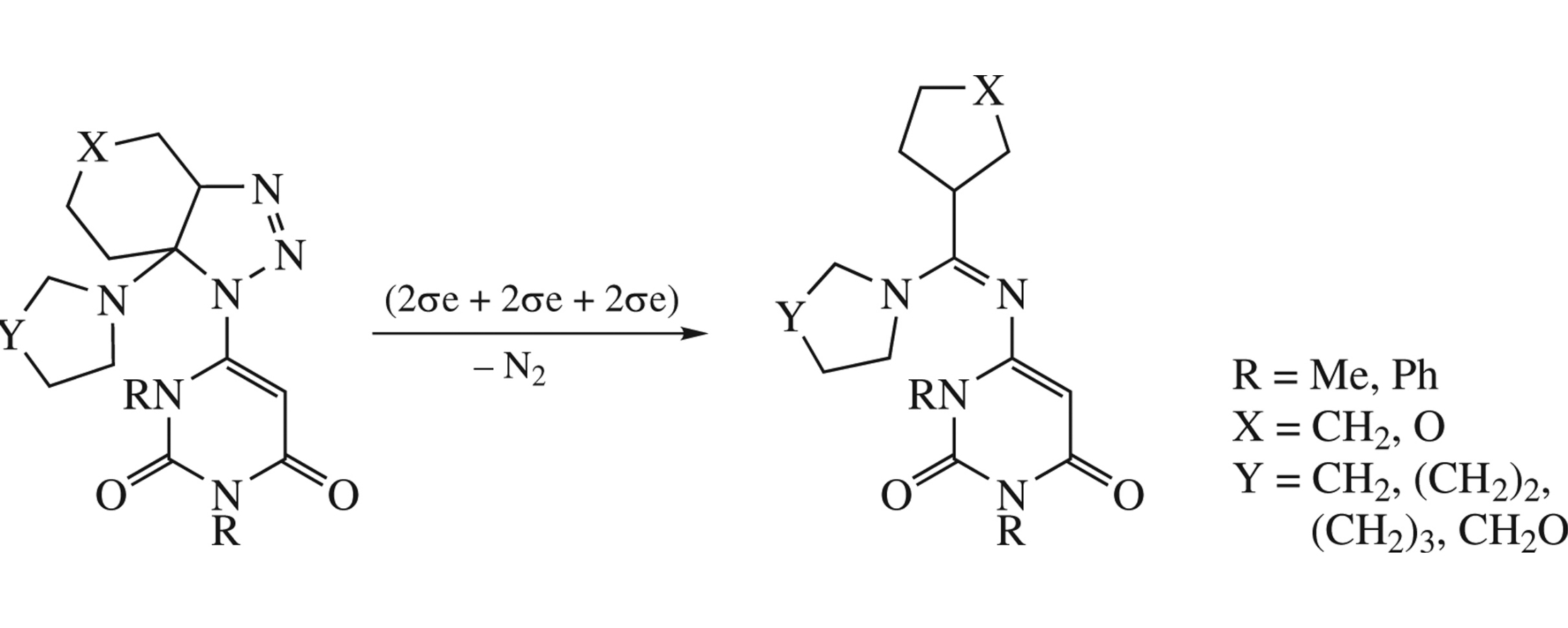
Abstract
Novel N-pyrimidyl amidines of alycyclic acids were obtained in one step from of 4-azidopyrimidines and endocyclic enamines. The reaction mechanism involves [3 + 2]-addition of azide at the double bond followed by cleavage of thus formed 1,2,3-triazoline ring, and a contraction of alicycle.
References
1.

Greenhill J.V., Lue P.
Progress in Medicinal Chemistry,
1993
2.
10.1016/j.mencom.2019.01.015_sbref0005b
Boyd
1991
3.

Donnini S., Monti M., Castagnini C., Solito R., Botta M., Schenone S., Giachetti A., Ziche M.
International Journal of Cancer,
2006
4.
![One-pot synthesis and antibacterial activities of pyrazolo[4′,3′:5,6]pyrido[2,3-d]pyrimidine-dione derivatives](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Bazgir A., Khanaposhtani M.M., Soorki A.A.
Bioorganic and Medicinal Chemistry Letters,
2008
5.

Shaban N.Z., Masoud M.S., Mawlawi M.A., Awad D., Sadek O.M.
Journal of Physiology and Biochemistry,
2012
6.

Pandey S., Suryawanshi S.N., Gupta S., M.L. Srivastava V.
European Journal of Medicinal Chemistry,
2004
7.

Amr A.E., Maigali S.S., Abdulla M.M.
Monatshefte fur Chemie,
2008
8.

Amr A.E., Mohamed A.M., Mohamed S.F., Abdel-Hafez N.A., Hammam A.E.
Bioorganic and Medicinal Chemistry,
2006
9.
B.J. Branstetter, J.G. Breitenbucher, A.D. Lebsack, W. Xiao, Patent WO 2008005303 A2, 2008.
10.

Flefel E.E., Salama M.A., El-Shahat M., El-Hashash M.A., El-Farargy A.F.
Phosphorus, Sulfur and Silicon and the Related Elements,
2007
11.
Verbitskiy E.V., Baskakova S.A., Gerasimova N.A., Evstigneeva N.P., Zil’berberg N.V., Kungurov N.V., Kravchenko M.A., Rusinov G.L., Chupakhina O.N., Charushin V.N.
Mendeleev Communications,
2018
12.

Liubchak K., Tolmachev A., Grygorenko O.O., Nazarenko K.
Tetrahedron,
2012
13.

Inamoto K., Shimizu M., Hayama N., Kimachi T.
Synthesis,
2017
14.

Dudkin S., Iaroshenko V.O., Sosnovskikh V.Y., Tolmachev A.A., Villinger A., Langer P.
Organic and Biomolecular Chemistry,
2013
15.
![A convenient synthesis of novel 5-aryl-pyrido[2,3-d]pyrimidines and screening of their preliminary antibacterial properties](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Saikia L., Das B., Bharali P., Thakur A.J.
Tetrahedron Letters,
2014
16.
![Recyclization in the Series of Spiro[indole-3,5′-pyrimido[4,5-b]quinoline]-2,2′,4′-triones Prepared by a Three-Component Reaction of Isatins with (Thio)barbituric Acids and Electron-Rich Anilines](/storage/images/resized/xqixcltwJYe6H8Uco2JbAFfIOzt7UNKH0OcPOPzO_small_thumb.webp)
Iaroshenko V., Dudkin S., Sosnovskikh V., Villinger A., Langer P.
Synthesis,
2013
17.

Efimov I., Beliaev N., Beryozkina T., Slepukhin P., Bakulev V.
Tetrahedron Letters,
2016
18.

Efimov I., Bakulev V., Beliaev N., Beryozkina T., Knippschild U., Leban J., Zhi-Jin F., Eltsov O., Slepukhin P., Ezhikova M., Dehaen W.
European Journal of Organic Chemistry,
2014
19.

Beliaev N.A., Shafikov M.Z., Efimov I.V., Beryozkina T.V., Lubec G., Dehaen W., Bakulev V.A.
New Journal of Chemistry,
2018
20.

Bakulev V.A., Beryozkina T., Thomas J., Dehaen W.
European Journal of Organic Chemistry,
2017
21.

Bakulev V.A., Efimov I.V., Belyaev N.A., Rozin Y.A., Volkova N.N., El’tsov O.S.
Chemistry of Heterocyclic Compounds,
2012
22.

Anderson A.A., Simonyan S.P., Lukevics E.
Chemistry of Heterocyclic Compounds,
1998
23.

Pellegrino S., Contini A., Gelmi M.L., Lo Presti L., Soave R., Erba E.
Journal of Organic Chemistry,
2014
24.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007