Abstract
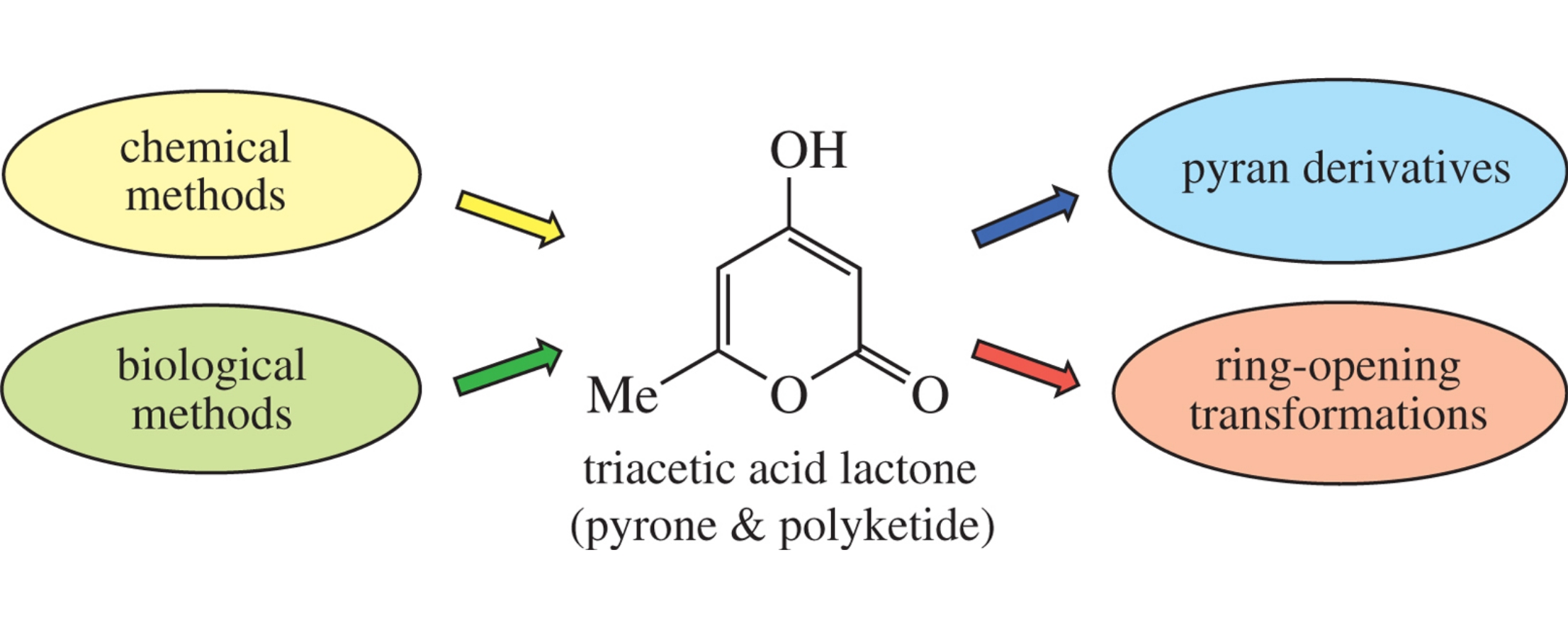
Major methods for the preparation of triacetic acid lactone and its application as a bioprivileged compound in the synthesis of various valuable materials are summarized. Due to its structural features, this lactone belongs to both pyrones and polyketides, which provides opportunities for its obtaining by chemical and biological methods. The presence of several electrophilic and nucleophilic centers in its molecule, as well as its capability of undergoing transformations with both preservation and opening of the ring, ensure its multiple reactivity. Reactions proceeding without the ring opening lead to substituted and fused pyrans, while the ring opening provides N-heterocycles and acyclic derivatives.
References
1.

Luterbacher J.S., Martin Alonso D., Dumesic J.A.
Green Chemistry,
2014
2.

Bardhan S.K., Gupta S., Gorman M.E., Haider M.A.
Renewable and Sustainable Energy Reviews,
2015
3.

Zhu H., Luo W., Ciesielski P.N., Fang Z., Zhu J.Y., Henriksson G., Himmel M.E., Hu L.
Chemical Reviews,
2016
4.

Chernyshev V.M., Kravchenko O.A., Ananikov V.P.
Russian Chemical Reviews,
2017
5.
Chernysheva D.V., Klushin V.A., Zubenko A.F., Pudova L.S., Kravchenko O.A., Chernyshev V.M., Smirnova N.V.
Mendeleev Communications,
2018
6.

Schäberle T.F.
Beilstein Journal of Organic Chemistry,
2016
7.

McGlacken G.P., Fairlamb I.J.
Natural Product Reports,
2005
8.
ZIRAK M., EFTEKHARI-SIS B.
Turkish Journal of Chemistry,
2015
9.

10.

Weeks P.D., Brennan T.M., Brannegan D.P., Kuhla D.E., Elliott M.L., Watson H.A., Wlodecki B., Breitenbach R.
Journal of Organic Chemistry,
1980
11.

Moreno-Mañas M., Pleixats R.
Advances in Heterocyclic Chemistry,
1992
12.

Shanks B.H., Keeling P.L.
Green Chemistry,
2017
13.

Schwartz T.J., Shanks B.H., Dumesic J.A.
Current Opinion in Biotechnology,
2016
14.

Mohammadi Ziarani G., Lashgari N., Moradi R., Zandiyeh M.
Heterocycles,
2018
15.

Kumagai M., Nishikawa K., Mishima T., Yoshida I., Ide M., Koizumi K., Nakamura M., Morimoto Y.
Bioorganic and Medicinal Chemistry Letters,
2017
16.

Fang S., Chen L., Yu M., Cheng B., Lin Y., Morris-Natschke S.L., Lee K., Gu Q., Xu J.
Organic and Biomolecular Chemistry,
2015
17.

Cheng B., Lin Y., Kuang M., Fang S., Gu Q., Xu J., Wang L.
Chemical Biology and Drug Design,
2015
18.
10.1016/j.mencom.2019.01.001_bib0040
Fadel
ARKIVOC,
2011
19.
10.1016/j.mencom.2019.01.001_sbref0045a
Collie
J. Chem. Soc.,
1891
20.

21.

Butt M.A., Elvidge J.A.
Journal of the Chemical Society (Resumed),
1963
22.

Dong Y., Nakagawa-Goto K., Lai C., Morris-Natschke S.L., Bastow K.F., Lee K.
Bioorganic and Medicinal Chemistry Letters,
2011
23.

Häusler J.
Monatshefte fur Chemie,
1982
24.

Cardenas J., Da Silva N.A.
Metabolic Engineering,
2014
25.

Saunders L.P., Bowman M.J., Mertens J.A., Da Silva N.A., Hector R.E.
Journal of Industrial Microbiology and Biotechnology,
2015
26.

Cardenas J., Da Silva N.A.
Metabolic Engineering,
2016
27.

Tang S., Qian S., Akinterinwa O., Frei C.S., Gredell J.A., Cirino P.C.
Journal of the American Chemical Society,
2013
28.

Xie D., Shao Z., Achkar J., Zha W., Frost J.W., Zhao H.
Biotechnology and Bioengineering,
2006
29.

Li Y., Qian S., Dunn R., Cirino P.C.
Journal of Industrial Microbiology and Biotechnology,
2018
30.

Markham K.A., Palmer C.M., Chwatko M., Wagner J.M., Murray C., Vazquez S., Swaminathan A., Chakravarty I., Lynd N.A., Alper H.S.
Proceedings of the National Academy of Sciences of the United States of America,
2018
31.

Yu J., Landberg J., Shavarebi F., Bilanchone V., Okerlund A., Wanninayake U., Zhao L., Kraus G., Sandmeyer S.
Biotechnology and Bioengineering,
2018
32.

Koskela S., Söderholm P.P., Ainasoja M., Wennberg T., Klika K.D., Ovcharenko V.V., Kylänlahti I., Auerma T., Yli-Kauhaluoma J., Pihlaja K., Vuorela P.M., Teeri T.H.
Planta,
2010
33.

Wang Y., Bao R., Huang S., Tang Y.
Beilstein Journal of Organic Chemistry,
2013
34.

Song L., Yao H., Zhu L., Tong R.
Organic Letters,
2012
35.

Burns M.J., Ronson T.O., Taylor R.J., Fairlamb I.J.
Beilstein Journal of Organic Chemistry,
2014
36.
10.1016/j.mencom.2019.01.001_sbref0065b
Zhang
Synthesis,
2007
37.

Kirsch S.F., Bach T.
Chemistry - A European Journal,
2005
38.

Xi M., Sun Z., Sun H., Jia J., Jiang Z., Tao L., Ye M., Yang X., Wang Y., Xue X., Huang J., Gao Y., Guo X., Zhang S., Yang Y., et. al.
European Journal of Medicinal Chemistry,
2013
39.

Mori T., Ujihara K., Matsumoto O., Yanagi K., Matsuo N.
Journal of Fluorine Chemistry,
2007
40.

Fairlamb I.J., Lee A.F., Loe-Mie F.E., Niemelä E.H., O'Brien C.T., Whitwood A.C.
Tetrahedron,
2005
41.

Fairlamb I.J., Marrison L.R., Dickinson J.M., Lu F., Schmidt J.P.
Bioorganic and Medicinal Chemistry,
2004
42.

Lei X., Gao L., Ding Q., Peng Y., Wu J.
Organic and Biomolecular Chemistry,
2011
43.

Lah H.U., Rasool F., Yousuf S.K.
RSC Advances,
2015
44.

Hu Y., Ding Q., Ye S., Peng Y., Wu J.
Tetrahedron,
2011
45.

Marrison L.R., Dickinson J.M., Fairlamb I.J.
Bioorganic and Medicinal Chemistry Letters,
2002
46.

Bellina F., Marchetti C., Rossi R.
European Journal of Organic Chemistry,
2009
47.

Kuroda J., Inamoto K., Hiroya K., Doi T.
European Journal of Organic Chemistry,
2009
48.

Hansen A.L., Skrydstrup T.
Organic Letters,
2005
49.

Prendergast A.M., McGlacken G.P.
European Journal of Organic Chemistry,
2017
50.

Nolan M., Bray J.T., Eccles K., Cheung M.S., Lin Z., Lawrence S.E., Whitwood A.C., Fairlamb I.J., McGlacken G.P.
Tetrahedron,
2014
51.

Pardo L.M., Prendergast A.M., Nolan M., Ó Muimhneacháin E., McGlacken G.P.
European Journal of Organic Chemistry,
2015
52.

Nolan M., Pardo L.M., Prendergast A.M., McGlacken G.P.
Journal of Organic Chemistry,
2015
53.

Burns M.J., Thatcher R.J., Taylor R.J., Fairlamb I.J.
Dalton Transactions,
2010
54.

Mackey K., Pardo L.M., Prendergast A.M., Nolan M., Bateman L.M., McGlacken G.P.
Organic Letters,
2016
55.

de March P., Moreno-Mañas M., Casado J., Pleixats R., Roca J.L., Trius A.
Journal of Heterocyclic Chemistry,
1984
56.
![A One-Pot Condensation of Pyrones and Enals. Synthesis of 1H,7H-5a,6,8,9-Tetrahydro-1-oxopyrano[4,3-b][1]benzopyrans](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Hua D.H., Chen Y., Sin H., Maroto M.J., Robinson P.D., Newell S.W., Perchellet E.M., Ladesich J.B., Freeman J.A., Perchellet J., Chiang P.K.
Journal of Organic Chemistry,
1997
57.
![Sequential 1,2-Addition−Electrocyclic Ring Closures Involving Acyclic α,β-Unsaturated Iminiums: A Formal [3 + 3] Cycloaddition Strategy to Unique Pyranyl Spirocycles](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Hsung R.P., Shen H.C., Douglas C.J., Morgan C.D., Degen S.J., Yao L.J.
Journal of Organic Chemistry,
1999
58.
![A Formal [3 + 3] Cycloaddition Reaction. Improved Reactivity Using α,β-Unsaturated Iminium Salts and Evidence for Reversibility of 6π-Electron Electrocyclic Ring Closure of 1-Oxatrienes](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Shen H.C., Wang J., Cole K.P., McLaughlin M.J., Morgan C.D., Douglas C.J., Hsung R.P., Coverdale H.A., Gerasyuto A.I., Hahn J.M., Liu J., Sklenicka H.M., Wei L., Zehnder L.R., Zificsak C.A., et. al.
Journal of Organic Chemistry,
2003
59.
![Brønsted Acid‐Catalyzed Synthesis of Pyrans via a Formal [3+3] Cycloaddition](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Hubert C., Moreau J., Batany J., Duboc A., Hurvois J., Renaud J.
Advanced Synthesis and Catalysis,
2008
60.
![Metal-Free Brønsted Acid Catalyzed Formal [3 + 3] Annulation. Straightforward Synthesis of Dihydro-2H-Chromenones, Pyranones, and Tetrahydroquinolinones](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Moreau J., Hubert C., Batany J., Toupet L., Roisnel T., Hurvois J., Renaud J.
Journal of Organic Chemistry,
2009
61.
![A Lewis Acid-Catalyzed Formal [3 + 3] Cycloaddition of α,β-Unsaturated Aldehydes with 4-Hydroxy-2-Pyrone, Diketones, and Vinylogous Esters](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Kurdyumov A.V., Lin N., Hsung R.P., Gullickson G.C., Cole K.P., Sydorenko N., Swidorski J.J.
Organic Letters,
2005
62.
![Efficient synthesis of substituted 7-methyl-2H,5H-pyrano[4,3-b]pyran-5-ones and evaluation of their in vitro antiproliferative/cytotoxic activities](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Leutbecher H., Williams L.A., Rösner H., Beifuss U.
Bioorganic and Medicinal Chemistry Letters,
2007
63.
![Diastereoselective Synthesis of Polycyclic Acetal-Fused Pyrano[3,2-c]pyran-5(2H)-one Derivatives](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Sagar R., Park J., Koh M., Park S.B.
Journal of Organic Chemistry,
2009
64.
![Regioselective synthesis of pyrano[3,2-c]coumarins via Cu(II)-catalyzed tandem reaction](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Bagdi A.K., Majee A., Hajra A.
Tetrahedron Letters,
2013
65.

Yang S., Shen L., Kim Y., Jeong J.
Organic and Biomolecular Chemistry,
2016
66.

Liu Y., Zhu J., Qian J., Jiang B., Xu Z.
Journal of Organic Chemistry,
2011
67.

Kausar N., Ghosh P.P., Pal G., Das A.R.
RSC Advances,
2015
68.

Rueping M., Merino E., Sugiono E.
Advanced Synthesis and Catalysis,
2008
69.

Yao H., Song L., Liu Y., Tong R.
Journal of Organic Chemistry,
2014
70.

Leutbecher H., Hajdok S., Braunberger C., Neumann M., Mika S., Conrad J., Beifuss U.
Green Chemistry,
2009
71.

Leutbecher H., Greiner G., Amann R., Stolz A., Beifuss U., Conrad J.
Organic and Biomolecular Chemistry,
2011
72.

Miao C., Liu R., Sun Y., Sun X., Yang H.
Tetrahedron Letters,
2017
73.

Asta C., Schmidt D., Conrad J., Frey W., Beifuss U.
Organic and Biomolecular Chemistry,
2013
74.

Zhao B., Du D.
Tetrahedron Asymmetry,
2014
75.
Anary-Abbasinejad M., Anaraki-Ardakani H., Mosslemin M.H., Khavasi H.R.
Journal of the Brazilian Chemical Society,
2010
76.

Ni C., Zhou W., Tong X.
Tetrahedron,
2017
77.

Moghaddam F.M., Mirjafary Z., Saeidian H., Taheri S., Soltanzadeh B.
Tetrahedron,
2010
78.

Ghosh M., Hajra A.
European Journal of Organic Chemistry,
2015
79.

Rueping M., Parra A., Uria U., Besselièvre F., Merino E.
Organic Letters,
2010
80.

Gurubrahamam R., Gao B., Chen Y.M., Chan Y., Tsai M., Chen K.
Organic Letters,
2016
81.

Chia M., Schwartz T.J., Shanks B.H., Dumesic J.A.
Green Chemistry,
2012
82.

Schwartz T.J., Johnson R.L., Cardenas J., Okerlund A., Da Silva N.A., Schmidt-Rohr K., Dumesic J.A.
Angewandte Chemie - International Edition,
2014
83.

Hansen C.A., Frost J.W.
Journal of the American Chemical Society,
2002
84.

Zha W., Shao Z., Frost J.W., Zhao H.
Journal of the American Chemical Society,
2004
85.

Sun C., Fürstner A.
Angewandte Chemie - International Edition,
2013
86.

Gärtner D., Stein A.L., Grupe S., Arp J., Jacobi von Wangelin A.
Angewandte Chemie - International Edition,
2015
87.

Stoyanov E., Ivanov I.
Molecules,
2004
88.

Vincetti P., Caporuscio F., Kaptein S., Gioiello A., Mancino V., Suzuki Y., Yamamoto N., Crespan E., Lossani A., Maga G., Rastelli G., Castagnolo D., Neyts J., Leyssen P., Costantino G., et. al.
Journal of Medicinal Chemistry,
2015
89.
10.1016/j.mencom.2019.01.001_sbref0245c
Castillo
Bull. Soc. Chim. Fr.,
1982
90.

Castillo S., Bouissou T., Favrot J., Brazier J.F., Fayet J.P.
Spectrochimica Acta Part A Molecular Spectroscopy,
1993
91.

Vucicevic J., Srdic-Rajic T., Pieroni M., Laurila J.M., Perovic V., Tassini S., Azzali E., Costantino G., Glisic S., Agbaba D., Scheinin M., Nikolic K., Radi M., Veljkovic N.
Bioorganic and Medicinal Chemistry,
2016
92.

Kraus G.A., Wanninayake U.K., Bottoms J.
Tetrahedron Letters,
2016
93.

Cheruvallath Z.S., Gwaltney S.L., Sabat M., Tang M., Feng J., Wang H., Miura J., Guntupalli P., Jennings A., Hosfield D., Lee B., Wu Y.
Bioorganic and Medicinal Chemistry Letters,
2013
94.

Bio-based triacetic acid lactone in the synthesis of azaheterocyclesviaa ring-opening transformation
Obydennov D.L., El-Tantawy A.I., Sosnovskikh V.Y.
New Journal of Chemistry,
2018
95.

Obydennov D.L., El-Tantawy A.I., Sosnovskikh V.Y.
Journal of Organic Chemistry,
2018
96.

Faidallah H.M., Rostom S.A., Khan K.A., Basaif S.A.
Journal of Enzyme Inhibition and Medicinal Chemistry,
2012
97.

Ainsworth C., Jones R.G.
Journal of the American Chemical Society,
1954
98.
B. Buettelmann, H. Knust, A. Thomas, US Patent, 20070082890, 2007.
99.

Jouha J., Loubidi M., Bouali J., Hamri S., Hafid A., Suzenet F., Guillaumet G., Dagcı T., Khouili M., Aydın F., Saso L., Armagan G.
European Journal of Medicinal Chemistry,
2017
100.
![A one-pot process for the microwave-assisted synthesis of 7-substituted pyrazolo[1,5-a]pyrimidine](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Bassoude I., Tber Z., Essassi E.M., Guillaumet G., Berteina-Raboin S.
RSC Advances,
2016
101.
![One-step reaction leading to new pyrazolo[1,5-a]pyrimidines by condensation of 2-pyrone with 5(3)-amino-3(5)-arylpyrazoles](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Bassoude I., Berteina-Raboin S., Leger J., Jarry C., Essassi E.M., Guillaumet G.
Tetrahedron,
2011
102.
![Synthesis of new compounds containing the pyrazolo[3,4-b]pyridine-3-one subunit](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Fadel S., Suzenet F., Hafid A., Rakib E.M., Khouili M., Pujol M.D., Guillaumet G.
Journal of Heterocyclic Chemistry,
2009
103.
![Unexpected Ring-Opening of a 2-Pyrone Ring in the Synthesis of 3-[(Z)-1-Hydroxy-3-Oxobut-1-Enyl]-2H-chromen-2-One Derivatives Catalysed by Kf-Alumina](/storage/images/resized/ruydfaB80LDjlkYqsfOeUAZohOIODyq7bQzis5O7_small_thumb.webp)
Wang X., Zeng Z., Zhang M., Shi D., Tu S.
Journal of Chemical Research,
2006
104.
![A novel and direct synthetic route to substituted 1,5-dihydro-4H-[1]benzopyrano[4,3-b]pyridine-4,5-diones](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Světlík J., Prónayová N., Hanuš V.
Journal of Heterocyclic Chemistry,
2000
105.

Ben Mohamed S., Rachedi Y., Hamdi M., Le Bideau F., Dejean C., Dumas F.
European Journal of Organic Chemistry,
2016
106.

Chen Z., Chen F., Sun Y., Liu H., He H., Zhang X., Wang S.
RSC Advances,
2017
107.

Shaikh T.S., Undale K.A., Gaikwad D.S., Pore D.M.
Comptes Rendus Chimie,
2011
108.
![Synthesis of novel 2H,8H-pyrano[2,3-f]chromene-2,8-diones from 8-formyl-7-hydroxy-4-methylcoumarin](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Al-Kawkabani A., Boutemeur-Kheddis B., Makhloufi-Chebli M., Hamdi M., Talhi O., Silva A.M.
Tetrahedron Letters,
2013
109.

Siddiqui Z.N., Khan K.
New Journal of Chemistry,
2013
110.

Siddiqui Z., Praveen S., Farooq F.
Chemical Papers,
2010
111.

Li X., Liu Y., Liu X., Zhang Z.
RSC Advances,
2015
112.

Velpula R., Deshineni R., Gali R., Bavantula R.
Research on Chemical Intermediates,
2015
113.

Tangeti V.S., Varma K. R., Siva Prasad G.V., Satyanarayana K.V.
Synthetic Communications,
2016
114.
![Synthesis, Characterization and Cytotoxic Investigations of Novel C3-Dihydrofuran Substituted 1H-benzo[g]chromene-2,5,10-triones besides Antimicrobial study](/storage/images/resized/Vb3hw5ha3GXFySkcFIRq7hedUzMGRVNprYvOwnlQ_small_thumb.webp)
Tangeti V.S., Vasundhara D., Kumar M.N., Mylapalli H., Pavan Kumar K.S.
Asian Journal of Chemistry,
2017
115.
![A Novel and Efficient Synthesis of 3-[(4,5-Dihydro-1H-pyrrol-3-yl)carbonyl]-2H-chromen-2-ones (=3-[(4,5-Dihydro-1H-pyrrol-3-yl)carbonyl]-2H-1-benzopyran-2-ones)](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Alizadeh A., Ghanbaripour R.
Helvetica Chimica Acta,
2013
116.
![Synthesis of 3-[(coumarinyl)carbonyl]-3a,8b-dihyroindeno[1,2-b]pyrrole-4(1H)-ones and their conversion to coumarin bearing spiro[isobenzofuran-1,2′-pyrrole] moiety compounds via oxidative cleavage reaction](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Alizadeh A., Ghanbaripour R., Feizabadi M., Zhu L., Dusek M.
RSC Advances,
2015
117.

Nagawade R.R., Khanna V.V., Bhagwat S.S., Shinde D.B.
European Journal of Medicinal Chemistry,
2005
118.

Filipponi P., Baxendale I.R.
European Journal of Organic Chemistry,
2016
119.

Mojahidi S., Rakib E.M., Sekkak H., Abouricha S., Benchat N., Mousse H.A., Zyad A.
Archiv der Pharmazie,
2010
120.
![Synthesis of New Azocompounds and Fused Pyrazolo[5,1-c][1,2,4]triazines Using Heterocyclic Components](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Ledenyova I.V., Didenko V.V., Shestakov A.S., Shikhaliev K.S.
Journal of Heterocyclic Chemistry,
2013
121.

Blackaby W.P., Atack J.R., Bromidge F., Lewis R., Russell M.G., Smith A., Wafford K., McKernan R.M., Street L.J., Castro J.L.
Bioorganic and Medicinal Chemistry Letters,
2005
122.
![[4-(Phenoxy)pyridin-3-yl]methylamines: A new class of selective noradrenaline reuptake inhibitors](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Fish P.V., Ryckmans T., Stobie A., Wakenhut F.
Bioorganic and Medicinal Chemistry Letters,
2008
123.

El Bakali J., Muccioli G.G., Renault N., Pradal D., Body-Malapel M., Djouina M., Hamtiaux L., Andrzejak V., Desreumaux P., Chavatte P., Lambert D.M., Millet R.
Journal of Medicinal Chemistry,
2010
124.
![SYNTHESIS OF 3-ACETOACETYL-7-METHYL-2H,5H-PYRANO[4,3-B]PYRAN-2,5-DIONE](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Boutemeur-Kheddis B., Bendaas A., Hamdi M., Sakellariou R., Spéziale V.
Organic Preparations and Procedures International,
1994
125.
![Study of the reactions of 3-acetoacetyl-7-methyl-2H,5H-pyrano[4,3-b]pyran-2,5-dione with aromatic amines](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Boutemeur-Kheddis B., Hamdi M., Sellier N., Silva A.M.
Journal of Heterocyclic Chemistry,
2001
126.

Preindl J., Schulthoff S., Wirtz C., Lingnau J., Fürstner A.
Angewandte Chemie - International Edition,
2017
127.

Qi C., Qin T., Suzuki D., Porco J.A.
Journal of the American Chemical Society,
2014
128.

Bhat Z.S., Rather M.A., Maqbool M., Lah H.U., Yousuf S.K., Ahmad Z.
Biomedicine and Pharmacotherapy,
2017
129.
![Cyclooctatrienes from pyran-2-ones via a tandem [4 + 4]-photocycloaddition/decarboxylation process](/storage/images/resized/leiAYcRDGTSl5B1eCnwpSGqmDEUEfDPPoYisFGhT_small_thumb.webp)
Li L., Chase C.E., West F.G.
Chemical Communications,
2008
130.
![Diastereocontrol in [4+4]-photocycloadditions of pyran-2-ones: effect of ring substituents and chiral ketal](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Li L., Bender J.A., West F.G.
Tetrahedron Letters,
2009
131.

Ghandi M., Feizi S., Nazeri M.T., Notash B.
Journal of the Iranian Chemical Society,
2016