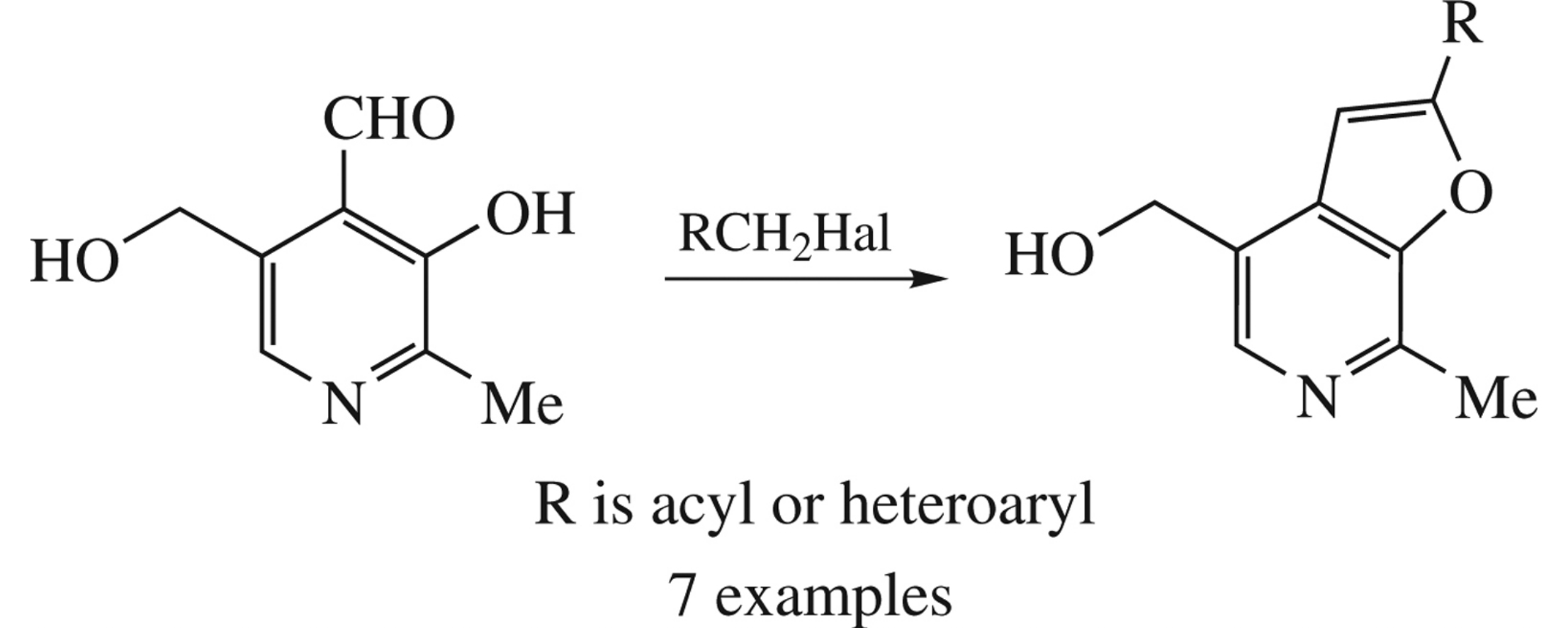
Abstract
A simple method for pyridoxal structural modification via furan ring closure was developed resulting in 2-acyl- and 2-heteroarylfuro[2,3-c]pyridines. The reaction products can be proposed as pyridoxal mimetics to inhibit pyridoxal 5′-phosphate-dependent enzymes.
References
1.

Newman D.J., Cragg G.M.
Journal of Natural Products,
2016
2.
10.1016/j.mencom.2019.01.040_sbref0010a
Anticancer Agents from Natural Products,
2011
3.

Yao H., Liu J., Xu S., Zhu Z., Xu J.
Expert Opinion on Drug Discovery,
2016
4.

Guo Z.
Acta Pharmaceutica Sinica B,
2017
5.

Chen J., Li W., Yao H., Xu J.
Fitoterapia,
2015
6.

Ganesan A.
Current Opinion in Chemical Biology,
2008
7.

Kaur G., Verma N.
Biotechnology Reports,
2015
8.

Maier M.E.
Organic and Biomolecular Chemistry,
2015
9.

Zhang J., Zhang H., Alexandre Muehlmann L., Jiang C., Guo Y.
Molecules,
2017
10.
10.1016/j.mencom.2019.01.040_sbref0010i
Pawar
Nat. Prod. Chem. Res.,
2014
11.

Zhang H., Ryu D., Wu Y., Gariani K., Wang X., Luan P., D’Amico D., Ropelle E.R., Lutolf M.P., Aebersold R., Schoonjans K., Menzies K.J., Auwerx J.
Science,
2016
12.

Oyewole A.O., Birch‐Machin M.A.
FASEB Journal,
2015
13.

Rossman M.J., Santos-Parker J.R., Steward C.A., Bispham N.Z., Cuevas L.M., Rosenberg H.L., Woodward K.A., Chonchol M., Gioscia-Ryan R.A., Murphy M.P., Seals D.R.
Hypertension,
2018
14.

Gioscia-Ryan R.A., LaRocca T.J., Sindler A.L., Zigler M.C., Murphy M.P., Seals D.R.
Journal of Physiology,
2014
15.

Tam K., Ho C., Tu S., Lee W., Huang C., Chen C., Wu C., Lee C., Ho Y.
Oncotarget,
2017
16.

Dong L., Low P., Dyason J.C., Wang X., Prochazka L., Witting P.K., Freeman R., Swettenham E., Valis K., Liu J., Zobalova R., Turanek J., Spitz D.R., Domann F.E., Scheffler I.E., et. al.
Oncogene,
2008
17.

Angulo-Molina A., Reyes-Leyva J., López-Malo A., Hernández J.
Nutrition and Cancer,
2013
18.
M. L. di Salvo, N. Budisa and R. Contestabile, Beilstein Bozen Symposium on Molecular Engineering and Control, Prien am Chiemsee, Germany, 2012, p. 27.
19.

Percudani R., Peracchi A.
EMBO Reports,
2003
20.

Pham V., Zhang W., Chen V., Whitney T., Yao J., Froese D., Friesen A.D., Diakur J.M., Haque W.
Journal of Medicinal Chemistry,
2003
21.

Müller I.B., Wu F., Bergmann B., Knöckel J., Walter R.D., Gehring H., Wrenger C.
PLoS ONE,
2009
22.
![Structure–Activity Relationships in Human Toll-like Receptor 8-Active 2,3-Diamino-furo[2,3-c]pyridines](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Salunke D.B., Yoo E., Shukla N.M., Balakrishna R., Malladi S.S., Serafin K.J., Day V.W., Wang X., David S.A.
Journal of Medicinal Chemistry,
2012
23.

Dowling D.J., Scott E.A., Scheid A., Bergelson I., Joshi S., Pietrasanta C., Brightman S., Sanchez-Schmitz G., Van Haren S.D., Ninković J., Kats D., Guiducci C., de Titta A., Bonner D.K., Hirosue S., et. al.
Journal of Allergy and Clinical Immunology,
2017
24.

Ponticelli F., Marinello E., Pagani R., Terzuoli L.
Journal of Heterocyclic Chemistry,
1991
25.
![Synthesis of 5-Hydroxymethyl-8-methyl-3-(3-aryl-[1,2,4]oxadiazol-5-yl)-2H-pyrano[2,3-c]pyridin-2-ones and Their Esters](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Zhuravel I.O., Kovalenko S.M., Zaremba O.V., Detistov O.S., Kovalenko S.S., Chernykh V.P.
Synthetic Communications,
2008
26.
10.1016/j.mencom.2019.01.040_sbref0080c
Zhuravel’
Izv. Vuzov, Khim. Khim. Tekhnol.,
2007
27.
![Synthesis of substituted 3-(5-amino-[1,3,4]thiadiazol-2-yl)-2H-pyrano[2,3-c]pyridin-2-ones](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Zhuravel' I.O., Kovalenko S.M., Ivachtchenko A.V., Chernykh V.P., Shinkarenko P.E.
Journal of Heterocyclic Chemistry,
2004
28.
10.1016/j.mencom.2019.01.040_sbref0080e
Shinkarenko
Zh. Org. Farm. Khim.,
2010
29.
Batyrkhanovna Derbisbekova U., Mahambetovich Datkhayev U., Alexandrovna Zhuravel I., Amirzhanovna Omarova R., Turgumbayeva A.A.
Oriental Journal Of Chemistry,
2017
30.

Sosnovskikh V.Y., Korotaev V.Y., Barkov A.Y., Sokovnina A.A., Kodess M.I.
Journal of Fluorine Chemistry,
2012
31.

Dale T.J., Sather A.C., Rebek J.
Tetrahedron Letters,
2009
32.

Zubenko A.A., Kartsev V.G., Morkovnik A.S., Divaeva L.N., Alexeenko D.V., Suponitsky K.Y., Borodkin G.S., Klimenko A.I.
Tetrahedron Letters,
2017
33.

Kartsev V.G., Zubenko A.A., Morkovnik A.S., Divaeva L.N.
Tetrahedron Letters,
2015
34.
Zubenko A.A., Morkovnik A.S., Divaeva L.N., Kartsev V.G., Borodkin G.S., Klimenko A.I.
Mendeleev Communications,
2018