M.R., Chiang L.Y.
European Journal of Medicinal Chemistry,
2013
46.

Yin R., Wang M., Huang Y., Landi G., Vecchio D., Chiang L.Y., Hamblin M.R.
Free Radical Biology and Medicine,
2015
47.

Zhang Y., Dai T., Wang M., Vecchio D., Chiang L.Y., Hamblin M.R.
Nanomedicine,
2015
48.
Khaybullin R.N., Strobykina I.Y., Gubskaya V.P., Fazleeva G.M., Latypov S.K., Kataev V.E.
Mendeleev Communications,
2011
49.
10.1016/j.mencom.2019.03.003_bib0090
Varvounis
2009
50.

Kafle B., Aher N.G., Khadka D., Park H., Cho H.
Chemistry - An Asian Journal,
2011
51.

Ishioka T., Kubo A., Koiso Y., Nagasawa K., Itai A., Hashimoto Y.
Bioorganic and Medicinal Chemistry,
2002
52.

Ishioka T., Tanatani A., Nagasawa K., Hashimoto Y.
Bioorganic and Medicinal Chemistry Letters,
2003
53.

Chande M.S., Verma R.S., Barve P.A., Khanwelkar R.R., Vaidya R.B., Ajaikumar K.B.
European Journal of Medicinal Chemistry,
2005
54.

Wallis R.B.
Thrombosis Research,
1983
55.

Buchanan M.R.
Thrombosis Research,
1983
56.
10.1016/j.mencom.2019.03.003_sbref0110c
Perez-Ruiz
Crystal-Induced Arthropathies. Gout, Pseudogout and Apatite-Associated Syndromes,,
2006
57.

Vesell E.S., Page J.G.
Science,
1968
58.

Beretta C., Garavaglia G., Cavalli M.
Pharmacological Research,
2005
59.

SOMA L.R., UBOH C.E., MAYLIN G.M.
Journal of Veterinary Pharmacology and Therapeutics,
2011
60.
10.1016/j.mencom.2019.03.003_bib0120
Barceloux
Medical Toxicology of Drug Abuse: Synthesized Chemicals and Psychoactive Plants,
2012
61.
10.1016/j.mencom.2019.03.003_sbref0125a
Feng
Rev.,
2011
62.

Jiao S., Yao X., Liu Y., Wang Q., Zeng F., Lu J., Liu J., Zhu C., Shen L., Liu C., Wang Y., Zeng G., Parikh A., Chen J., Liang C., et. al.
Proceedings of the National Academy of Sciences of the United States of America,
2015
63.

Yuan C., Liang Y., Hernandez T., Berriochoa A., Houk K.N., Siegel D.
Nature,
2013
64.

Griffith J.C., Jones K.M., Picon S., Rawling M.J., Kariuki B.M., Campbell M., Tomkinson N.C.
Journal of the American Chemical Society,
2010
65.

Zhao R., Chang D., Shi L.
Synthesis,
2017
66.
Lapitskaya M.A., Vil’ V.A., Daeva E.D., Terent’ev A.O., Pivnitsky K.K.
Mendeleev Communications,
2018