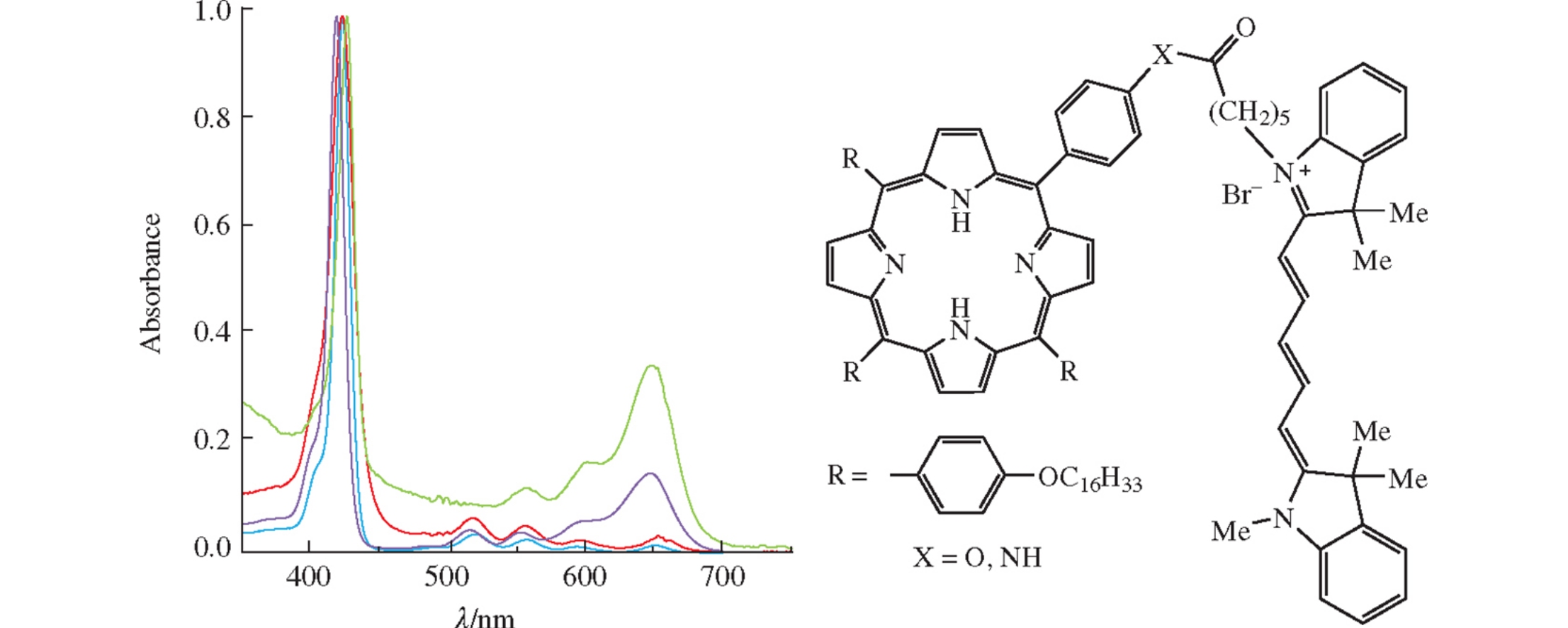
Abstract
New amphiphilic conjugates of porphyrins and cyanine dye Cy5COOH were prepared by the creation of ester or amide bond between Cy5-carboxylic acid and hydroxy- or amino-substituted porphyrins. Study of self-aggregation of these conjugates in aqueous-organic media by dynamic light scattering and UV-VIS spectroscopy showed that the conjugates form micelles in THF–H2O with different ratios.
References
1.

Oniszczuk A., Wojtunik-Kulesza K.A., Oniszczuk T., Kasprzak K.
Biomedicine and Pharmacotherapy,
2016
2.

Celli J.P., Spring B.Q., Rizvi I., Evans C.L., Samkoe K.S., Verma S., Pogue B.W., Hasan T.
Chemical Reviews,
2010
3.

Lucky S.S., Soo K.C., Zhang Y.
Chemical Reviews,
2015
4.

Moret F., Reddi E.
Journal of Porphyrins and Phthalocyanines,
2017
5.

Suvorov N.V., Mironov A.F., Grin M.A.
Russian Chemical Bulletin,
2017
6.
Gushchina O.I., Gramma V.A., Larkina E.A., Mironov A.F.
Mendeleev Communications,
2017
7.

Cheng L., Wang C., Feng L., Yang K., Liu Z.
Chemical Reviews,
2014
8.

Lin L., Xiong L., Wen Y., Lei S., Deng X., Liu Z., Chen W., Miao X.
Journal of Biomedical Nanotechnology,
2014
9.

Panchenko P.A., Grin M.A., Fedorova O.A., Zakharko M.A., Pritmov D.A., Mironov A.F., Arkhipova A.N., Fedorov Y.V., Jonusauskas G., Yakubovskaya R.I., Morozova N.B., Ignatova A.A., Feofanov A.V.
Physical Chemistry Chemical Physics,
2017
10.

Bhattarai P., Liang X., Xu Y., Dai Z.
Journal of Biomedical Nanotechnology,
2017
11.

Liu X., Yang G., Zhang L., Liu Z., Cheng Z., Zhu X.
Nanoscale,
2016
12.

Chen Y., Gryshuk A., Achilefu S., Ohulchansky T., Potter W., Zhong T., Morgan J., Chance B., Prasad P.N., Henderson B.W., Oseroff A., Pandey R.K.
Bioconjugate Chemistry,
2005
13.
James N.S., Ohulchanskyy T.Y., Chen Y., Joshi P., Zheng X., Goswami L.N., Pandey R.K.
Theranostics,
2013
14.

James N.S., Joshi P., Ohulchanskyy T.Y., Chen Y., Tabaczynski W., Durrani F., Shibata M., Pandey R.K.
European Journal of Medicinal Chemistry,
2016
15.

Samaroo D., Perez E., Aggarwal A., Wills A., O'Connor N.
Therapeutic Delivery,
2014
16.

Ahmed S., Davoust E., Savoie H., Boa A.N., Boyle R.W.
Tetrahedron Letters,
2004
17.
Lebedeva N.S., Yurina E.S., Gubarev Y.A., Koifman O.I.
Mendeleev Communications,
2017
18.

Babu S.S., Bonifazi D.
ChemPlusChem,
2014
19.

Bragina N.A., Zhdanova K.A., Mironov A.F.
Russian Chemical Reviews,
2016
20.

Kvach M.V., Ustinov A.V., Stepanova I.A., Malakhov A.D., Skorobogatyi M.V., Shmanai V.V., Korshun V.A.
European Journal of Organic Chemistry,
2008
21.

Johansson M.K., Fidder H., Dick D., Cook R.M.
Journal of the American Chemical Society,
2002
22.

Niu H., Jiang X., He J., Cheng J.
Tetrahedron Letters,
2009
23.
Fomina M.V., Nikiforov A.S., Vedernikov A.I., Kurchavov N.A., Gromov S.P.
Mendeleev Communications,
2014
24.

Zhdanova K.A., Cherepanova K.S., Bragina N.A., Mironov A.F.
Macroheterocycles,
2016
25.

Zhdanova K.A., Zhdanov A.P., Ezhov A.V., Bragina N.A., Zhizhin K.Y., Ushakova I.P., Mironov A.F., Kuznetsov N.T.
Russian Chemical Bulletin,
2014
26.
Zhdanova K.A., Bragina N.A., Bagratashvili V.N., Timashev P.S., Mironov A.F.
Mendeleev Communications,
2014
27.

Fedulova I.N., Bragina N.A., Novikov N.V., Ugol’nikova O.A., Mironov A.F.
Russian Journal of Bioorganic Chemistry,
2007
28.

Hirata T., Kogiso H., Morimoto K., Miyamoto S., Taue H., Sano S., Muguruma N., Ito S., Nagao Y.
Bioorganic and Medicinal Chemistry,
1998
29.

Jung M.E., Kim W.
Bioorganic and Medicinal Chemistry,
2006
30.

Hsieh M., Chien C., Chang C., Chang T.
Journal of Materials Chemistry B,
2013