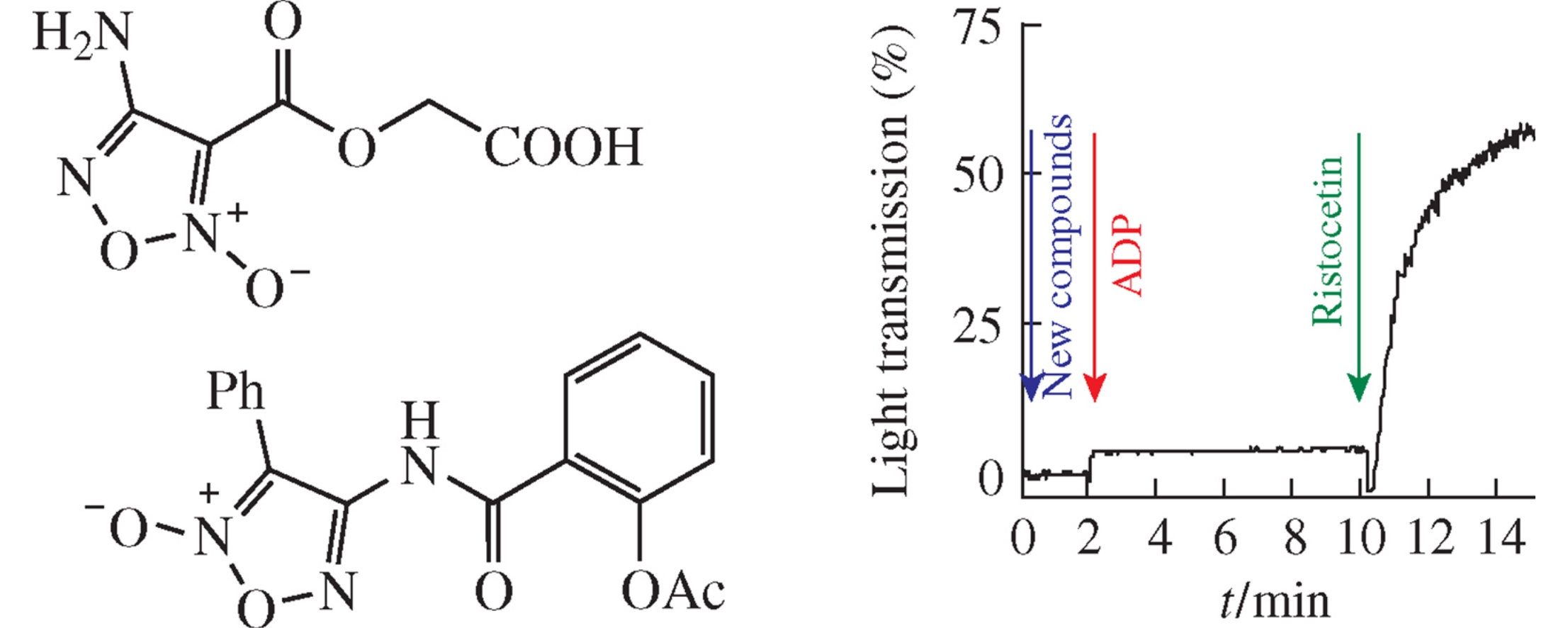
Abstract
A series of functionalized furoxans was synthesized, and their effect on the platelet aggregation was estimated using a set of inducers. New hybrid structures comprising the furoxan ring and glycolic or acetylsalicylic acid motifs effectively inhibit aggregate formation induced by adenosine diphosphate and adrenaline. Studies of their NO-donor ability showed that their antiplatelet effects were mainly independent of the NO action.
References
1.

Verstraete M.
European Heart Journal,
1995
2.

Warner T.D., Nylander S., Whatling C.
British Journal of Clinical Pharmacology,
2011
3.
10.1016/j.mencom.2018.11.010_bibe0020
Tetik
Clin. Appl. Thromb. Hemost.,
2010
4.

Hohlfeld T., Scharnowski F., Braun M., Schrör K.
Thrombosis and Haemostasis,
1994
5.
10.1016/j.mencom.2018.11.010_bibe0030
Verstraete
Drugs Today,
1993
6.

Ferioli R., Folco G.C., Ferretti C., Gasco A.M., Medana C., Fruttero R., Civelli M., Gasco A.
British Journal of Pharmacology,
1995
7.

Del Grosso E., Boschi D., Lazzarato L., Cena C., Di Stilo A., Fruttero R., Moro S., Gasco A.
Chemistry and Biodiversity,
2005
8.

Nortcliffe A., Botting N.P., O'Hagan D.
Organic and Biomolecular Chemistry,
2013
9.

Rosseto L., Pires M., Melchior A., Bosquesi P., Pavan A., Marcondes S., Chung M., Santos J.
Molecules,
2015
10.
Ananikov V.P., Galkin K.I., Egorov M.P., Sakharov A.M., Zlotin S.G., Redina E.A., Isaeva V.I., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2016
11.

Vereshchagin A.N.
Russian Chemical Bulletin,
2017
12.
Ananikov V.P., Eremin D.B., Yakukhnov S.A., Dilman A.D., Levin V.V., Egorov M.P., Karlov S.S., Kustov L.M., Tarasov A.L., Greish A.A., Shesterkina A.A., Sakharov A.M., Nysenko Z.N., Sheremetev A.B., Stakheev A.Y., et. al.
Mendeleev Communications,
2017
13.
Zlotin S.G., Churakov A.M., Dalinger I.L., Luk’yanov O.A., Makhova N.N., Sukhorukov A.Y., Tartakovsky V.A.
Mendeleev Communications,
2017
14.

Fershtat L.L., Makhova N.N.
ChemMedChem,
2017
15.

Makhova N.N., Rakitin O.A.
Chemistry of Heterocyclic Compounds,
2017
16.
Ustyuzhanina N.E., Fershtat L.L., Gening M.L., Nifantiev N.E., Makhova N.N.
Mendeleev Communications,
2016
17.
K. Schönafinger and H. Bohn, Patent US 5486531, 1996.
18.

Bohn H., Brendel J., Martorana P.A., Schönafinger K.
British Journal of Pharmacology,
1995
19.

Balbo S., Lazzarato L., Di Stilo A., Fruttero R., Lombaert N., Kirsch-Volders M.
Toxicology Letters,
2008
20.
Ustyuzhanina N.E., Fershtat L.L., Gening M.L., Nifantiev N.E., Makhova N.N.
Mendeleev Communications,
2018
21.

Ovchinnikov I.V., Kulikov A.S., Makhova N.N., Tosco P., Di Stilo A., Fruttero R., Gasco A.
Il Farmaco,
2003
22.

23.
Ovchinnikov I.V., Blinnikov A.N., Makhova N.N., Khmel’nitskii L.I.
Mendeleev Communications,
1995
24.

Gagneux A.R., Meier R.
Helvetica Chimica Acta,
1970
25.
Kulikov A.S., Larin A.A., Fershtat L.L., Anikina L.V., Pukhov S.A., Klochkov S.G., Struchkova M.I., Romanova A.A., Ananyev I.V., Makhova N.N.
Arkivoc,
2017
26.

Puri R.N., Colman R.W., Liberman M.A.
Critical Reviews in Biochemistry and Molecular Biology,
1997
27.

Ueno M., Kodali M., Tello-Montoliu A., Angiolillo D.J.
Journal of Atherosclerosis and Thrombosis,
2011