Abstract
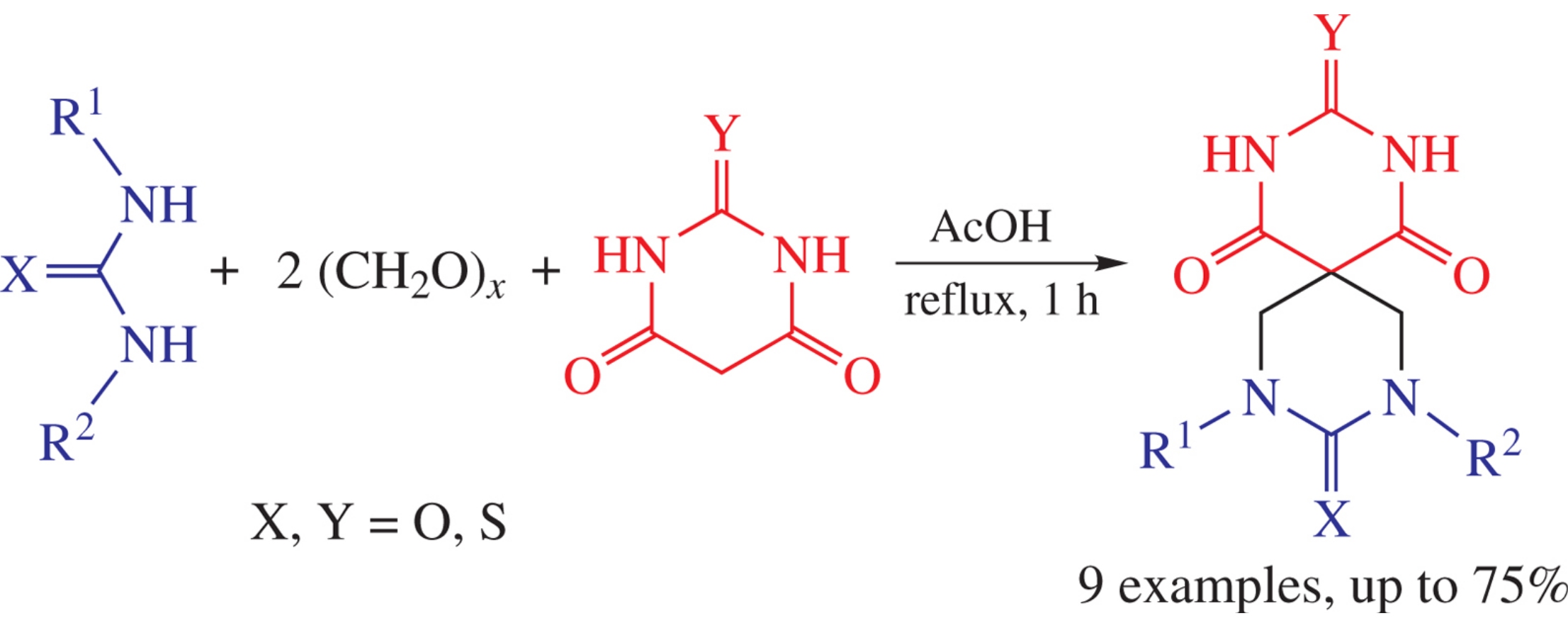
Multicomponent condensations of alkyl(thio)ureas, formaldehyde and (thio)barbituric acids afford new spiro heterocyclic compounds, viz., 8- and 8,10-substituted 2,4,8,10-tetraazaspiro[5.5]undecane-1,3,5,9-tetraones (barbiturils) and their thio analogues.
References
1.

Kravchenko A.N., Baranov V.V., Gazieva G.A.
Russian Chemical Reviews,
2018
2.

Barsegyan Y.A., Baranov V.V., Kravchenko A.N.
Chemistry of Heterocyclic Compounds,
2017
3.
![Synthesis, Structure, and Biological Activity of 2,6-Disubstituted 2,3a,4a,6,7a,8a-Hexaazaperhydrocyclopenta[ def ]fluorene-4-thioxo-8-ones](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
Barsegyan Y., Baranov V., Kravchenko A., Strelenko Y., Anikina L., Karnoukhova V., Kolotyrkina N.
Synthesis,
2018
4.

Ivanov E.V., Lebedeva E.Y., Petrovskaya S.G., Baranov V.V., Kravchenko A.N., Ivanova N.G.
Journal of Molecular Liquids,
2017
5.
Anikina L.V., Vikharev Y.B., Baranov V.V., Malyshev O.R., Kravchenko A.N.
Mendeleev Communications,
2018
6.

New access to thioglycolurils by condensation of 4,5-dihydroxyimidazolidin-2-ones(thiones) with HSCN
Baranov V.V., Nelyubina Y.V., Kravchenko A.N., Kolotyrkina N.G., Biriukova K.A.
Tetrahedron Letters,
2015
7.
![Helianthus-like cucurbit[4]uril and cucurbit[5]uril analogues](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Wu Y., Xu L., Shen Y., Wang Y., Wang Q.
New Journal of Chemistry,
2017
8.

Shen Y., Zou L., Wang Q.
New Journal of Chemistry,
2017
9.

Wu Y., Xu L., Shen Y., Wang Y., Zou L., Wang Q., Jiang X., Liu J., Tian H.
Chemical Communications,
2017
10.

Bugnet E.A., Nixon T.D., Kilner C.A., Greatrex R., Kee T.P.
Tetrahedron Letters,
2003
11.

Abelman M.M., Smith S.C., James D.R.
Tetrahedron Letters,
2003
12.

Xu Z., Tu Y.
Chinese Journal of Organic Chemistry,
2016
13.
![The Effect of Aryl Substituents in Some Spiro[2-oxo-4, 6-bis (aryl)hexahydropyrimidine-5,5′-barbituric acids]](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Mokrosz J.L., Paluchowska M.H., Szneler E., Drozdz B.
Archiv der Pharmazie,
1989
14.
10.1016/j.mencom.2019.01.032_bib0025
Jetti
J. Chem. Pharm. Res.,
2012
15.

Gupta P., Gupta S., Sachar A., Sharma R.L.
Journal of Heterocyclic Chemistry,
2010
16.

Prajapati D., Bhuyan D., Gohain M., Hu W.
Molecular Diversity,
2010
17.

Shaabani A., Bazgir A., Bijanzadeh H.R.
Molecular Diversity,
2004
18.
10.1016/j.mencom.2019.01.032_bib0045
Jetti
J. Catal.,
2013
19.

Shaabani A., Bazgir A.
Tetrahedron Letters,
2004
20.

Amini M.M., Shaabani A., Bazgir A.
Catalysis Communications,
2006
21.

Kaur G., Gupta P., Harjai K., Singh V.
Drug Development Research,
2014
22.

23.
Belyakov P.A., Kadentsev V.I., Chizhov A.O., Kolotyrkina N.G., Shashkov A.S., Ananikov V.P.
Mendeleev Communications,
2010
24.

Valizadeh H., Dinparast L.
Monatshefte fur Chemie,
2011
25.

Abdul Hai S.M., Perveen S., Khan R.A., Khan K.M., Afza N.
Natural Product Research,
2003