Abstract
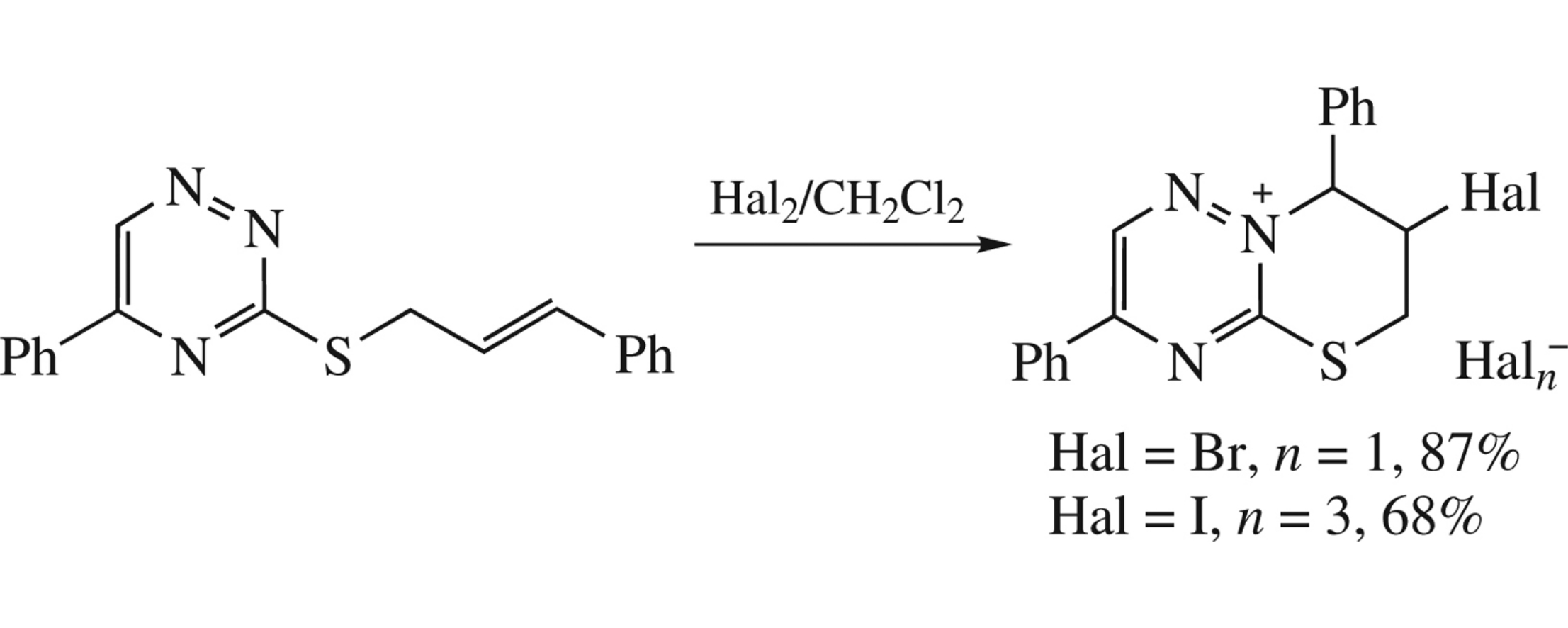
Treatment of trans-5-phenyl-3-cinnamylsulfanyl[1,2,4]triazine with bromine or iodine in dichloromethane leads to haloannulation onto the thiazine moiety with formation of (7r,8t)-7-halo-3,8-diphenyl-7,8-dihydro-6H-[1,3]thiazino[3,2-b][1,2,4]-triazin-9-ium salts. The product structure was confirmed by X-ray study.
References
1.

Savateev K.V., Ulomsky E.N., Butorin I.I., Charushin V.N., Rusinov V.L., Chupakhin O.N.
Russian Chemical Reviews,
2018
2.

Fershtat L.L., Makhova N.N.
Russian Chemical Reviews,
2016
3.
![New Conversions and Functionalization Possibilities in Pyrimido[4,5-e][1,2,4]triazine-6,8-diones](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Azev Y.A., Gabel D., Doerfler U., Él'‐Zaria M.E., Bauer K., Shorshnev S.V., Klyuev N.A.
Pharmaceutical Chemistry Journal,
2003
4.
![The first stable examples of compounds containing both diazonium and acyl azide, and synthesis of a new pyrazino[2′,3′:3,4]pyrazolo[5,1- c ][1,2,4]triazin-4(6 H )-one heterocyclic system](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Ivanov S.M., Mironovich L.M., Rodinovskaya L.A., Shestopalov A.M.
Tetrahedron Letters,
2017
5.

Ali R.S., Al Harthi R.R., Saad H.A., Amin M.A.
Russian Journal of General Chemistry,
2016
6.

Rezaei B., Fazlollahi M.
Chemistry Central Journal,
2013
7.

Smagin S.S., Bogachev V.E., Yakubovskii A.K., Metkalova S.E., Privol'neva T.P., Chugunov V.V., Lavretskaya É.F.
Pharmaceutical Chemistry Journal,
1975
8.

Tuylu Kucukkilinc T., Safari Yanghagh K., Ayazgok B., Ali Roknipour M., Homayouni Moghadam F., Moradi A., Emami S., Amini M., Irannejad H.
Medicinal Chemistry Research,
2017
9.
10.1016/j.mencom.2019.01.019_bib0045
Mizutani
Agric. Biol. Chem.,
1987
10.
![1,2,4-Triazino-[5,6b]indole derivatives: effects of the trifluoromethyl group on in vitro antimalarial activity](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Kgokong J.L., Smith P.P., Matsabisa G.M.
Bioorganic and Medicinal Chemistry,
2005
11.
10.1016/j.mencom.2019.01.019_bib0055
Polat
Turk. J. Chem.,
2013
12.
10.1016/j.mencom.2019.01.019_bib0060
Tehrani
Int. J. Anal. Chem.,
2014
13.
10.1016/j.mencom.2019.01.019_bib0065
Tehrani
Chem. Sci. Trans.,
2015
14.
Chepchugov N.V., Kopchuk D.S., Kovalev I.S., Zyryanov G.V., Rusinov V.L., Chupakhin O.N.
Mendeleev Communications,
2016
15.

Kopchuk D.S., Chepchugov N.V., Taniya O.S., Khasanov A.F., Giri K., Kovalev I.S., Santra S., Zyryanov G.V., Majee A., Rusinov V.L., Chupakhin O.N.
Tetrahedron Letters,
2016
16.

Kopchuk D.S., Chepchugov N.V., Kim G.A., Zyryanov G.V., Kovalev I.S., Rusinov V.L., Chupakhin O.N.
Tetrahedron Letters,
2016
17.
Kopchuk D.S., Krinochkin A.P., Kim G.A., Kozhevnikov D.N.
Mendeleev Communications,
2017
18.

Gazieva G.A., Karpova T.B., Nechaeva T.V., Kravchenko A.N.
Russian Chemical Bulletin,
2016
19.
Voinkov E.K., Ulomskiy E.N., Rusinov V.L., Drokin R.A., Fedotov V.V., Gorbunov E.B.
Mendeleev Communications,
2017
20.
![Synthesis of 3-methyl-7-phenylimidazo-[1,2-b]-1,2,4-triazine](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Rudakov B.V., Kim D.G., Aleskeev S.G.
Chemistry of Heterocyclic Compounds,
1998
21.
![Halocyclization of 3-allylthio-5H-[1,2,4]triazino[5,6-b]indole](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Kim D.G., Zhuravlyova A.V.
Chemistry of Heterocyclic Compounds,
2010
22.
![Regioselectivity of cyclization of 3-allyl(propargyl)sulfanyl-5H-[1,2,4]triazino[5,6-b]indoles](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Vasˈkevich R.I., Vasˈkevich A.I., Turov A.V., Staninets V.I., Vovk M.V.
Chemistry of Heterocyclic Compounds,
2011
23.
![Direction of the heterocyclization reaction of 3-allyl- and 3-propargyl-sulfanyl-5H-[1,2,4]triazino[5,6-b]indoles](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Rybakova A.V., Slepukhin P.A., Kim D.G.
Chemistry of Heterocyclic Compounds,
2013
24.
![Synthesis of novel [1,3]thiazino[3´,2´:2,3][1,2,4]triazino[5,6-b]indole derivatives](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Rybakova A.V., Kim D.G., Ezhikova M.A., Kodess M.I., Taherc I.A.
Russian Chemical Bulletin,
2015
25.
10.1016/j.mencom.2019.01.019_bib0125
SMART and SAINT-Plus, Versions 5.0,
1998
26.
10.1016/j.mencom.2019.01.019_bib0130
SHELXTL/PC, Versions 5.10,
1998
27.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
28.

Mantina M., Chamberlin A.C., Valero R., Cramer C.J., Truhlar D.G.
Journal of Physical Chemistry A,
2009