Abstract
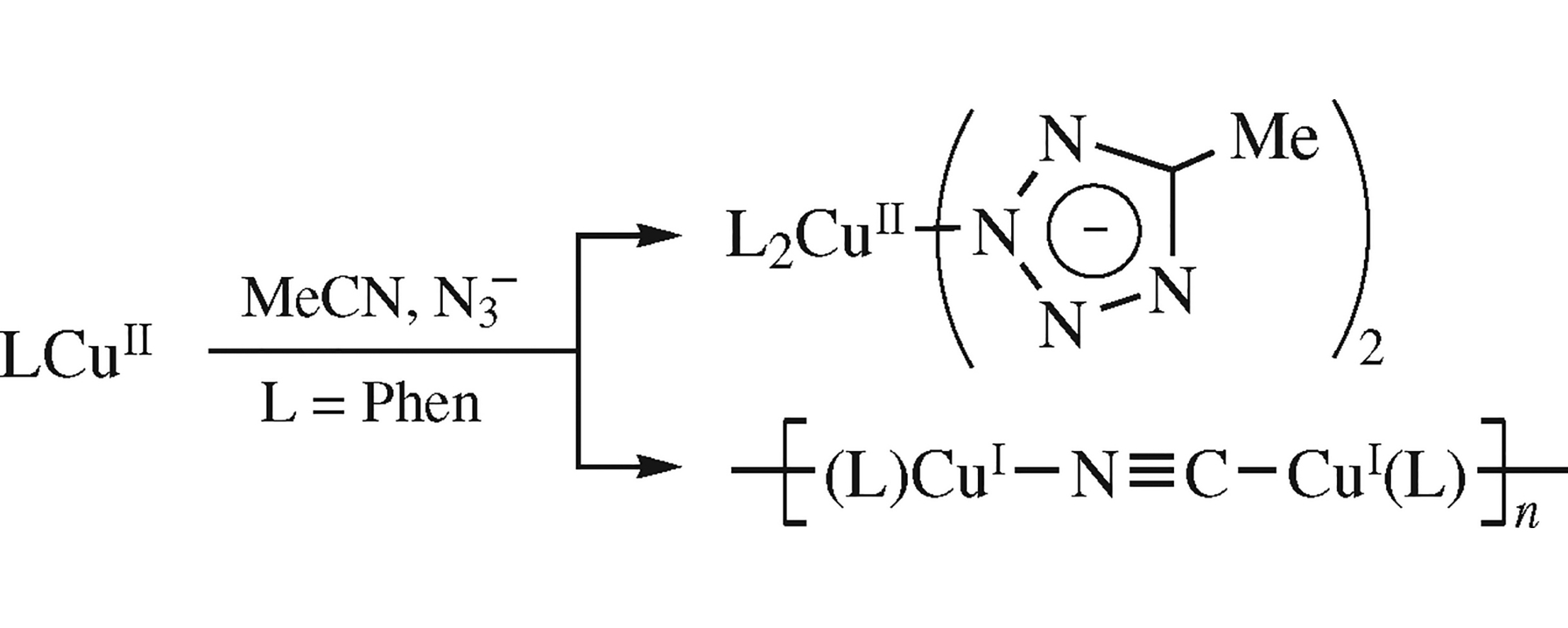
The hydrothermal reaction of NaN3 with Cu(Phen)Cl2 in acetonitrile solutions results in the formation of two complexes: new d9 mononuclear cis-[CuII(Phen)2(mtz)2]·H2O and d10 coordination polymer catena-[CuI(Phen)(μ-CN)]n (mtz is 5-methyltetrazolate anion and Phen is 1,10-phenanthroline). The process involves in situ formation of mtz ligand via cycloaddition of acetonitrile and azide (in the case of [CuII(Phen)2(mtz)2]) and cleavage of acetonitrile C–C bond (in the case of [CuI(Phen)(μ-CN)]n). Both complexes were fully characterized by a comprehensive set of methods, including the single crystal X-ray diffraction data.
References
1.
10.1016/j.mencom.2018.11.014_bibe0010
Comprehensive Coordination Chemistry II,
2003
2.

Aromí G., Barrios L.A., Roubeau O., Gamez P.
Coordination Chemistry Reviews,
2011
3.
10.1016/j.mencom.2018.11.014_sbrefe0015b
Popova
Arkivoc,
2012
4.

Du C., Fan J., Wang X., Zhou S., Wang D.
Journal of Molecular Structure,
2017
5.

Takayama A., Suzuki T., Ikeda M., Sunatsuki Y., Kojima M.
Dalton Transactions,
2013
6.

Lin Q., Wu T., Zheng S., Bu X., Feng P.
Journal of the American Chemical Society,
2011
7.

Li X., Li Z., Xu X., Guo X.
Journal of Coordination Chemistry,
2014
8.

Jeong S., Song X., Jeong S., Oh M., Liu X., Kim D., Moon D., Lah M.S.
Inorganic Chemistry,
2011
9.

Xiong S., Gong Y., Wang H., Wang H., Liu Q., Gu M., Wang X., Chen B., Wang Z.
Chemical Communications,
2014
10.
![A multicentre-bonded [ZnI]8 cluster with cubic aromaticity](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Cui P., Hu H., Zhao B., Miller J.T., Cheng P., Li J.
Nature Communications,
2015
11.

Wu T., Yi B., Li D.
Inorganic Chemistry,
2005
12.

Lu Y., Wang M., Zhou W., Xu G., Guo G., Huang J.
Inorganic Chemistry,
2008
13.

Zhao F., Che Y., Zheng J., Grandjean F., Long G.J.
Inorganic Chemistry,
2012
14.

Gaspar A.B., Ksenofontov V., Seredyuk M., Gütlich P.
Coordination Chemistry Reviews,
2005
15.

Liu J., Dong H., Yang E., Zhao X.
Zeitschrift fur Anorganische und Allgemeine Chemie,
2013
16.

Łukowska-Chojnacka E., Mierzejewska J., Milner-Krawczyk M., Bondaryk M., Staniszewska M.
Bioorganic and Medicinal Chemistry,
2016
17.

Surendra Babu M.S., Rao B.U., Krishna V., Mustafa S., Rao G.N.
Journal of Saudi Chemical Society,
2017
18.

Ostrovskii V.A., Popova E.A., Trifonov R.E.
Advances in Heterocyclic Chemistry,
2017
19.

Demko Z.P., Sharpless K.B.
Journal of Organic Chemistry,
2001
20.

Stawicka K., Drazkiewicz K., Ziolek M.
Microporous and Mesoporous Materials,
2018
21.

Liang L., Astruc D.
Coordination Chemistry Reviews,
2011
22.

Aldhoun M., Massi A., Dondoni A.
Journal of Organic Chemistry,
2008
23.

Jewett J.C., Bertozzi C.R.
Chemical Society Reviews,
2010
24.

Xiong R., Xue X., Zhao H., You X., Abrahams B.F., Xue Z.
Angewandte Chemie - International Edition,
2002
25.

Nasani R., Saha M., Mobin S.M., Mukhopadhyay S.
Polyhedron,
2013
26.

Ma L., Qiu Y., Peng G., Cai J., Deng H.
European Journal of Inorganic Chemistry,
2011
27.

Gerhards H., Krest A., Eulgem P.J., Naumann D., Rokitta D., Valldor M., Klein A.
Polyhedron,
2015
28.

Dehghani F., Sardarian A.R., Esmaeilpour M.
Journal of Organometallic Chemistry,
2013
29.

Xu F., Huang W., You X.
Dalton Transactions,
2010
30.

Burmeister J.L., Edwards L.M.
Journal of the Chemical Society A Inorganic Physical Theoretical,
1971
31.
![Synthesis, Structure and Photoluminescent Studies of a Novel Supramolecular [Ag(phen)(CN)]·(phen) Complex](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Huang X., Zheng S., Zhang J., Chen X.
European Journal of Inorganic Chemistry,
2004
32.

Zhang X., Dong G.Y., Yu B., Van Hecke K., Cui G.H.
Transition Metal Chemistry,
2015
33.

Lu T., Zhuang X., Li Y., Chen S.
Journal of the American Chemical Society,
2004
34.

Liu Y., Wang Z., He X., Shao M., Li M.
Inorganic Chemistry Communication,
2017
35.

Chandraleka S., Ramya K., Chandramohan G., Dhanasekaran D., Priyadharshini A., Panneerselvam A.
Journal of Saudi Chemical Society,
2014
36.

He X., Lu C., Yuan D., Chen S., Chen J.
European Journal of Inorganic Chemistry,
2005
37.

Kaur A., Pintauer T.
European Journal of Inorganic Chemistry,
2013
38.
10.1016/j.mencom.2018.11.014_bibe0095
Nie
Acta Crystallogr.,
2010
39.
10.1016/j.mencom.2018.11.014_bibe0100
Shao
Acta Crystallogr.,
2004
40.

Wang C., Li J., Fan X., Zhao F., Zhang W., Zhang G., Gao Z.
European Journal of Inorganic Chemistry,
2015
41.

Szafranowska B., Beck J.
European Journal of Inorganic Chemistry,
2013
42.
10.1016/j.mencom.2018.11.014_bibe0115
Zhang
Acta Crystallogr.,
2007
43.
10.1016/j.mencom.2018.11.014_bibe0120
Huang
Jiegou Huaxue (J. Struct. Chem.),
2007
44.
![Hydrothermal synthesis, crystal structure and third-order non-linear optical property of a novel one-dimensional copper(I) cyanide–organodiimine coordination polymer [Cu6(CN)6(phen)4]n (phen=1,10-phenanthroline)](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Yu J., Xu J., Yang Q., Pan L., Wang T., Lü C., Ma T.
Journal of Molecular Structure,
2003
45.
![Solvothermal Synthesis, Crystal Structure and DNA Binding Studies of a 3D Supramolecular Complex {[Cu(phen)CN][Cu(phen)][Cu(CN)2]} n Assembled by Double Curvy Chains](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Dong F., Li Y., Wu Z., Sun Y., Sun W., Liu Z., Song Y.
Journal of Inorganic and Organometallic Polymers and Materials,
2008
46.

Zhao H., Qu Z., Ye H., Xiong R.
Chemical Society Reviews,
2008
47.

Himo F., Demko Z.P., Noodleman L., Sharpless K.B.
Journal of the American Chemical Society,
2002
48.
10.1016/j.mencom.2018.11.014_bibe0145
Ostrovskii
2008
49.

Feierman D.E., Cederbaum A.I.
Chemical Research in Toxicology,
1989
50.

Hu W., Teng F., Peng H., Yu J., Sun S., Cheng J., Shao Y.
Tetrahedron Letters,
2015
51.
10.1016/j.mencom.2018.11.014_bibe0160
Sheldrick
Acta Crystallogr.,
2015
52.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
53.
10.1016/j.mencom.2018.11.014_bibe0170
Spek
Acta Crystallogr.,
2015
54.
CrysAlis(Pro), Rigaku Oxford Diffraction, 2017, Version 1.171.39.35a.