Abstract
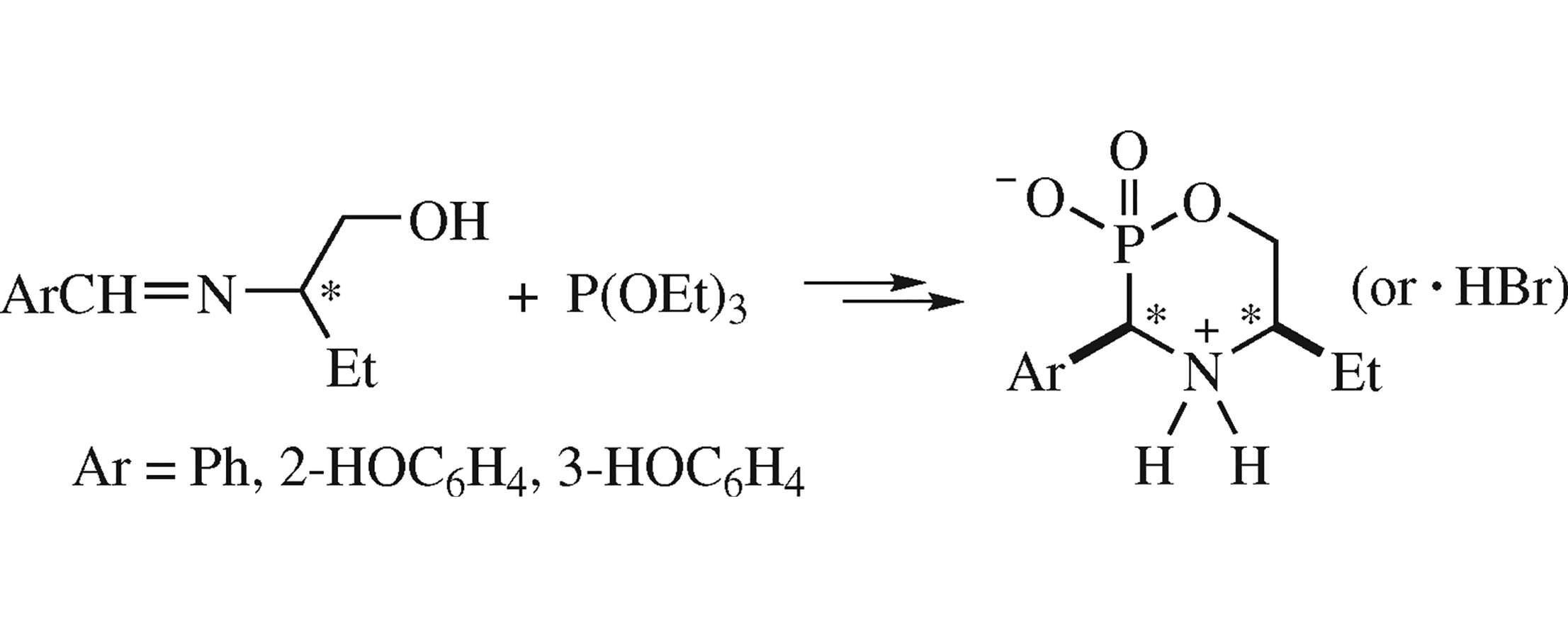
Diastereoselective synthesis of racemic and enantiopure 3-aryl-5-ethyl-1,4,2-oxazaphosphorines, including those bearing phenolic hydroxyl groups in the exocyclic aromatic fragment, was implemented by the reaction of imines derived from (±)-and (R)-(−)-2-aminobutan-1-ol and (hydroxy)benzaldehydes with triethyl phosphite and trifluoroacetic acid, followed by the one-pot dealkylation of the intermediate esters.
References
1.
10.1016/j.mencom.2018.11.004_sbref0005a
Aminophosphonic and Aminophosphinic Acids: Chemistry and Biological Activity,
2000
2.

Orsini F., Sello G., Sisti M.
Current Medicinal Chemistry,
2009
3.
10.1016/j.mencom.2018.11.004_bib0010
Pudovik
Dokl. Akad. Nauk SSSR,
1973
4.
10.1016/j.mencom.2018.11.004_bib0015
Kibardina
Zh. Obshch. Khim.,
1975
5.
10.1016/j.mencom.2018.11.004_sbref0020a
Samitov
Zh. Obshch. Khim.,
1975
6.

Sigalov M.V., Pestunovich V.A., Nikitin V.M., Atavin A.S., Kukharev B.F.
Russian Chemical Bulletin,
1978
7.

Maury C., Gharbaoui T., Royer J., Husson H.
Journal of Organic Chemistry,
1996
8.

Maury C., Wang Q., Gharbaoui T., Chiadmi M., Tomas A., Royer J., Husson H.
Tetrahedron,
1997
9.

Fadel A., Tesson N.
Tetrahedron Asymmetry,
2000
10.
Dimukhametov M.N., Bajandina E.V., Davydova E.Y., Dobrynin A.B., Gubaidullin A.T., Litvinov I.A., Alfonsov V.A.
Mendeleev Communications,
2001
11.
Dimukhametov M.N., Davydova E.Y., Bayandina E.V., Dobrynin A.B., Litvinov I.A., Alfonsov V.A.
Mendeleev Communications,
2001
12.

Dimukhametov M.N., Bayandina E.V., Davydova E.Y., Zyablikova T.A., Dobrynin A.B., Litvinov I.A., Al"fonsov V.A.
Russian Chemical Bulletin,
2001
13.

Dimukhametov M.N., Bajandina E.V., Davydova E.Y., Litvinov I.A., Gubaidullin A.T., Dobrynin A.B., Zyablikova T.A., Alfonsov V.A.
Heteroatom Chemistry,
2003
14.

Alfonsov V.A., McKenna C.E., Bayandina E.V., Kashemirov B.A., Yarmieva L.N., Punegova L.N., Kataeva O.N.
Heteroatom Chemistry,
2008
15.

NMR and X-ray crystallographic studies of axial and equatorial 2-ethoxy-2-oxo-1,4,2-oxazaphosphinane
Linzaga I., Escalante J., Muñoz M., Juaristi E.
Tetrahedron,
2002
16.

Cristau H., Monbrun J., Tillard M., Pirat J.
Tetrahedron Letters,
2003
17.

Pirat J., Monbrun J., Virieux D., Volle J., Tillard M., Cristau H.
Journal of Organic Chemistry,
2005
18.

Pirat J., Monbrun J., Virieux D., Cristau H.
Tetrahedron,
2005
19.
![Chiral phosphinyl analogues of 2-C-arylmorpholinols: 2-aryl-3,5-diphenyl-[1,4,2]-oxazaphosphinanes](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Volle J., Virieux D., Starck M., Monbrun J., Clarion L., Pirat J.
Tetrahedron Asymmetry,
2006
20.
![Phosphinyl analogues of hydroxybupropion: (±)-2-aryl-3,3,5,5-tetramethyl-[1,4,2]-oxazaphosphinanes](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Volle J., Kaloyanov N., Saada M.C., Virieux D., Pirat J.
Tetrahedron Letters,
2007
21.

Volle J., Filippini D., Krawczy B., Kaloyanov N., Van der Lee A., Maurice T., Pirat J., Virieux D.
Organic and Biomolecular Chemistry,
2010
22.

Metlushka K., Kashemirov B., Zheltukhin V., Sadkova D., Büchner B., Hess C., Kataeva O., McKenna C., Alfonsov V.
Chemistry - A European Journal,
2009
23.

Metlushka K.E., Sadkova D.N., Shaimardanova L.N., Nikitina K.A., Tufatullin A.I., Kataeva O.N., Alfonsov V.A.
Russian Chemical Bulletin,
2014
24.
Nikitina K.A., Metlushka K.E., Sadkova D.N., Shaimardanova L.N., Alfonsov V.A.
Mendeleev Communications,
2016
25.

Salehi P., Dabiri M., Kozehgary G.
Journal of the Iranian Chemical Society,
2010
26.

Paul A., Figuerola A., Bertolasi V., Manna S.C.
Polyhedron,
2016
27.

Dimukhametov M.N., Bayandina E.V., Davydova E.Y., Al'fonsov V.A.
Russian Journal of General Chemistry,
2004
28.

Juhász M., Lázár L., Fülöp F.
Journal of Heterocyclic Chemistry,
2007
29.
10.1016/j.mencom.2018.11.004_bib0070
Gu
Chiral Analysis,
2006
30.
APEX3 Crystallography Software Suite, Bruker AXS, Madison, WI, USA, 2016.
31.
SAINT Crystallography Software Suite, Bruker AXS, Madison, WI, USA, 2016.
32.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007
33.

Krause L., Herbst-Irmer R., Sheldrick G.M., Stalke D.
Journal of Applied Crystallography,
2015
34.

Berlicki Ł., Rudzińska E., Kafarski P.
Tetrahedron Asymmetry,
2003