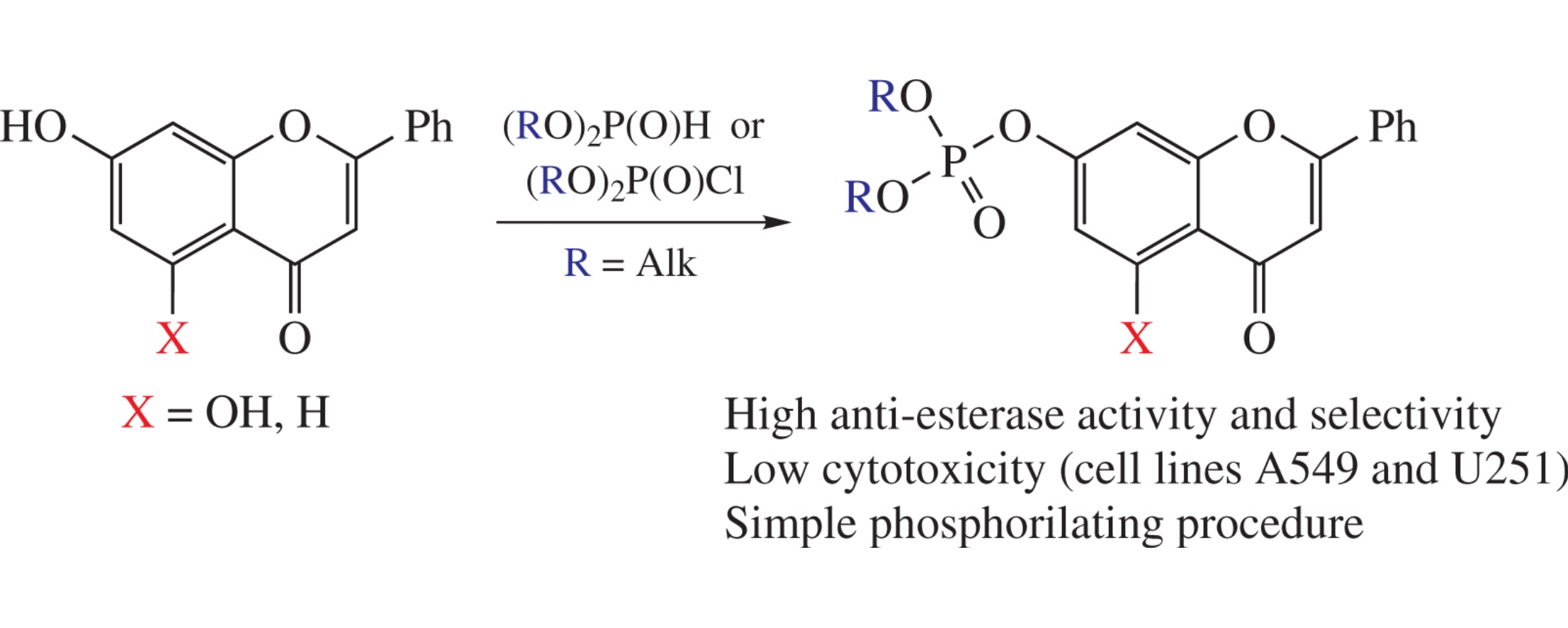
Abstract
A series of phosphorylated flavonoids has been synthesized and evaluated in vitro for inhibitory activity against carboxylesterase, acetylcholinesterase and butyrylcholinesterase as well as for their cytotoxicity towards human adenocarcinoma A549 and human glioblastoma U251 cell lines. Diethylphosphoryl derivatives of chrysin and 7-hydroxyflavone were found to be the most effective with bimolecular rate constants for carboxylesterase inhibition ki=2.0×106 and 5.7×106dm3mol−1min−1, respectively.
References
1.

Zhou Y., Zheng J., Li Y., Xu D., Li S., Chen Y., Li H.
Nutrients,
2016
2.
10.1016/j.mencom.2019.01.020_sbref0005b
Mileo
Oxid. Med. Cell. Longevity,
2016
3.

Bensasson R.V., Zoete V., Jossang A., Bodo B., Arimondo P.B., Land E.J.
Free Radical Biology and Medicine,
2011
4.

Boege F., Straub T., Kehr A., Boesenberg C., Christiansen K., Andersen A., Jakob F., Köhrle J.
Journal of Biological Chemistry,
1996
5.

Zemanova L., Hofman J., Novotna E., Musilek K., Lundova T., Havrankova J., Hostalkova A., Chlebek J., Cahlikova L., Wsol V.
Journal of Natural Products,
2015
6.

Kasala E.R., Bodduluru L.N., Madana R.M., V A.K., Gogoi R., Barua C.C.
Toxicology Letters,
2015
7.

Jiang Y., Du Z., Xue G., Chen Q., Lu Y., Zheng X., Conney A., Zhang K.
Molecules,
2013
8.

Kukongviriyapan V., Phromsopha N., Tassaneeyakul W., Kukongviriyapan U., Sripa B., Hahnvajanawong V., Bhudhisawasdi V.
Xenobiotica,
2006
9.

Peluso M.R.
Experimental Biology and Medicine,
2006
10.

Brás N.F., Gonçalves R., Mateus N., Fernandes P.A., Ramos M.J., de Freitas V.
Journal of Agricultural and Food Chemistry,
2010
11.

Griffiths D.W.
Advances in Experimental Medicine and Biology,
1986
12.
10.1016/j.mencom.2019.01.020_sbref0030b
Padilla-Camberos
BioMed Res. Int.,
2015
13.

Buchholz T., Zhang X., Melzig M.
Planta Medica,
2015
14.

Srivastava A.K.
Biochemical and Biophysical Research Communications,
1985
15.

Sudan S., Rupasinghe H.P.
Nutrition and Cancer,
2014
16.
10.1016/j.mencom.2019.01.020_sbref0040b
Sudan
Anticancer Res.,
2014
17.

MIZUSHINA Y., SHIOMI K., KURIYAMA I., TAKAHASHI Y., YOSHIDA H.
International Journal of Oncology,
2013
18.

Wei Y., Peng A., Wang B., Ma L., Peng G., Du Y., Tang J.
European Journal of Medicinal Chemistry,
2014
19.

Djeridane A., Brunel J.M., Vidal N., Yousfi M., Ajandouz E.H., Stocker P.
Chemico-Biological Interactions,
2008
20.

Wei Y., Peng A., Huang J.
Chemico-Biological Interactions,
2013
21.

Potter P., Wadkins R.
Current Medicinal Chemistry,
2006
22.

Li B., Sedlacek M., Manoharan I., Boopathy R., Duysen E.G., Masson P., Lockridge O.
Biochemical Pharmacology,
2005
23.

Bahar F.G., Ohura K., Ogihara T., Imai T.
Journal of Pharmaceutical Sciences,
2012
24.

Na K., Lee E., Lee H., Kim K., Lee H., Jeong S., Jeong A., Cho S.Y., Kim S.A., Song S.Y., Kim K.S., Cho S.W., Kim H., Paik Y.
Proteomics,
2009
25.

Koitka M., Höchel J., Gieschen H., Borchert H.
Journal of Pharmaceutical and Biomedical Analysis,
2010
26.

Law K., Acey R.A., Smith C.R., Benton D.A., Soroushian S., Eckenrod B., Stedman R., Kantardjieff K.A., Nakayama K.
Biochemical and Biophysical Research Communications,
2007
27.

Clement J.G.
Biochemical Pharmacology,
1984
28.

Maxwell D.M., Brecht K.M., O'Neill B.L.
Toxicology Letters,
1987
29.

Yang Z.P., Dettbarn W.
Biochemical Pharmacology,
1998
30.

Grubič Z., Sket D., Brzin M.
Archives of Toxicology,
1988
31.

Gupta R.C., Dettbarn W.-.
Archives of Toxicology,
1987
32.
10.1016/j.mencom.2019.01.020_bib0085
Prokofieva
Toksikol. Vestn.,
2016
33.

Skau K.A.
Comparative Biochemistry and Physiology Part C Comparative Pharmacology,
1985
34.

Carletti E., Schopfer L.M., Colletier J., Froment M., Nachon F., Weik M., Lockridge O., Masson P.
Chemical Research in Toxicology,
2011
35.

Makhaeva G.F., Aksinenko A.Y., Sokolov V.B., Baskin I.I., Palyulin V.A., Zefirov N.S., Hein N.D., Kampf J.W., Wijeyesakere S.J., Richardson R.J.
Chemico-Biological Interactions,
2010