Abstract
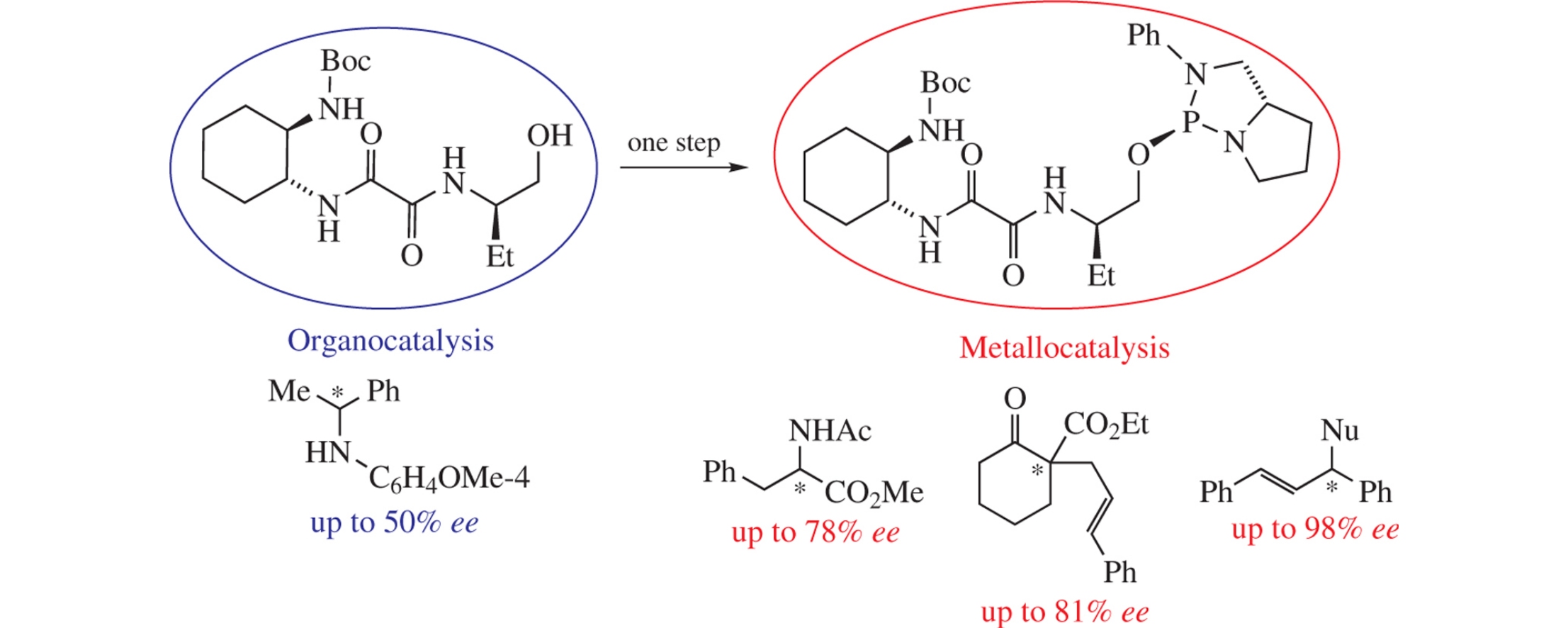
A P*-chiral diamidophosphite ligand of the 1,3,2-diazaphospholidine series was obtained by direct phosphorylation of N-[(1R,2R)-2-(tert-butoxycarbonylaminocyclohexyl)]-N′-[(1R)-1-(hydroxymethyl)propyl]oxalamide. The use of this ligand made it possible to reach 98% ee in the Pd-catalyzed allylation involving (E)-1,3-diphenylallyl acetate, 81% ee in the allylation of ethyl 2-oxocyclohexane-1-carboxylate with cinnamyl acetate, 78% ee in the Rh-catalyzed hydrogenation of methyl (Z)-2-acetamido-3-phenylacrylate, and 32% ee in the Rh- and Ir-catalyzed hydrosilylation of (E)-N-(4-methoxyphenyl)-1-phenylethan-1-imine with diphenylsilane. The above mentioned oxalamide provides up to 50% ee in the organocatalytic reduction of the same imine.
References
1.

Burk M.J.
Accounts of Chemical Research,
2000
2.
10.1016/j.mencom.2019.01.010_sbref0005b
Asymmetric Catalysis on Industrial Scale,
2010
3.
Beletskaya I.P., Kabachnik M.M.
Mendeleev Communications,
2008
4.

Abakumov G.A., Piskunov A.V., Cherkasov V.K., Fedushkin I.L., Ananikov V.P., Eremin D.B., Gordeev E.G., Beletskaya I.P., Averin A.D., Bochkarev M.N., Trifonov A.A., Dzhemilev U.M., D'yakonov V.A., Egorov M.P., Vereshchagin A.N., et. al.
Russian Chemical Reviews,
2018
5.

Gavrilov K.N., Bondarev O.G., Polosukhin A.I.
Russian Chemical Reviews,
2004
6.

Swennenhuis B.H., Chen R., van Leeuwen P.W., de Vries J.G., Kamer P.C.
European Journal of Organic Chemistry,
2009
7.

Teichert J., Feringa B.
Angewandte Chemie - International Edition,
2010
8.

Lühr S., Holz J., Börner A.
ChemCatChem,
2011
9.

Leeuwen P.W., Kamer P.C., Claver C., Pàmies O., Diéguez M.
Chemical Reviews,
2010
10.

Fu W., Tang W.
ACS Catalysis,
2016
11.

Sandee A.J., van der Burg A.M., Reek J.N.
Chemical Communications,
2007
12.

Laungani A.C., Breit B.
Chemical Communications,
2008
13.

Pignataro L., Carboni S., Civera M., Colombo R., Piarulli U., Gennari C.
Angewandte Chemie - International Edition,
2010
14.

Xia H., Yan H., Shen C., Shen F., Zhang P.
Catalysis Communications,
2011
15.

Margalef J., Lega M., Ruffo F., Pàmies O., Diéguez M.
Tetrahedron Asymmetry,
2012
16.

Lega M., Margalef J., Ruffo F., Pàmies O., Diéguez M.
Tetrahedron Asymmetry,
2013
17.
10.1016/j.mencom.2019.01.010_sbref0020a
Buono
2008
18.
![Development of Diamidophosphite Ligands and Their Application to the Palladium-Catalyzed Vinyl-Substituted Trimethylenemethane Asymmetric [3 + 2] Cycloaddition](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Trost B.M., Lam T.M.
Journal of the American Chemical Society,
2012
19.

Bravo M.J., Ceder R.M., Muller G., Rocamora M.
Organometallics,
2013
20.

Bravo M.J., Ceder R.M., Grabulosa A., Muller G., Rocamora M., Font-Bardia M.
Journal of Organometallic Chemistry,
2017
21.
10.1016/j.mencom.2019.01.010_sbref0025a
Privileged Chiral Ligands and Catalysts,
2011
22.

Recent Developments in Catalytic Asymmetric Hydrogenation Employing P-Chirogenic Diphosphine Ligands
Crépy K. ., Imamoto T.
Advanced Synthesis and Catalysis,
2003
23.
10.1016/j.mencom.2019.01.010_sbref0025c
P-Stereogenic Ligands in Enantioselective Catalysis,
2011
24.

Tsarev V., Lyubimov S., Shiryaev A., Zheglov S., Bondarev O., Davankov V., Kabro A., Moiseev S., Kalinin V., Gavrilov K.
European Journal of Organic Chemistry,
2004
25.
![A Practical Method for the Large-Scale Synthesis of Diastereomerically Pure (2R,5S)- 3-Phenyl-2-(8-quinolinoxy)- 1,3-diaza-2-phosphabicyclo-[3.3.0]-octane Ligand (QUIPHOS). Synthesis and X-ray Structure of Its Corresponding Chiral π-Allyl Palladium Complex](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Brunel J.M., Constantieux T., Buono G.
Journal of Organic Chemistry,
1999
26.

Barta K., Hölscher M., Franciò G., Leitner W.
European Journal of Organic Chemistry,
2009
27.
![Development of New P-Chiral Phosphorodiamidite Ligands Having a Pyrrolo[1,2-c]diazaphosphol-1-one Unit and Their Application to Regio- and Enantioselective Iridium-Catalyzed Allylic Etherification](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Kimura M., Uozumi Y.
Journal of Organic Chemistry,
2006
28.

Fernández-Pérez H., Etayo P., Panossian A., Vidal-Ferran A.
Chemical Reviews,
2011
29.

Lu Z., Ma S.
Angewandte Chemie - International Edition,
2008
30.

Diéguez M., Pàmies O.
Accounts of Chemical Research,
2009
31.

Lafrance D., Bowles P., Leeman K., Rafka R.
Organic Letters,
2011
32.

Trost B.M.
Organic Process Research and Development,
2012
33.

Nag S., Batra S.
Tetrahedron,
2011
34.

Chavan S.P., Khairnar L.B., Chavan P.N.
Tetrahedron Letters,
2014
35.

Liu Y., Han S., Liu W., Stoltz B.M.
Accounts of Chemical Research,
2015
36.
![Synthesis of gem-Difluoromethylenated Bicyclo[m.n.0]alkan-1-ols and Their Ring-Expansion to gem-Difluoromethylenated Macrocyclic Lactones](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Punirun T., Peewasan K., Kuhakarn C., Soorukram D., Tuchinda P., Reutrakul V., Kongsaeree P., Prabpai S., Pohmakotr M.
Organic Letters,
2012
37.
10.1016/j.mencom.2019.01.010_sbref0035j
Yen
Applications of Transition Metal Catalysis in Drug Discovery and Development: an Industrial Perspective,
2012
38.

Etayo P., Vidal-Ferran A.
Chemical Society Reviews,
2013
39.

Li B., Sortais J., Darcel C.
RSC Advances,
2016
40.
![(S)-2-[(N-arylamino)methyl]pyrrolidines-Based Phosphoramidite P,N-Ligand Library for Asymmetric Metal-Catalyzed Allylic Substitution and Conjugate 1,4-Addition](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Gavrilov K.N., Mikhel I.S., Chuchelkin I.V., Zheglov S.V., Gavrilov V.K., Birin K.P., Tafeenko V.A., Chernyshev V.V., Goulioukina N.S., Beletskaya I.P.
ChemistrySelect,
2016
41.

Goulioukina N., Bondarenko G., Lyubimov S., Davankov V., Gavrilov K., Beletskaya I.
Advanced Synthesis and Catalysis,
2008
42.

Brzezińska-Rodak M., Klimek-Ochab M., Żymańczyk-Duda E., Kafarski P.
Molecules,
2011
43.

44.

Kowalczyk D., Albrecht Ł.
Chemical Communications,
2015
45.

Yan Z., Wu B., Gao X., Chen M., Zhou Y.
Organic Letters,
2016
46.

Nemoto T., Matsumoto T., Masuda T., Hitomi T., Hatano K., Hamada Y.
Journal of the American Chemical Society,
2004
47.

Nemoto T., Masuda T., Matsumoto T., Hamada Y.
Journal of Organic Chemistry,
2005
48.
Kochetkov S.V., Kucherenko A.S., Zlotin S.G.
Mendeleev Communications,
2017
49.
Fedorova O.V., Titova Y.A., Ovchinnikova I.G., Rusinov G.L., Charushin V.N.
Mendeleev Communications,
2018