Abstract
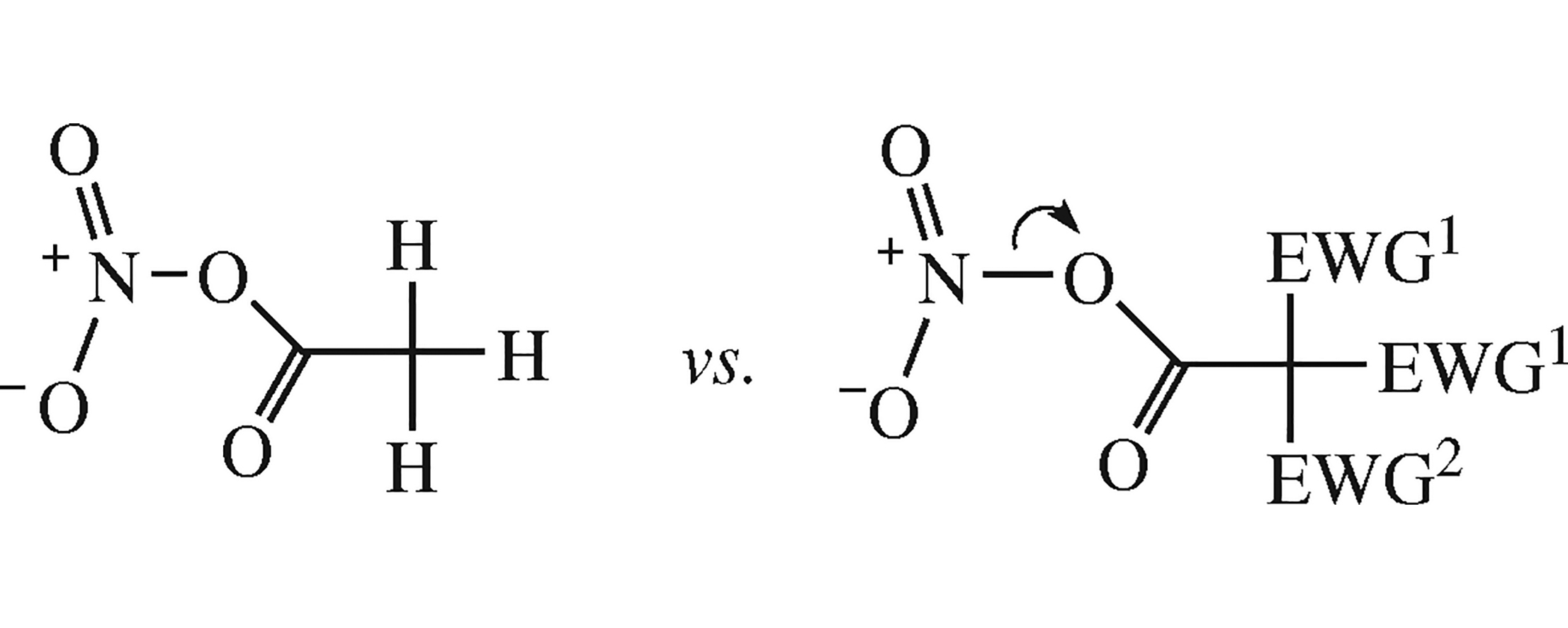
Regularities in the Raman (\( \mathrm{v_{NO_2}^s} \)) and 14NMR spectra of acetyl nitrates containing various electron-withdrawing α-substituents were established by quantum chemical calculations. The experimental data on (poly)-α-haloacetyl nitrates (in solutions) revealed no reliable relationship between the halogen nature/number and the spectral characteristics, whereas the computed values of δN and O-NO2 bond lengths disclose the substituent effect on the polarization of O-N bond and hence on the nitrating power of acyl nitrates.
References
1.
10.1016/j.mencom.2018.11.026_bibe0010
Olah
Superacid Chemistry,
2009
2.

Krasnikov P.E., Osyanin V.A., Osipov D.V., Klimochkin Y.N.
Russian Journal of General Chemistry,
2016
3.

Olah G.A., Lin H.C., Olah J.A., Narang S.C.
Proceedings of the National Academy of Sciences of the United States of America,
1978
4.

Bachman G.B., Dever J.L.
Journal of the American Chemical Society,
1958
5.

Evans J.C., Rinn H.W., Kuhn S.J., Olah G.A.
Inorganic Chemistry,
1964
6.
10.1016/j.mencom.2018.11.026_bibe0035
Kranke
Isotopenpraxis,
1984
7.

Sprague R.W., Garrett A.B., Sisler H.H.
Journal of the American Chemical Society,
1960
8.
10.1016/j.mencom.2018.11.026_bibe0045
Zyk
Zh. Org. Khim.,
1995
9.

Zelenov V.P., Bukalov S.S., Leites L.A., Bushmarinov I.S., Struchkova M.I., Dmitrienko A.O., Tartakovsky V.A.
ChemistrySelect,
2017
10.
Zelenov V.P., Bukalov S.S., Leites L.A., Aysin R.R., Subbotin A.N., Struchkova M.I., Fedyanin I.V.
Mendeleev Communications,
2017
11.
Zelenov V.P., Bukalov S.S., Subbotin A.N.
Mendeleev Communications,
2017
12.
V. P. Zelenov, A.N. Subbotin and I. A. Troyan, Fluorine Notes, 2017, http://notes.fluorine1.ru/public/2017/6_2017/article_4.html.
13.
10.1016/j.mencom.2018.11.026_bibe0070
Hoggett
Nitration and Aromatic Reactivity,
1971
14.
10.1016/j.mencom.2018.11.026_bibe0075
Odokienko
Zh. Prikl. Khim.,
1978
15.
10.1016/j.mencom.2018.11.026_bibe0080
Frisch
Gaussian 09,
2009
16.

Harrar J.E., Rigdon L.P., Rice S.F.
Journal of Raman Spectroscopy,
1997
17.

Witanowski M., Biedrzycka Z., Grela K., Wejroch K.
Magnetic Resonance in Chemistry,
1998
18.
Epishina M.A., Ovchinnikov I.V., Kulikov A.S., Makhova N.N., Tartakovsky V.A.
Mendeleev Communications,
2011
19.

Zapol'skii V.A., Namyslo J.C., Sergeev G., Brönstrup M., Gjikaj M., Kaufmann D.E.
European Journal of Organic Chemistry,
2015
20.

Witanowski M., Stefaniak L.
Journal of the Chemical Society B Physical Organic,
1967
21.

Harriss M.G., Milne J.B.
Canadian Journal of Chemistry,
1971
22.
10.1016/j.mencom.2018.11.026_bibe0115
Tsvetkov
J. Gen. Chem. USSR,
1989
23.
10.1016/j.mencom.2018.11.026_bibe0120
Shirokova
Russ. J. Inorg. Chem.,
1971
24.

Ryason R., Wilson M.K.
Journal of Chemical Physics,
1954
25.

Trimithioti M., Akimov A.V., Prezhdo O.V., Hayes S.C.
Journal of Chemical Physics,
2014
26.

Durig J.R., Kim Y.H., Guirgis G.A., McDonald J.K.
Spectrochimica Acta Part A Molecular Spectroscopy,
1994