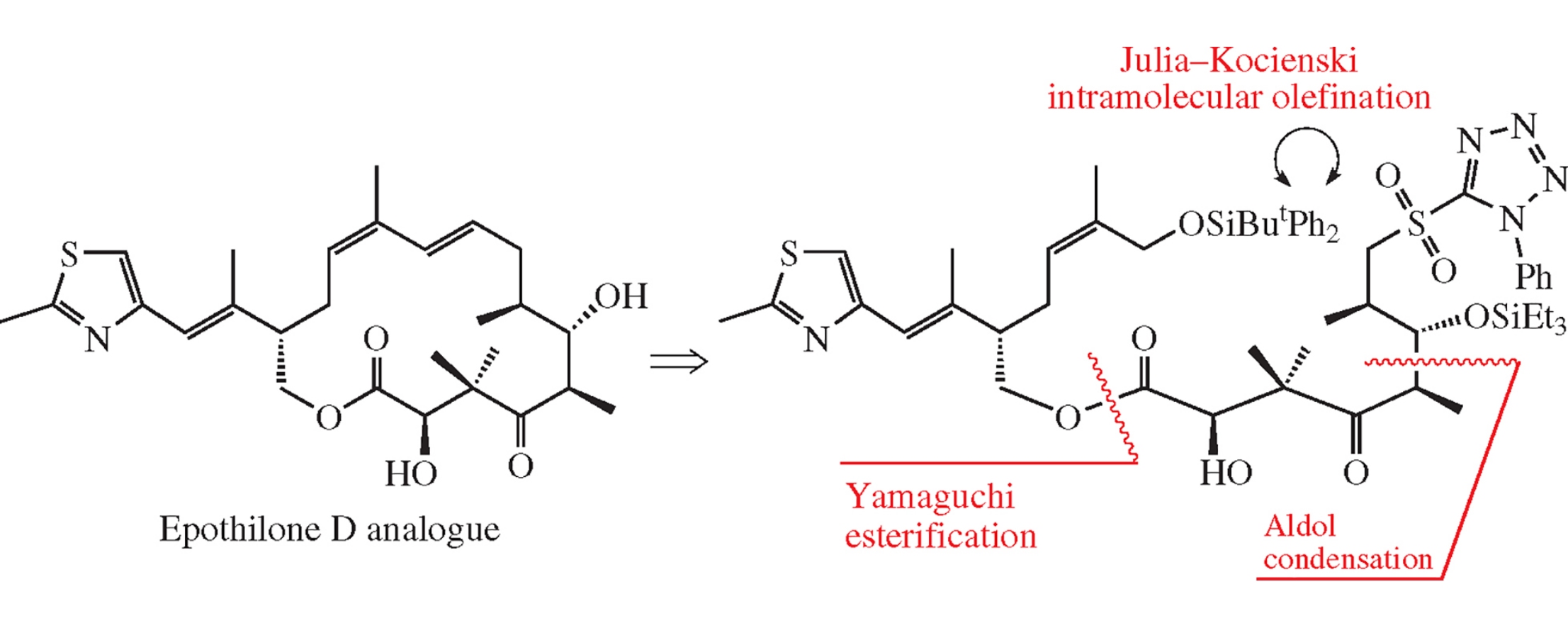
Abstract
Condensation of separately obtained C1–C9 and C10–C21 chiral blocks under Yamaguchi conditions affords the corresponding ester, an acyclic precursor of an epothilone D analogue. The reaction is accompanied by the formation of a side product of acylation of the starting alcohol by the Yamaguchi reagent.
References
1.

Altmann K., Pfeiffer B., Arseniyadis S., Pratt B., Nicolaou K. .
ChemMedChem,
2007
2.

Cao Y., Zheng L., Wang D., Liang X., Gao F., Zhou X.
European Journal of Medicinal Chemistry,
2018
3.

Watkins E.B., Chittiboyina A.G., Avery M.A.
European Journal of Organic Chemistry,
2006
4.

Inanaga J., Hirata K., Saeki H., Katsuki T., Yamaguchi M.
Bulletin of the Chemical Society of Japan,
1979
5.

Cachoux F., Isarno T., Wartmann M., Altmann K.
Angewandte Chemie - International Edition,
2005
6.

Nicolaou K.C., Namoto K., Ritzén A., Ulven T., Shoji M., Li J., D'Amico G., Liotta D., French C.T., Wartmann M., Altmann K., Giannakakou P.
Journal of the American Chemical Society,
2001
7.

Valeev R.F., Bikzhanov R.F., Yagafarov N.Z., Miftakhov M.S.
Tetrahedron,
2012
8.
Valeev R.F., Bikzhanov R.F., Miftakhov M.S.
Mendeleev Communications,
2014
9.

Valeev R.F., Sunagatullina G.R., Miftakhov M.S.
Russian Journal of Organic Chemistry,
2017
10.

De Mico A., Margarita R., Parlanti L., Vescovi A., Piancatelli G.
Journal of Organic Chemistry,
1997
11.

Nicolaou K.C., Hepworth D., King N.P., Finlay M.R., Scarpelli R., Pereira M.M., Bollbuck B., Bigot A., Werschkun B., Winssinger N.
Chemistry - A European Journal,
2000
12.

Nicolaou K.C., Ritzén A., Namoto K., Buey R.M., Dı́az J.F., Andreu J.M., Wartmann M., Altmann K., O'Brate A., Giannakakou P.
Tetrahedron,
2002
13.

Fürstner A., Mathes C., Grela K.
Chemical Communications,
2001
14.

Chakraborty T.K., Dutta S.
Tetrahedron Letters,
1998