Abstract
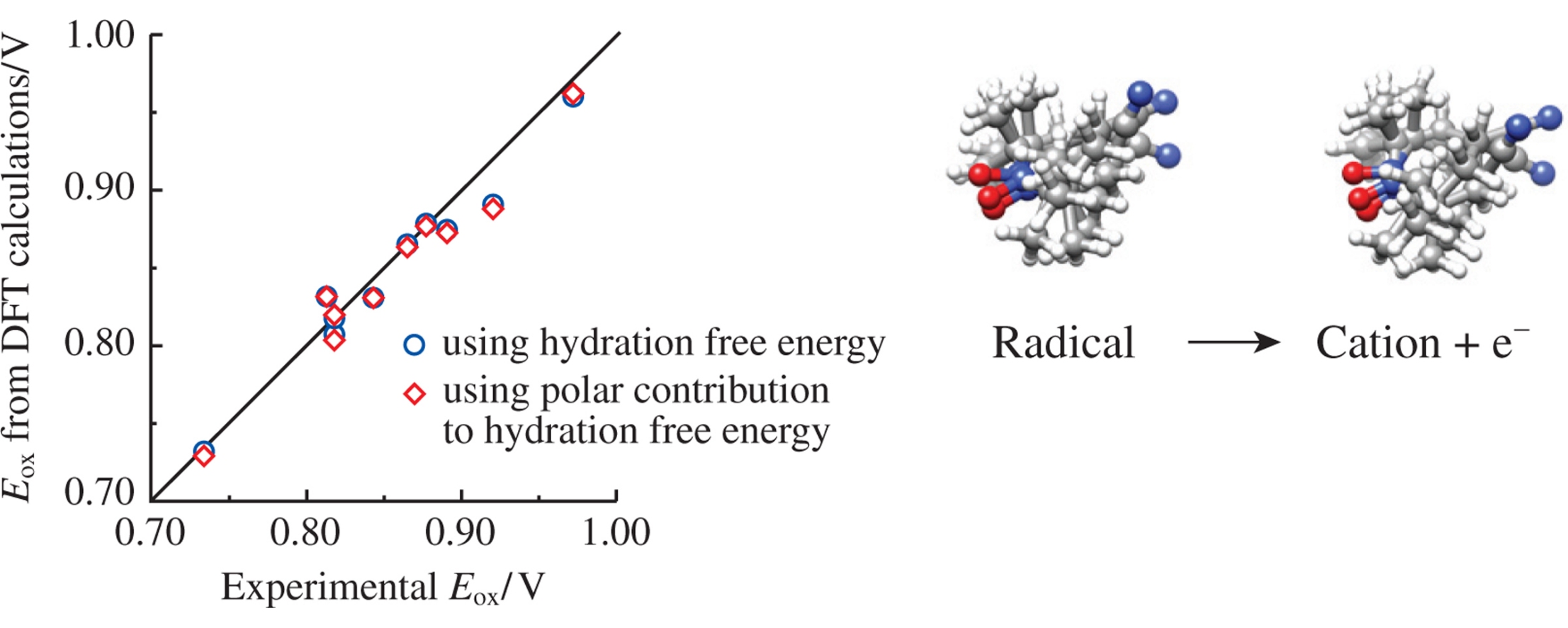
The oxidation potential Eox of cyclic nitroxide radicals in water has been calculated using density functional theory methods and the polarizable continuum solvent model. Robust correlations were obtained between the calculated and experimental Eox values with the lowest mean unsigned error of 10mV. The electrostatic model was demonstrated as a useful approximation for the calculations of Eox.
References
1.
10.1016/j.mencom.2019.01.026_bib0005
Golubev
Bioactive Spin Labels,
1992
2.

SOULE B., HYODO F., MATSUMOTO K., SIMONE N., COOK J., KRISHNA M., MITCHELL J.
Free Radical Biology and Medicine,
2007
3.

4.

Wilcox C.S.
Pharmacology and Therapeutics,
2010
5.

Tebben L., Studer A.
Angewandte Chemie - International Edition,
2011
6.
Zlotin S.G., Churakov A.M., Dalinger I.L., Luk’yanov O.A., Makhova N.N., Sukhorukov A.Y., Tartakovsky V.A.
Mendeleev Communications,
2017
7.

Nutting J.E., Rafiee M., Stahl S.S.
Chemical Reviews,
2018
8.

Mitchell J.B., Samuni A., Krishna M.C., DeGraff W.G., Ahn M.S., Samuni U., Russo A.
Biochemistry,
1990
9.

Griesser M., Shah R., Van Kessel A.T., Zilka O., Haidasz E.A., Pratt D.A.
Journal of the American Chemical Society,
2018
10.

Sen' V.D., Sokolova E.M., Neshev N.I., Kulikov A.V., Pliss E.M.
Reactive and Functional Polymers,
2017
11.

Mendkovich A.S., Luzhkov V.B., Syroeshkin M.A., Sen´ V.D., Khartsii D.I., Rusakov A.I.
Russian Chemical Bulletin,
2017
12.

Hodgson J.L., Namazian M., Bottle S.E., Coote M.L.
Journal of Physical Chemistry A,
2007
13.

Blinco J.P., Hodgson J.L., Morrow B.J., Walker J.R., Will G.D., Coote M.L., Bottle S.E.
Journal of Organic Chemistry,
2008
14.

Hickey D.P., Schiedler D.A., Matanovic I., Doan P.V., Atanassov P., Minteer S.D., Sigman M.S.
Journal of the American Chemical Society,
2015
15.

Zhang K., Noble B.B., Mater A.C., Monteiro M.J., Coote M.L., Jia Z.
Physical Chemistry Chemical Physics,
2018
16.
Levitskiy O.A., Magdesieva T.V.
Mendeleev Communications,
2018
17.

Yamasaki T., Mito F., Ito Y., Pandian S., Kinoshita Y., Nakano K., Murugesan R., Sakai K., Utsumi H., Yamada K.
Journal of Organic Chemistry,
2010
18.

Billone P.S., Johnson P.A., Lin S., Scaiano J.C., DiLabio G.A., Ingold K.U.
Journal of Organic Chemistry,
2010
19.

Sen’ V.D., Tikhonov I.V., Borodin L.I., Pliss E.M., Golubev V.A., Syroeshkin M.A., Rusakov A.I.
Journal of Physical Organic Chemistry,
2014
20.

Marenich A.V., Ho J., Coote M.L., Cramer C.J., Truhlar D.G.
Physical Chemistry Chemical Physics,
2014
21.

Becke A.D.
Journal of Chemical Physics,
1993
22.

Lee C., Yang W., Parr R.G.
Physical Review B,
1988
23.

Zhao Y., Schultz N.E., Truhlar D.G.
Journal of Chemical Theory and Computation,
2006
24.

Zhao Y., Truhlar D.G.
Theoretical Chemistry Accounts,
2007
25.

Dunning T.H.
Journal of Chemical Physics,
1989
26.

Marenich A.V., Cramer C.J., Truhlar D.G.
Journal of Physical Chemistry B,
2009
27.

Martins F.A., Silla J.M., Freitas M.P.
Carbohydrate Research,
2017
28.

Korneichuk A.Y., Senyavin V.M., Kuramshina G.M.
Russian Journal of Physical Chemistry A,
2017