Abstract
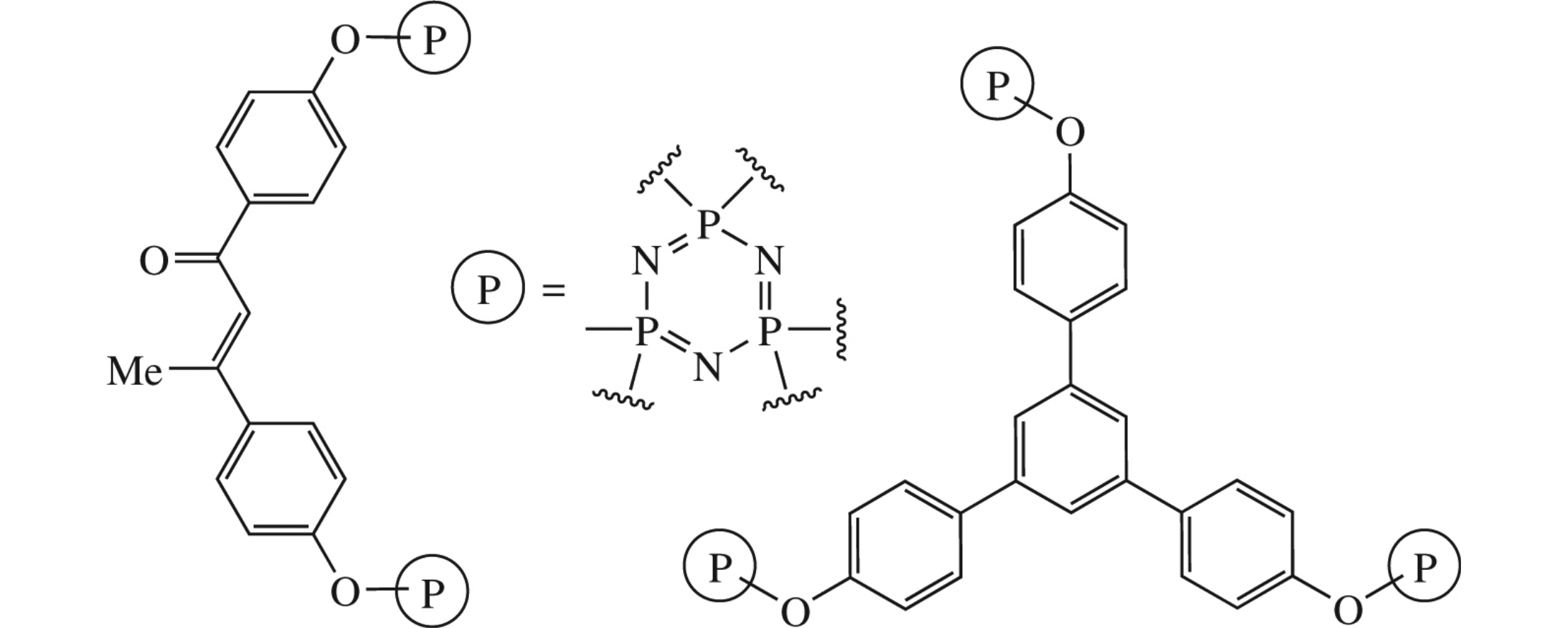
The thermal polycondensation of acetophenoxy groups in hexakis(p-acetylphenoxy)cyclotriphosphazene proceeds in two ways, namely, dimerization and trimerization, to produce chalcone or 1,3,5-triarylbenzene moieties, respectively. The polymers formed in both cases are thermally stable up to 400°C. The structure of hexakis(p-acetylphenoxy)cyclotriphosphazene has been established by X-ray study.
References
1.

Wang J., Su X., Mao Z.
Polymer Degradation and Stability,
2014
2.

Qian L., Ye L., Qiu Y., Qu S.
Polymer,
2011
3.

Huang X., Wei W., Wei H., Li Y., Gu X., Tang X.
Journal of Applied Polymer Science,
2013
4.
![Poly[di(sodium carboxylatoethylphenoxy)phosphazene] (PCEP) is a potent enhancer of mixed Th1/Th2 immune responses in mice immunized with influenza virus antigens](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Mutwiri G., Benjamin P., Soita H., Townsend H., Yost R., Roberts B., Andrianov A.K., Babiuk L.A.
Vaccine,
2007
5.

Khan R.U., Wang L., Yu H., Zain-ul-Abdin, Akram M., Wu J., Haroon M., Ullah R.S., Deng Z., Xia X.
Russian Chemical Reviews,
2018
6.

Stone M.L., Wilson A.D., Harrup M.K., Stewart F.F.
Desalination,
2013
7.

Chandrasekhar V., Suriya Narayanan R.
Dalton Transactions,
2013
8.

Yuan W., Zhu L., Huang X., Zheng S., Tang X.
Polymer Degradation and Stability,
2005
9.

Kireev V.V., Chistyakov E.M., Filatov S.N., Tupikov A.S., Panfilova D.V., Chetverikova A.I.
Russian Journal of Applied Chemistry,
2015
10.

11.

Miyata K., Watanabe Y., Itaya T., Tanigaki T., Inoue K.
Macromolecules,
1996
12.

Chistyakov E.M., Kireev V.V., Filatov S.N., Terekhov I.V., Buzin M.I., Komarova L.I.
Polymer Science - Series B,
2012
13.
![Synthesis, thermal property and hydrolytic degradation of a novel star-shaped hexa[p-(carbonylglycinomethylester)phenoxy]cyclotriphosphazene](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Bing B., Li B.
Science in China Series B Chemistry,
2009
14.

Çil E., Arslan M., Görgülü A.O.
Polyhedron,
2006
15.

Palatinus L., Chapuis G.
Journal of Applied Crystallography,
2007
16.
10.1016/j.mencom.2019.01.034_bib0080
Petřiček
Jana 2006 Software Package,
2006
17.

Zhang X., Higashihara T., Ueda M., Wang L.
Polymer Chemistry,
2014
18.

Maya E.M., Rangel-Rangel E., Díaz U., Iglesias M.
Journal of CO2 Utilization,
2018
19.
Ilgach D.M., Nosova G.I., Kopylova T.N., Nikonova E.N., Gadirov R.M., Smyslov R.Y., Litvinova L.S., Yakimansky A.V.
Mendeleev Communications,
2017