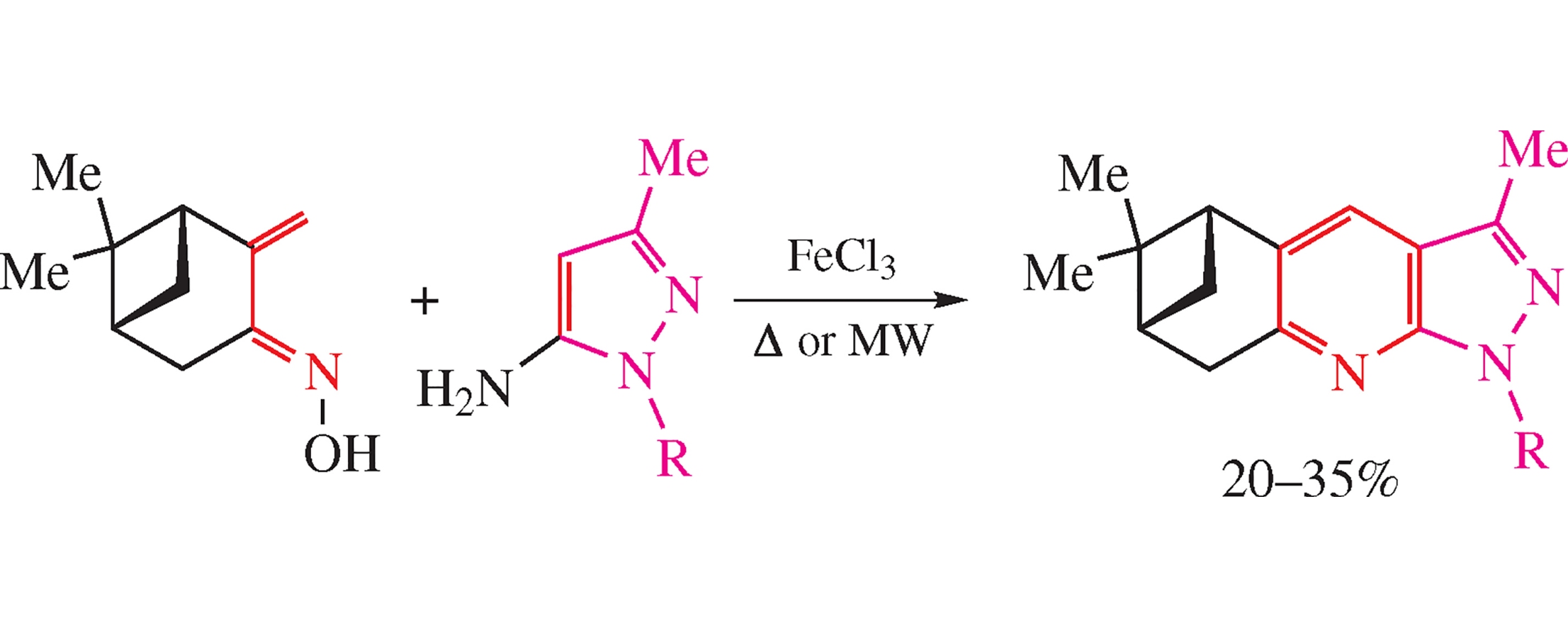
Abstract
Chiral 1H-pyrazolo[3,4-b]pyridines fused with nopinane frame were obtained by FeCl3-catalyzed assembling of pinocarvone oxime and 1-aryl-1H-pyrazol-5-amines. Chemical structures of new compounds were solved by NMR spectroscopy and confirmed by quantum chemical calculations and X-ray crystallography.
References
1.

von Zelewsky A., Mamula O.
Journal of the Chemical Society Dalton Transactions,
2000
2.

Chelucci G., Thummel R.P.
Chemical Reviews,
2002
3.
![Synthesis of novel dipyrazolo[3,4-b:3,4-d]pyridines and study of their fluorescence behavior](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Kendre D.B., Toche R.B., Jachak M.N.
Tetrahedron,
2007
4.
![Effect of substituents on absorption and fluorescence properties of pyrazolo[3,4-b]pyrrolo[2,3-d]pyridines](/storage/images/resized/qZfu8vVyaDIo81Au73p6SNE8AGGzf73yfiTC33bu_small_thumb.webp)
Ghotekar B.K., Kazi M.A., Jachak M.N., Toche R.B.
Canadian Journal of Chemistry,
2008
5.
![An Efficient Synthesis and Fluorescent Properties of Pyrazole[3,4-b]thieno[2,3-e]pyridine Derivatives](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Yao C., Lu K., Song B., Liu B., Li T., Yu C.
Journal of Heterocyclic Chemistry,
2014
6.
![An efficient Synthesis and Photophysical Properties of 1H-pyrazolo-[3,4-b]pyridine and pyrazolo[3,4-b][1,8]naphthyridines](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Deore R., Dingore K., Jachak M.
Journal of Fluorescence,
2015
7.
![Synthesis and properties of fluorescence dyes: tetracyclic pyrazolo[3,4-b]pyridine-based coumarin chromophores with intramolecular charge transfer character.](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Chen J., Liu W., Ma J., Xu H., Wu J., Tang X., Fan Z., Wang P.
Journal of Organic Chemistry,
2012
8.
![Turn-on fluorescence sensor based on the aggregation of pyrazolo[3,4-b]pyridine-based coumarin chromophores induced by Hg2+](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Chen J., Liu W., Wang Y., Zhang H., Wu J., Xu H., Ju W., Wang P.
Tetrahedron Letters,
2013
9.
![Interactions of Lysozyme with 6-Amino-4-aryl-3-methyl-1-phenyl-1N-pyrazolo[3,4-b]pyridine-5-carbonitriles: a Fluorescence Quenching Study](/storage/images/resized/MriDiasGda8aAymM983PIz18dzVMXwO888XReHom_small_thumb.webp)
Wu H., Lian S., Shen Y., Wan Y.
Analytical Sciences,
2007
10.
![A Fluorescence Study of New Angular Polycyclic Blue Light-Emitting pyrazolo[3,4-h][1,6]naphthyridine and their Interaction with Bovine Serum Albumin (BSA)](/storage/images/resized/voXLqlsvTwv5p3iMQ8Dhs95nqB4AXOG7Taj7G4ra_small_thumb.webp)
Patil S.R., Shelar D.P., Rote R.V., Jachak M.N.
Journal of Fluorescence,
2011
11.
10.1016/j.mencom.2018.11.006_bibe0060
Dubey
Asian J. Chem.,
2002
12.

Costantino L., Barlocco D.
Current Medicinal Chemistry,
2005
13.

Duarte C., Barreiro E., Fraga C.
Mini-Reviews in Medicinal Chemistry,
2007
14.

Meiners B.A., Salama A.I.
European Journal of Pharmacology,
1982
15.

Revesz L., Blum E., Di Padova F.E., Buhl T., Feifel R., Gram H., Hiestand P., Manning U., Neumann U., Rucklin G.
Bioorganic and Medicinal Chemistry Letters,
2006
16.
![Synthesis and 3D QSAR of New Pyrazolo[3,4-b]pyridines: Potent and Selective Inhibitors of A1 Adenosine Receptors](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Manetti F., Schenone S., Bondavalli F., Brullo C., Bruno O., Ranise A., Mosti L., Menozzi G., Fossa P., Trincavelli M.L., Martini C., Martinelli A., Tintori C., Botta M.
Journal of Medicinal Chemistry,
2005
17.
![6-Aryl-pyrazolo[3,4-b]pyridines: potent inhibitors of glycogen synthase kinase-3 (GSK-3)](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Witherington J., Bordas V., Gaiba A., Garton N.S., Naylor A., Rawlings A.D., Slingsby B.P., Smith D.G., Takle A.K., Ward R.W.
Bioorganic and Medicinal Chemistry Letters,
2003
18.

Wenglowsky S., Ren L., Ahrendt K.A., Laird E.R., Aliagas I., Alicke B., Buckmelter A.J., Choo E.F., Dinkel V., Feng B., Gloor S.L., Gould S.E., Gross S., Gunzner-Toste J., Hansen J.D., et. al.
ACS Medicinal Chemistry Letters,
2011
19.

Chibiryaev A.M., De Kimpe N., Tkachev A.V.
Tetrahedron Letters,
2000
20.

Vasilyev E.S., Agafontsev A.M., Tkachev A.V.
Synthetic Communications,
2014
21.
Vasilyev E.S., Bagryanskaya I.Y., Tkachev A.V.
Mendeleev Communications,
2017
22.

Allais C., Grassot J., Rodriguez J., Constantieux T.
Chemical Reviews,
2014
23.

Gallaher K.L., Lukco D., Grasselli J.G.
Canadian Journal of Chemistry,
1985
24.

Moir J.
Journal of the Chemical Society Transactions,
1902
25.

Ganesan A., Heathcock C.H.
Journal of Organic Chemistry,
1993
26.

Volochnyuk D.M., Ryabukhin S.V., Plaskon A.S., Dmytriv Y.V., Grygorenko O.O., Mykhailiuk P.K., Krotko D.G., Pushechnikov A., Tolmachev A.A.
Journal of Combinatorial Chemistry,
2010
27.
10.1016/j.mencom.2018.11.006_bibe0140
Tkachev
Ross. Khim. Zh. (Zh. Ross. Khim. Ob-va im. D. I. Mendeleeva),
1998
28.

Petukhov P.A., Bizyaev S.N., Tkachev À.V.
Russian Chemical Bulletin,
2001
29.
![A Green Approach to the Synthesis of Biologically Important Indeno[2,1-e]pyrazolo[5,4-b]pyridines via Microwave-assisted Multi-component Reactions in Water](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
SHI F., ZHANG Y., TU S., ZHOU D., LI C., SHAO Q., CAO L.
Chinese Journal of Chemistry,
2008
30.

Zhu S., Ji S., Zhao K., Liu Y.
Tetrahedron Letters,
2008
31.
![An efficient and clean synthesis of indeno[1,2-b]pyrazolo[4,3-e] pyridin-5(1H)-one derivatives under microwave irradiation in water](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Wang S., Ma N., Zhang G., Shi F., Jiang B., Lu H., Gao Y., Tu S.
Journal of Heterocyclic Chemistry,
2010
32.
![Facile Three-Component Synthesis of Macrocyclane-Fused Pyrazolo[3,4-b]pyridine Derivatives](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Jiang B., Liu Y., Tu S.
European Journal of Organic Chemistry,
2011
33.

34.

Lidström P., Tierney J., Wathey B., Westman J.
Tetrahedron,
2001
35.

Neese F.
Wiley Interdisciplinary Reviews: Computational Molecular Science,
2011
36.

Toomsalu E., Burk P.
Journal of Molecular Modeling,
2015
37.

Foresman J.B., Keith T.A., Wiberg K.B., Snoonian J., Frisch M.J.
The Journal of Physical Chemistry,
1996
38.

Aidas K., Angeli C., Bak K.L., Bakken V., Bast R., Boman L., Christiansen O., Cimiraglia R., Coriani S., Dahle P., Dalskov E.K., Ekström U., Enevoldsen T., Eriksen J.J., Ettenhuber P., et. al.
Wiley Interdisciplinary Reviews: Computational Molecular Science,
2013
39.
APEX2, Version 2.0, SAINT, Version 8.18c and SADABS, Version 2.11, Bruker Advanced X-ray Solutions, Bruker AXS, Madison, WI, USA, 2000-2012.
40.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007
41.

Dolomanov O.V., Bourhis L.J., Gildea R.J., Howard J.A., Puschmann H.
Journal of Applied Crystallography,
2009
42.
M. J. Turner, J. J. McKinnon, S. K. Wolff, D. J. Grimwood, P. R. Spackman, D. Jayatilaka and M. A. Spackman, CrystalExplorer17, University of Western Australia, 2017, http://hirshfeldsurface.net.
43.

Jayatilaka D., Grimwood D.J.
Lecture Notes in Computer Science,
2003
44.

Turner M.J., Grabowsky S., Jayatilaka D., Spackman M.A.
Journal of Physical Chemistry Letters,
2014
45.
![Efficient Synthesis of 3-Substituted 1,2,4-Triazolo[4,3-a]pyridine by [Bis(Trifluroacetoxy)iodo]benzene-Catalyzed Oxidative Intramolecular Cyclization of Heterocyclic Hydrazones](/storage/images/resized/5YZtvLvkPZuc2JHOaZsjCvGSHFCuC3drUwN3YAc5_small_thumb.webp)
Padalkar V.S., Patil V.S., Phatangare K.R., Umape P.G., Sekar N.
Synthetic Communications,
2011