Abstract
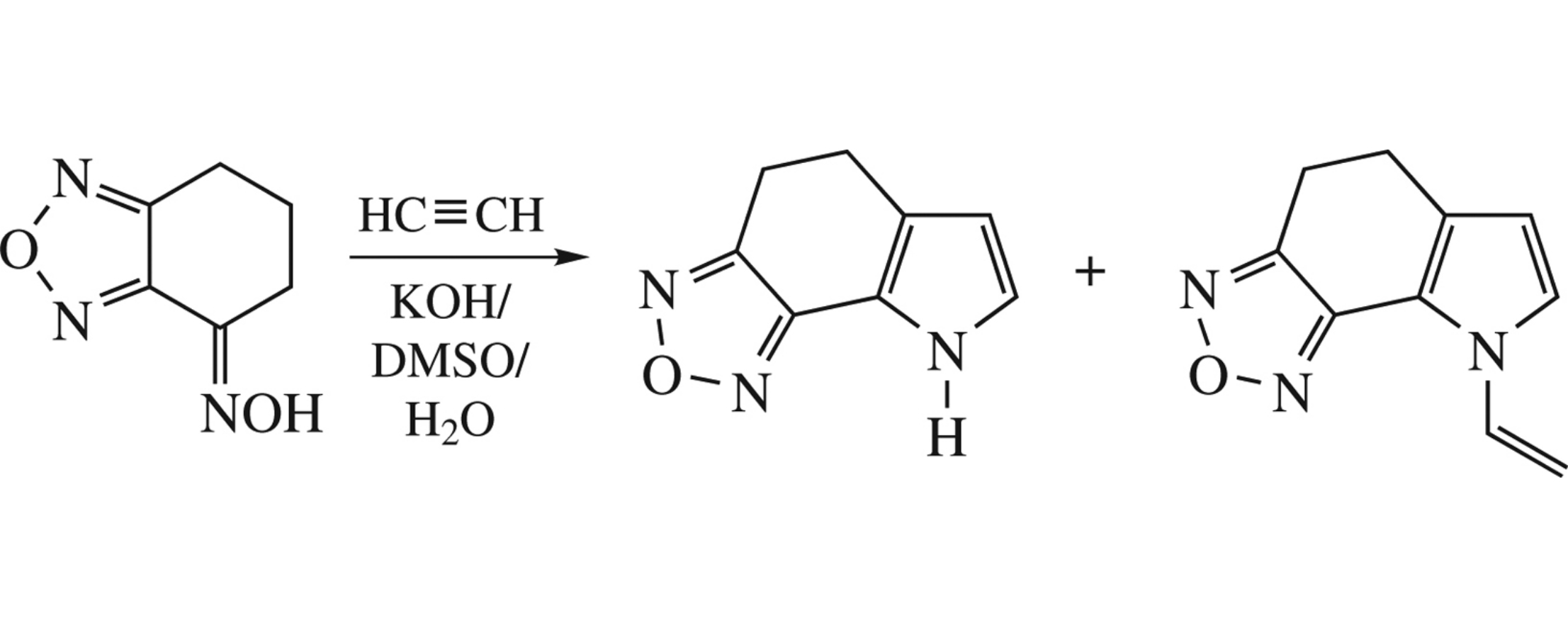
6,7-Dihydrobenzo[c][1,2,5]oxadiazol-4(5H)-one oxime is annelated with acetylene in the KOH/DMSO/H2O system under pressure to give NH- or N-vinyl-5,8-dihydro-4H-[1,2,5]oxadiazolo[3,4-g]indoles in 29 and 68% yields, respectively. These derivatives are readily aromatized with DDQ at room temperature to the expected indoles.
References
1.

Sheremetev A.B.
Russian Chemical Reviews,
1999
2.

Fershtat L.L., Makhova N.N.
Russian Chemical Reviews,
2016
3.

Christoff R.M., Murray G.L., Kostoulias X.P., Peleg A.Y., Abbott B.M.
Bioorganic and Medicinal Chemistry,
2017
4.

Lai Y., Shen L., Zhang Z., Liu W., Zhang Y., Ji H., Tian J.
Bioorganic and Medicinal Chemistry Letters,
2010
5.

Chegaev K., Riganti C., Lazzarato L., Rolando B., Guglielmo S., Campia I., Fruttero R., Bosia A., Gasco A.
ACS Medicinal Chemistry Letters,
2011
6.

Zhao J., Gou S., Sun Y., Fang L., Wang Z.
Inorganic Chemistry,
2012
7.
![A facile synthesis and microtubule-destabilizing properties of 4-(1H-benzo[d]imidazol-2-yl)-furazan-3-amines](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Stepanov A.I., Astrat'ev A.A., Sheremetev A.B., Lagutina N.K., Palysaeva N.V., Tyurin A.Y., Aleksandrova N.S., Sadchikova N.P., Suponitsky K.Y., Atamanenko O.P., Konyushkin L.D., Semenov R.V., Firgang S.I., Kiselyov A.S., Semenova M.N., et. al.
European Journal of Medicinal Chemistry,
2015
8.

Li X., Wang X., Xu C., Huang J., Wang C., Wang X., He L., Ling Y.
MedChemComm,
2015
9.

Duan W., Li J., Inks E.S., Chou C.J., Jia Y., Chu X., Li X., Xu W., Zhang Y.
Journal of Medicinal Chemistry,
2015
10.

Rai G., Thomas C.J., Leister W., Maloney D.J.
Tetrahedron Letters,
2009
11.

Guglielmo S., Cortese D., Vottero F., Rolando B., Kommer V.P., Williams D.L., Fruttero R., Gasco A.
European Journal of Medicinal Chemistry,
2014
12.

Chegaev K., Lazzarato L., Tamboli Y., Boschi D., Blangetti M., Scozzafava A., Carta F., Masini E., Fruttero R., Supuran C.T., Gasco A.
Bioorganic and Medicinal Chemistry,
2014
13.
A. V. Anikin, V.R. Gantla, V.E. Gregor, L. Jiang, Y. Liu, D.P. C. Mcgee, C.C. Mikel, J.C. Pickens, T.R. Webb, Y. Zheng, T. Zhu, A. Kadushkin, S. Zozulya, A. Chucholowski, D.E. Mcgrath, S. Sviridov, Patent WO 2007056155A1, 2007.(Chem. Abstr., 2007, 146, 521800).
14.
O. Dirat, J.M. Elliott, A.P. Owens, D.E. Shaw, F. Sternfeld, B. J. Williams, Patent WO 2007003965, 2007.(Chem. Abstr., 2007, 146, 142372).
15.
W. B. Trotter, K.K. Nanda, S. Wolkenberg, B.M. Nolt, P. Manley, N.R. Kett, M. T. Bilodeau, Patent WO 2007050348, 2007.(Chem. Abstr., 2007, 146, 481931).
16.

Gonzalez F., Sanz-Cervera J.F., Williams R.M.
Tetrahedron Letters,
1999
17.
10.1016/j.mencom.2019.01.016_sbref0050b
Tedeschi
2001
18.
10.1016/j.mencom.2019.01.016_sbref0050c
The Chemistry of Hydroxylamines, Oximes and Hydroxamic Acids,
2008
19.
10.1016/j.mencom.2019.01.016_sbref0050d
Wang
Comprehensive Organic Name Reactions and Reagents,
2009
20.

Trofimov B.A., Mikhaleva A.I., Schmidt E.Y., Sobenina L.N.
Advances in Heterocyclic Chemistry,
2010
21.
10.1016/j.mencom.2019.01.016_sbref0050f
Modern Heterocyclic Chemistry,
2011
22.
10.1016/j.mencom.2019.01.016_sbref0050g
Name Reactions in Heterocyclic Chemistry II,
2011
23.

Samsonov V.A., Volodarskii L.B.
Chemistry of Heterocyclic Compounds,
1991
24.

Petrova O.V., Sobenina L.N., Budaev A.B., Ivanov A.V., Samsonov V.A., Tikhonov A.Y., Trofimov B.A.
Russian Journal of Organic Chemistry,
2017
25.
Petrova O.V., Sobenina L.N., Ushakov I.A., Budaev A.B., Ivanov A.V., Samsonov V.A., Tikhonov A.Y., Trofimov B.A.
Mendeleev Communications,
2017
26.

Giorgis M., Lolli M.L., Rolando B., Rao A., Tosco P., Chaurasia S., Marabello D., Fruttero R., Gasco A.
European Journal of Medicinal Chemistry,
2011
27.
10.1016/j.mencom.2019.01.016_bib0075
Trofimov
Chemistry of Pyrroles,
2014