Abstract
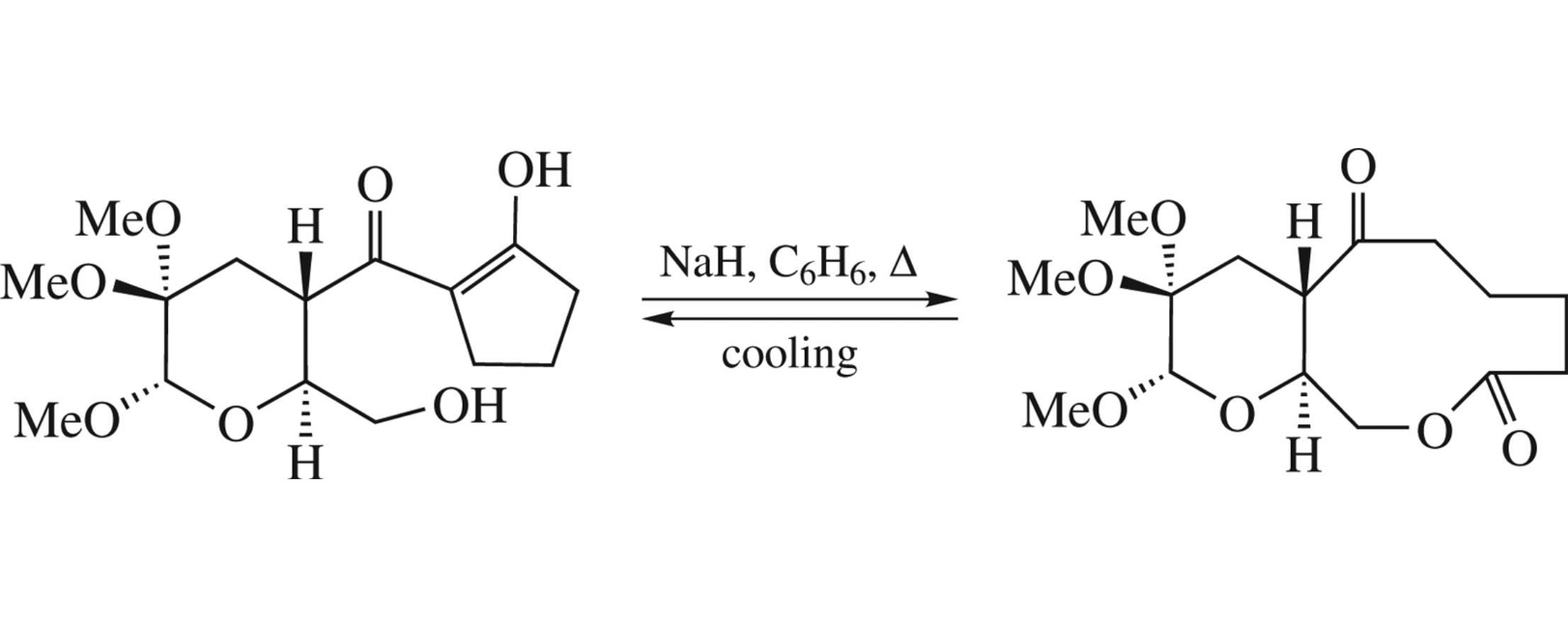
Levoglucosenone-derived 2-[(2S,3S,6S)-2-hydroxymethyl-5,5,6-trimethoxytetrahydro-2H-pyran-3-ylcarbonyl]cyclo-pentanone upon boiling in benzene in the presence of NaH undergoes the retro-Dieckmann-type reaction to afford 10-membered lactone, while storing this reaction mixture at room temperature brings about the starting cyclopentanone derivative. Several structurally analogous compounds were examined in respect of similar transformations.
References
1.
Sharipov B.T., Pershin A.A., Valeev F.A.
Mendeleev Communications,
2017
2.
Biktagirov I.M., Faizullina L.K., Salikhov S.M., Galin F.Z., Valeev F.A.
Mendeleev Communications,
2017
3.
Faizullina L.K., Galimova Y.S., Khalilova Y.A., Salikhov S.M., Valeev F.A.
Mendeleev Communications,
2018
4.

MAHAJAN J.R.
Synthesis,
1976
5.

Mahajan J.R., Resck I.S.
Synthesis,
1980
6.

Scott P.W., Harrison I.T., Bittner S.
Journal of Organic Chemistry,
1981
7.

Honeychuck R.V., Bonnesen P.V., Farahi J., Hersh W.H.
Journal of Organic Chemistry,
1987
8.

Suginome H., Yamada S.
Tetrahedron,
1987
9.
10.1016/j.mencom.2019.01.021_bib0015
Ho
Heterolytic Fragmentation of Organic Molecules,
1993
10.

Tagirov A.R., Fayzullina L.K., Enikeeva D.R., Galimova Y.S., Salikhov S.M., Valeev F.A.
Russian Journal of Organic Chemistry,
2018
11.

Galimova Y.S., Tagirov A.R., Faizullina L.K., Salikhov S.M., Valeev F.A.
Russian Journal of Organic Chemistry,
2017
12.

Khalilova Y.A., Tagirov A.R., Doronina O.Y., Spirikhin L.V., Salikhov S.M., Valeev F.A.
Russian Journal of Organic Chemistry,
2014
13.
14.

Smith A.B., Levenberg P.A., Jerris P.J., Scarborough R.M., Wovkulich P.M.
Journal of the American Chemical Society,
1981
15.

Karim M.R., Sampson P.
Tetrahedron Letters,
1988
16.

Luche J.L., Petrier C., Dupuy C.
Tetrahedron Letters,
1984
17.

Biktagirov I.M., Faizullina L.K., Salikhov S.M., Safarov M.G., Valeev F.A.
Russian Journal of Organic Chemistry,
2015