Abstract
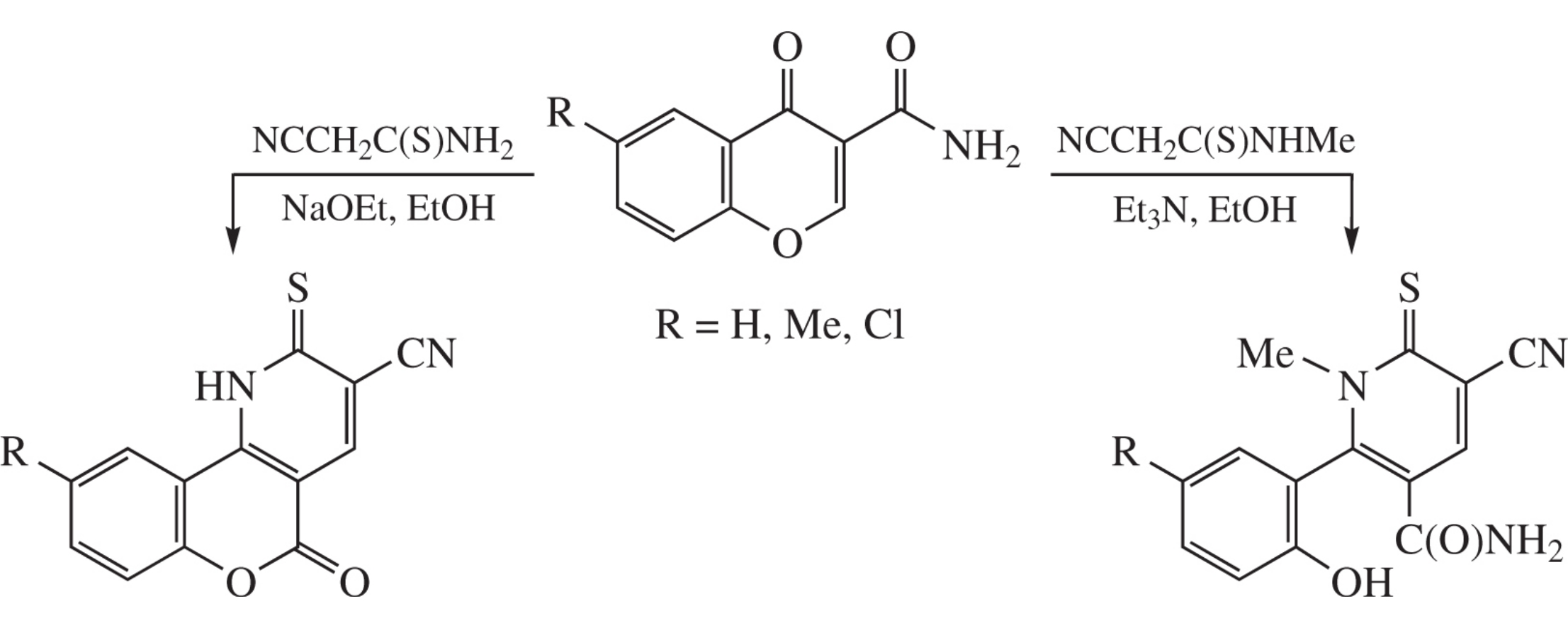
Chromone-3-carboxamides react with cyanothioacetamide in the presence of NaOEt in boiling ethanol to form 5-oxo-2-thioxo-1,5-dihydro-2H-chromeno[4,3-b]pyridine-3-carbonitriles (67–87% yields), whereas their reaction with N-methylcyanothioacetamide and a catalytic amount of Et3N in boiling ethanol affords 5-cyano-2-(2-hydroxyphenyl)-1-methyl-6-thioxo-1,6-dihydropyridine-3-carboxamides in 57–73% yields.
References
1.

Dotsenko V.V., Krivokolysko S.G., Polovinko V.V., Litvinov V.P.
Chemistry of Heterocyclic Compounds,
2012
2.

Dyachenko V.D., Dyachenko I.V., Nenajdenko V.G.
Russian Chemical Reviews,
2018
3.

Szabo M., Klein Herenbrink C., Christopoulos A., Lane J.R., Capuano B.
Journal of Medicinal Chemistry,
2014
4.

Narushyavichus É.V., Garalene V.N., Krauze A.A., Dubur G.Y.
Pharmaceutical Chemistry Journal,
1989
5.

Antczak M.I., Zhang Y., Wang C., Doran J., Naidoo J., Voruganti S., Williams N.S., Markowitz S.D., Ready J.M.
Journal of Medicinal Chemistry,
2017
6.

Krauze A.A., Bomika Z.A., Shestopalov A.M., Rodinovskaya L.A., Pelcher Y.�., Dubur G.Y., Sharanin Y.A., Promonenkov V.K.
Chemistry of Heterocyclic Compounds,
1981
7.
10.1016/j.mencom.2019.01.022_bib0035
Hagen
Pharmazie,
1989
8.
![Synthesis and anti-tumor activities of some new pyridines and pyrazolo[1,5-a]pyrimidines](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Ahmed O.M., Mohamed M.A., Ahmed R.R., Ahmed S.A.
European Journal of Medicinal Chemistry,
2009
9.

Kornev M.Y., Sosnovskikh V.Y.
Chemistry of Heterocyclic Compounds,
2016
10.
Demin D.Y., Myannik K.A., Ermolich P.A., Krayushkin M.M., Yarovenko V.N.
Mendeleev Communications,
2018
11.
10.1016/j.mencom.2019.01.022_bib0050
Ibrahim
J. Braz. Chem. Soc.,
2013
12.
Kornev M.Y., Moshkin V.S., Eltsov O.S., Sosnovskikh V.Y.
Mendeleev Communications,
2016
13.

Kornev M.Y., Moshkin V.S., Sosnovskikh V.Y.
Chemistry of Heterocyclic Compounds,
2015
14.

Dankova E.F., Bakulev V.A., Grishakov A.N., Mokrushin V.S.
Russian Chemical Bulletin,
1988
15.

Stefaniak L.
Magnetic Resonance in Chemistry,
1979
16.
10.1016/j.mencom.2019.01.022_sbref0070b
Gao
Synthesis,
2012