Abstract
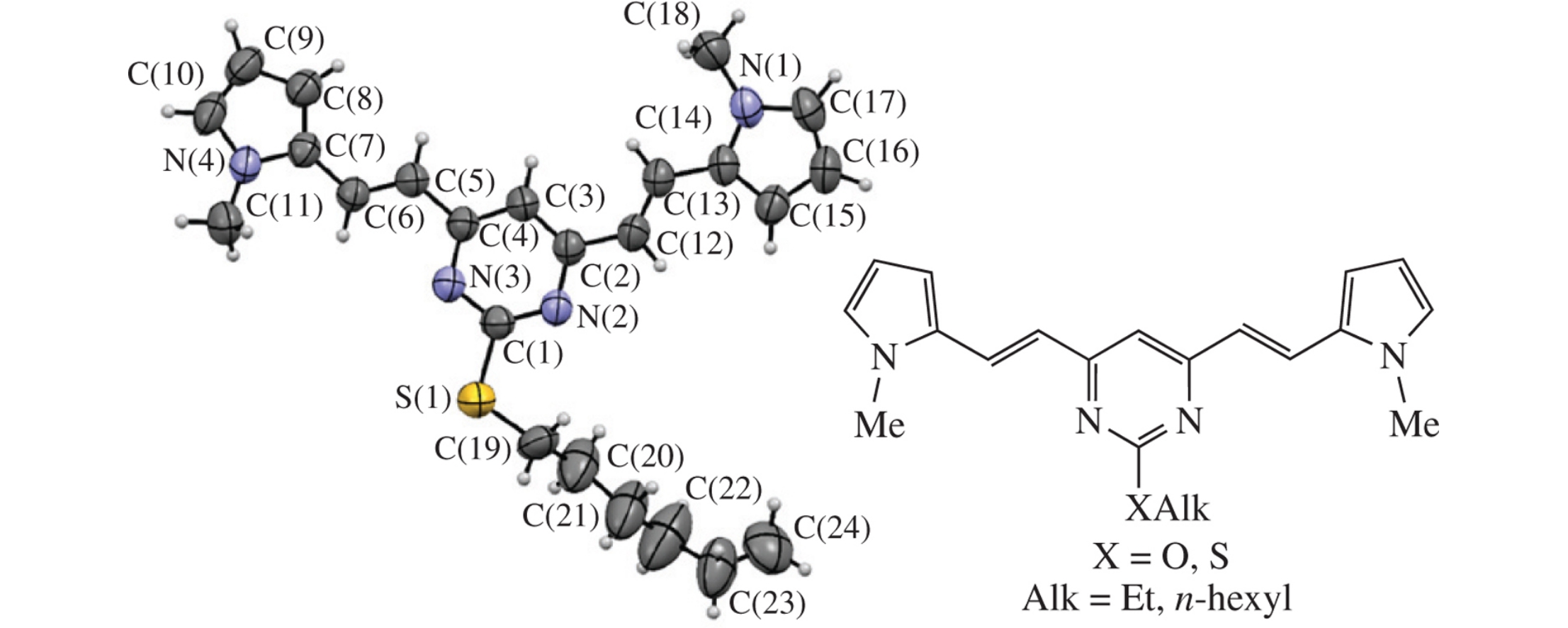
A series of novel Y-shaped dyes bearing the D–π–A–π–D fragment comprising central 2-alkoxy-or 2-alkylthio-substituted pyrimidine core and terminal 2-(1-methyl-1H-pyrrol-2-yl)vinyl moieties have been synthesized and studied as potential materials for the organic electronics. The obtained compounds demonstrated an efficient absorption at ∼410nm with the high molar absorption coefficient (29790–40620 dm3 mol−1 cm−1) and fluorescence emission at ∼480nm (ΦF ∼ 7.5–8.6%). Thin films of 2-ethyloxy- or 2-ethylthiopyrimidines exhibited the red-shifted photoluminescence (∼600nm).
References
1.

Kaur M., Ahn Y., Choi K., Cho M.J., Choi D.H.
Organic and Biomolecular Chemistry,
2015
2.

Kaur M., Cho M.J., Choi D.H.
Dyes and Pigments,
2016
3.

Achelle S., Nouira I., Pfaffinger B., Ramondenc Y., Plé N., Rodríguez-López J.
Journal of Organic Chemistry,
2009
4.
B. Schmidt, D. Kieser, A. Boländer, J. Herms, R. Heyny-Von Haussen, J. Gu, US Patent 0287700 A1, 2013.
5.

Kato S., Yamada Y., Hiyoshi H., Umezu K., Nakamura Y.
Journal of Organic Chemistry,
2015
6.

Verbitskiy E.V., Dinastiya E.M., Baranova A.A., Khokhlov K.O., Chuvashov R.D., Yakovleva Y.A., Makarova N.I., Vetrova E.V., Metelitsa A.V., Slepukhin P.A., Rusinov G.L., Chupakhin O.N., Charushin V.N.
Dyes and Pigments,
2018
7.

Achelle S., Rodríguez-López J., Cabon N., Guen F.R.
RSC Advances,
2015
8.

Kido J., Sasabe H., Komatsu R.
Journal of Photonics for Energy,
2018
9.

Gunathilake S.S., Huang P., Bhatt M.P., Rainbolt E.A., Stefan M.C., Biewer M.C.
RSC Advances,
2014
10.

Kusama H., Arakawa H.
Journal of Photochemistry and Photobiology A: Chemistry,
2003
11.

Verbitskiy E.V., Cheprakova E.M., Subbotina J.O., Schepochkin A.V., Slepukhin P.A., Rusinov G.L., Charushin V.N., Chupakhin O.N., Makarova N.I., Metelitsa A.V., Minkin V.I.
Dyes and Pigments,
2014
12.

Achelle S., Rodríguez-López J., Guen F.R.
ChemistrySelect,
2018
13.
Bushueva A.Y., Shklyaeva E.V., Abashev G.G.
Mendeleev Communications,
2009
14.

Abashev G., Sosnin E., Shklyaeva E., Ustalova T., Osorgina I., Romanova V.
physica status solidi (c),
2012
15.
Komissarova E.A., Sosnin E.A., Shklyaeva E.V., Osorgina I.V., Abashev G.G.
Arkivoc,
2017
16.

Komissarova E.A., Lunegov I.V., Shklyaeva E.V., Abashev G.G.
Chemistry of Heterocyclic Compounds,
2016
17.
10.1016/j.mencom.2019.01.014_sbref0045e
Komissarova
Butlerovskie Soobshcheniya,
2015
18.

Facchetti A., Abbotto A., Beverina L., van der Boom M.E., Dutta P., Evmenenko G., Marks T.J., Pagani G.A.
Chemistry of Materials,
2002
19.

Vlád G., Horváth I.T.
Journal of Organic Chemistry,
2002
20.
10.1016/j.mencom.2019.01.014_bib0060
Hunt
J. Chem. Soc.,
1959
21.
T. Nagasawa, K. Kuroiwa, K. Narita, US Patent 3904612 A, 1972.
22.

Chen D., Zhong C., Dong X., Liu Z., Qin J.
Journal of Materials Chemistry A,
2012
23.
K. Brunner, M.M. De Kok-Van Breemen, B.M.W. Langeveld, N.M.M. Kiggen, J.J. A.M. Bastiaansen, J.W. Hofstraat, H.F. Boerner, H.F.M. Schoo, US Patent 2006/0073357 A1, 2006.
24.

Vanden Eynde J.J., Pascal L., Van Haverbeke Y., Dubois P.
Synthetic Communications,
2001
25.
10.1016/j.mencom.2019.01.014_bib0085
Birks
Photophysics of Aromatic Molecules,
1970
26.

Sabouraud G., Sadki S., Brodie N.
Chemical Society Reviews,
2000
27.

28.

Pommerehne J., Vestweber H., Guss W., Mahrt R.F., Bässler H., Porsch M., Daub J.
Advanced Materials,
1995
29.

Li Y., Xue L., Li H., Li Z., Xu B., Wen S., Tian W.
Macromolecules,
2009
30.

Schmidt M.W., Baldridge K.K., Boatz J.A., Elbert S.T., Gordon M.S., Jensen J.H., Koseki S., Matsunaga N., Nguyen K.A., Su S., Windus T.L., Dupuis M., Montgomery J.A.
Journal of Computational Chemistry,
1993