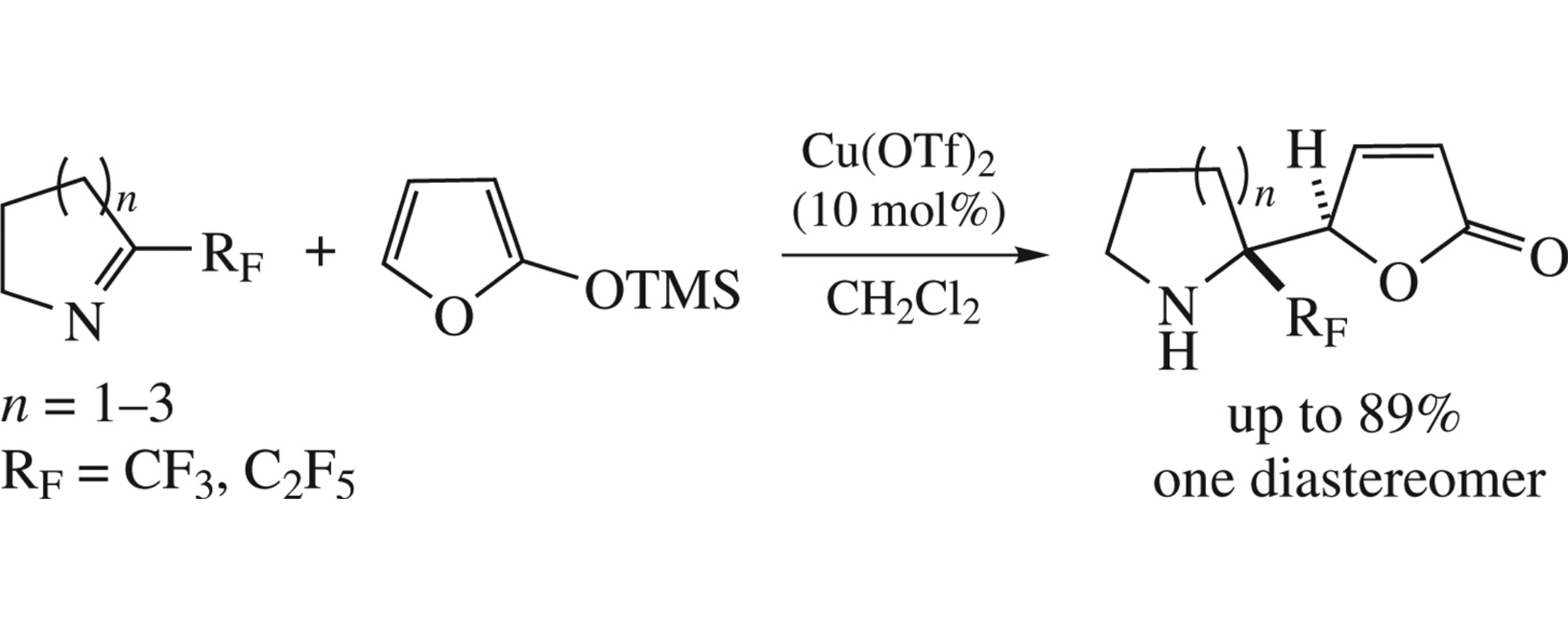
Abstract
The reaction between 2-trimethylsilyloxyfuran and perfluoroalkylated cyclic ketimines with different ring sizes affords 5-(2-perfluoroalkyl-1-azacycloalk-2-yl)furan-2(5H)-ones in yields up to 89% and with excellent rel-(R,R)-diastereoselectivity.
References
1.

Salwiczek M., Nyakatura E.K., Gerling U.I., Ye S., Koksch B.
Chemical Society Reviews,
2012
2.

Ahrens T., Kohlmann J., Ahrens M., Braun T.
Chemical Reviews,
2014
3.

Wang J., Sánchez-Roselló M., Aceña J.L., del Pozo C., Sorochinsky A.E., Fustero S., Soloshonok V.A., Liu H.
Chemical Reviews,
2013
4.

Zhou Y., Wang J., Gu Z., Wang S., Zhu W., Aceña J.L., Soloshonok V.A., Izawa K., Liu H.
Chemical Reviews,
2016
5.

Tomashenko O.A., Grushin V.V.
Chemical Reviews,
2011
6.

Shibata N., Matsnev A., Cahard D.
Beilstein Journal of Organic Chemistry,
2010
7.

Ma J., Cahard D.
Chemical Reviews,
2004
8.

Oishi M., Kondo H., Amii H.
Chemical Communications,
2009
9.

Prakash G.K., Yudin A.K.
Chemical Reviews,
1997
10.
Kuvshinova S.S., Smol’yakov A.F., Vorobyeva D.V., Osipov S.N., Loginov D.A.
Mendeleev Communications,
2018
11.
Ivanova A.E., Burgart Y.V., Saloutin V.I., Orshanskaya Y.R., Zarubaev V.V.
Mendeleev Communications,
2018
12.
10.1016/j.mencom.2019.01.018_sbref0015a
Shevchenko
Synthesis,
2010
13.

Shevchenko N.E., Nenajdenko V.G., Röschenthaler G.
Journal of Fluorine Chemistry,
2008
14.

Shmatova O.I., Nenajdenko V.G.
European Journal of Organic Chemistry,
2013
15.

Shmatova O.I., Shevchenko N.E., Nenajdenko V.G.
European Journal of Organic Chemistry,
2015
16.
Shmatova O.I., Shevchenko N.E., Balenkova E.S., Röschenthaler G., Nenajdenko V.G.
Mendeleev Communications,
2013
17.

Shmatova O.I., Khrustalev V.N., Nenajdenko V.G.
Organic Letters,
2016
18.

Shmatova O.I., Shevchenko N.E., Balenkova E.S., Röschenthaler G., Nenajdenko V.G.
European Journal of Organic Chemistry,
2013
19.
Kutovaya I.V., Shmatova O.I., Nenajdenko V.G.
Mendeleev Communications,
2018
20.

Takayama H., Ichikawa T., Kitajima M., Nonato M.G., Aimi N.
Journal of Natural Products,
2001
21.

Blanco P., Busqué F., de March P., Figueredo M., Font J., Sanfeliu E.
European Journal of Organic Chemistry,
2004
22.

Busqué F., de March P., Figueredo M., Font J., Sanfeliu E.
Tetrahedron Letters,
2002
23.

Ye J., Zhang Y., Liu Y., Zhang J., Ruan Y., Huang P.
Organic Chemistry Frontiers,
2015
24.

Liu L., Liu J.
Tetrahedron,
2014
25.

Spanedda M.V., Ourévitch M., Crousse B., Bégué J., Bonnet-Delpon D.
Tetrahedron Letters,
2004
26.

27.

Zhao Q., Yuan Z., Shi M.
Tetrahedron Asymmetry,
2010
28.

Zhao Q., Yuan Z., Shi M.
Advanced Synthesis and Catalysis,
2011
29.

Yu J., Miao Z., Chen R.
Organic and Biomolecular Chemistry,
2011
30.
10.1016/j.mencom.2019.01.018_bib0035
Frisch
Gaussian 03W, Revision B.05,
2003