Abstract
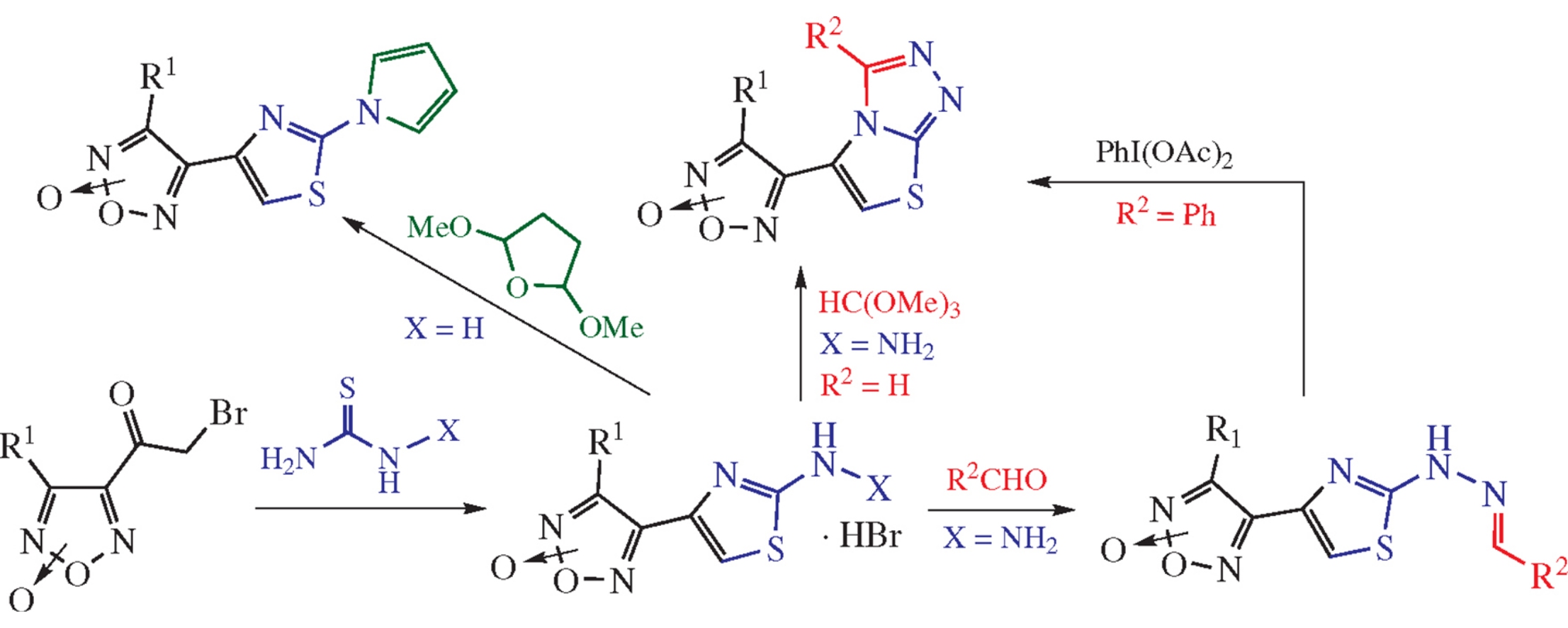
The effective and regioselective synthesis of new (2-hydrazinylthiazol-4-yl)furoxan hydrobromides based on the condensation of (bromoacetyl)furoxans with thiosemicarbazide has been developed. The cytotoxic activity of their derivatives (with hydrazone, 4-thiazolo[2.3-c][1,2,4]triazole or pyrrole moieties) against two human cancer cell lines (A549, HCT116) was tested and several structures revealed moderate cytotoxic activity.
References
1.
10.1016/j.mencom.2018.11.020_sbrefe0010a
Gasco
Nitric Oxide Donors: For Pharmaceutical and Biological Applications,
2005
2.

Guglielmo S., Cortese D., Vottero F., Rolando B., Kommer V.P., Williams D.L., Fruttero R., Gasco A.
European Journal of Medicinal Chemistry,
2014
3.

Zhao N., Tian K., Cheng K., Han T., Hu X., Li D., Li Z., Hua H.
Bioorganic and Medicinal Chemistry,
2016
4.

Fang Y., Wang R., He M., Huang H., Wang Q., Yang Z., Li Y., Yang S., Jin Y.
Bioorganic and Medicinal Chemistry Letters,
2017
5.

Makhova N.N., Rakitin O.A.
Chemistry of Heterocyclic Compounds,
2017
6.

Fershtat L.L., Makhova N.N.
ChemMedChem,
2017
7.
Ustyuzhanina N.E., Fershtat L.L., Gening M.L., Nifantiev N.E., Makhova N.N.
Mendeleev Communications,
2016
8.
Ustyuzhanina N.E., Fershtat L.L., Gening M.L., Nifantiev N.E., Makhova N.N.
Mendeleev Communications,
2018
9.
![An efficient access to (1H-tetrazol-5-yl)furoxan ammonium salts via a two-step dehydration/[3+2]-cycloaddition strategy](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Fershtat L.L., Epishina M.A., Kulikov A.S., Ovchinnikov I.V., Ananyev I.V., Makhova N.N.
Tetrahedron,
2015
10.

Fershtat L.L., Ananyev I.V., Makhova N.N.
RSC Advances,
2015
11.

Fershtat L.L., Larin A.A., Epishina M.A., Kulikov A.S., Ovchinnikov I.V., Ananyev I.V., Makhova N.N.
Tetrahedron Letters,
2016
12.

Fershtat L.L., Epishina M.A., Ovchinnikov I.V., Struchkova M.I., Romanova A.A., Ananyev I.V., Makhova N.N.
Tetrahedron Letters,
2016
13.

Fershtat L.L., Makhova N.N.
Russian Chemical Reviews,
2016
14.
![Versatile approach to heteroarylfuroxan derivatives from oximinofuroxans via a one-pot, nitration/thermolysis/[3+2]-cycloaddition cascade](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Larin A.A., Fershtat L.L., Ananyev I.V., Makhova N.N.
Tetrahedron Letters,
2017
15.
Zlotin S.G., Churakov A.M., Dalinger I.L., Luk’yanov O.A., Makhova N.N., Sukhorukov A.Y., Tartakovsky V.A.
Mendeleev Communications,
2017
16.

Fershtat L.L., Larin A.A., Epishina M.A., Ovchinnikov I.V., Kulikov A.S., Ananyev I.V., Makhova N.N.
RSC Advances,
2016
17.
Fershtat L.L., Radzhabov M.R., Romanova A.A., Ananyev I.V., Makhova N.N.
Arkivoc,
2017
18.
Kulikov A.S., Larin A.A., Fershtat L.L., Anikina L.V., Pukhov S.A., Klochkov S.G., Struchkova M.I., Romanova A.A., Ananyev I.V., Makhova N.N.
Arkivoc,
2017
19.

Anacona J.R., Rodriguez J.L., Camus J.
Spectrochimica Acta - Part A: Molecular and Biomolecular Spectroscopy,
2014
20.

Taylor B.S., Hunt G., Abrams E.J., Coovadia A., Meyers T., Sherman G., Strehlau R., Morris L., Kuhn L.
AIDS Research and Human Retroviruses,
2011
21.
10.1016/j.mencom.2018.11.020_bibe0035
Al-mamari
Der Pharma Chemica,
2014
22.
10.1016/j.mencom.2018.11.020_bibe0040
Siddiqui
Int. J. Pharm. Sci. Drug Res.,
2009
23.
10.1016/j.mencom.2018.11.020_bibe0045
Siddiqui
Int. J. Drug Dev. Res.,
2011
24.
10.1016/j.mencom.2018.11.020_bibe0050
Kapadiya
Chem. Biol. Interface,
2015
25.
10.1016/j.mencom.2018.11.020_sbrefe0055a
Chawla
Adv. J. Pharm. Life Sci. Res.,
2014
26.

Gomha S.M., Abdelaziz M.R., Kheder N.A., Abdel-aziz H.M., Alterary S., Mabkhot Y.N.
Chemistry Central Journal,
2017
27.

Kulikova L.B., Ezhova G.I., Kravchenko N.E., Dorofeeva O.V., Kulikov A.S., Zavozin A.G.
Russian Chemical Bulletin,
2007
28.

Beyer H.
Zeitschrift für Chemie,
2010
29.
10.1016/j.mencom.2018.11.020_sbrefe0065b
Busby
J. Chem. Soc.,
1980
30.

Sheremetev A.B., Kulikov A.S., Khmel'nitskii L.I.
Russian Chemical Bulletin,
1993
31.

Rollas S., Küçükgüzel S.
Molecules,
2007
32.

Kamal R., Kumar V., Kumar R.
Chemistry - An Asian Journal,
2016
33.

Makhova N.N., Fershtat L.L.
Tetrahedron Letters,
2018
34.

Fernandes G.F., de Souza P.C., Marino L.B., Chegaev K., Guglielmo S., Lazzarato L., Fruttero R., Chung M.C., Pavan F.R., dos Santos J.L.
European Journal of Medicinal Chemistry,
2016
35.

Dutra L.A., Guanaes J.F., Johmann N., Lopes Pires M.E., Chin C.M., Marcondes S., Dos Santos J.L.
Bioorganic and Medicinal Chemistry Letters,
2017
36.

Cabrera M., Mastandrea I., Otero G., Cerecetto H., González M.
Bioorganic and Medicinal Chemistry,
2016
37.
![The First Reliable, General Synthesis of the 5-Oxo Derivatives of 5,6-Dihydro-1,2,4-triazolo[4,3-c]pyrimidine and the Rates of Isomerization of the [4,3-c] Compounds into Their [1,5-c] Isomers](/storage/images/resized/SovkqI6gX6to8o5XBnS9RNxEOYVicn7hatkMzrvl_small_thumb.webp)
38.

Kulikov A.S., Makhova N.N.
Russian Chemical Bulletin,
1998