Abstract
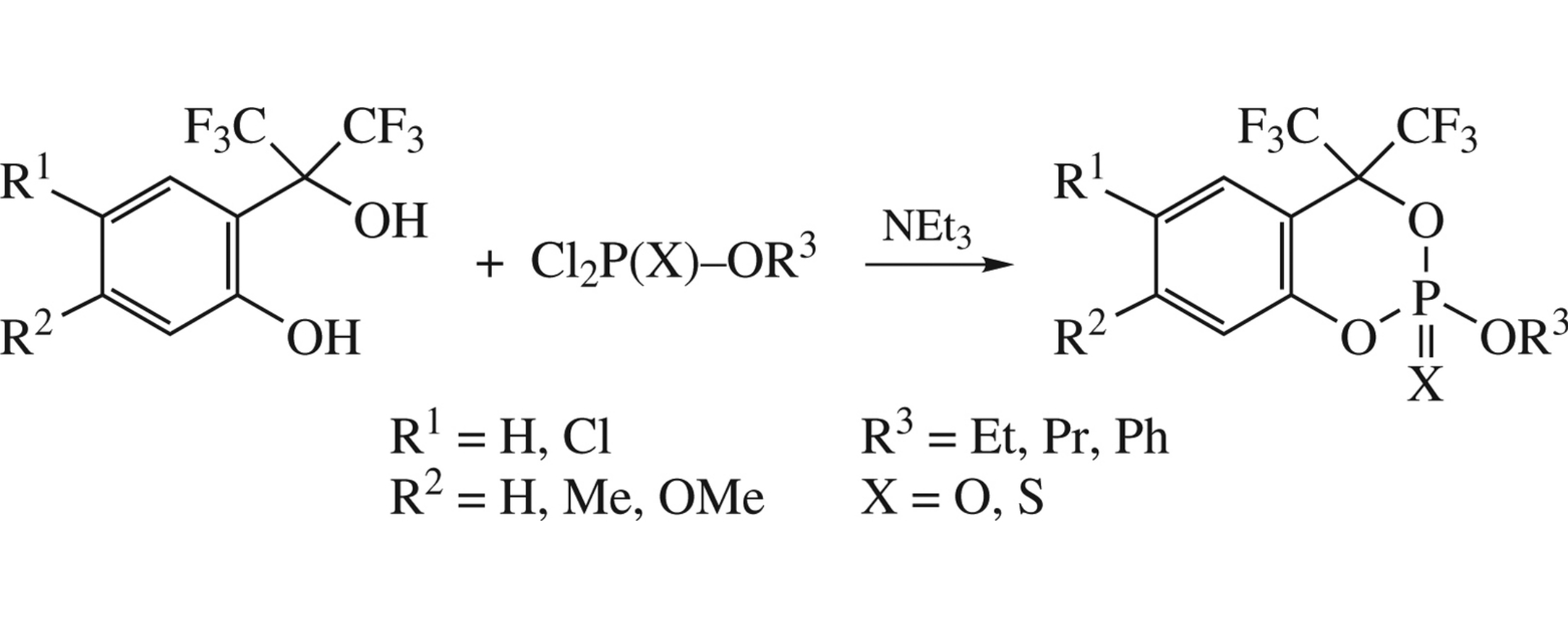
Substituted α,α-bis(trifluoromethyl)salicylic alcohols react with dichlorido(thio)phosphates in the presence of NEt3 to afford 2-alkoxy-4,4-bis(trifluoromethyl)-4H-1,3,2-benzodioxaphosphinine 2-oxide(sulfides) in high yields. The latter can be of interest as potential cholinesterase inhibitors.
References
1.

2.

Semina I.I., Baichurina A.Z., Garaev R.S.
Bulletin of Experimental Biology and Medicine,
1996
3.

Bajgar J.
Advances in Clinical Chemistry,
2004
4.

Smulders C.J.
Toxicological Sciences,
2004
5.

Böhm H., Banner D., Bendels S., Kansy M., Kuhn B., Müller K., Obst-Sander U., Stahl M.
ChemBioChem,
2004
6.
10.1016/j.mencom.2019.01.030_bib0030
Hiyama
Organofluorine Compounds: Chemistry and Applications,
2000
7.
10.1016/j.mencom.2019.01.030_sbref0035a
Nifant’ev
Russ. J. Gen. Chem.,
1993
8.

Nifantyev E.E., Kukhareva T.S., Dyachenko V.I., Kolomietz A.O., Magomedova N.S., Belsky V.K., Vasyanina L.K.
Phosphorus, Sulfur and Silicon and the Related Elements,
1994
9.
10.1016/j.mencom.2019.01.030_bib0040
Nifant’ev
Russ. J. Gen. Chem.,
1996
10.

Farah B.S., Gilbert E.E., Litt M., Otto J.A., Sibilia J.P.
Journal of Organic Chemistry,
1965
11.

Dyachenko V.I., Kolomiets A.F., Fokin A.V.
Russian Chemical Bulletin,
1988
12.

Allen F.H., Kennard O., Watson D.G., Brammer L., Orpen A.G., Taylor R.
Journal of the Chemical Society Perkin Transactions 2,
1987
13.
10.1016/j.mencom.2019.01.030_bib0060
Anticholinesterase Pesticides: Metabolism, Neurotoxicity and Epidemiology,
2011
14.
10.1016/j.mencom.2019.01.030_bib0065
Sheldrick
Acta Crystallogr.,
2015
15.
10.1016/j.mencom.2019.01.030_bib0070
Sheldrick
Acta Crystallogr.,
2015
16.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009