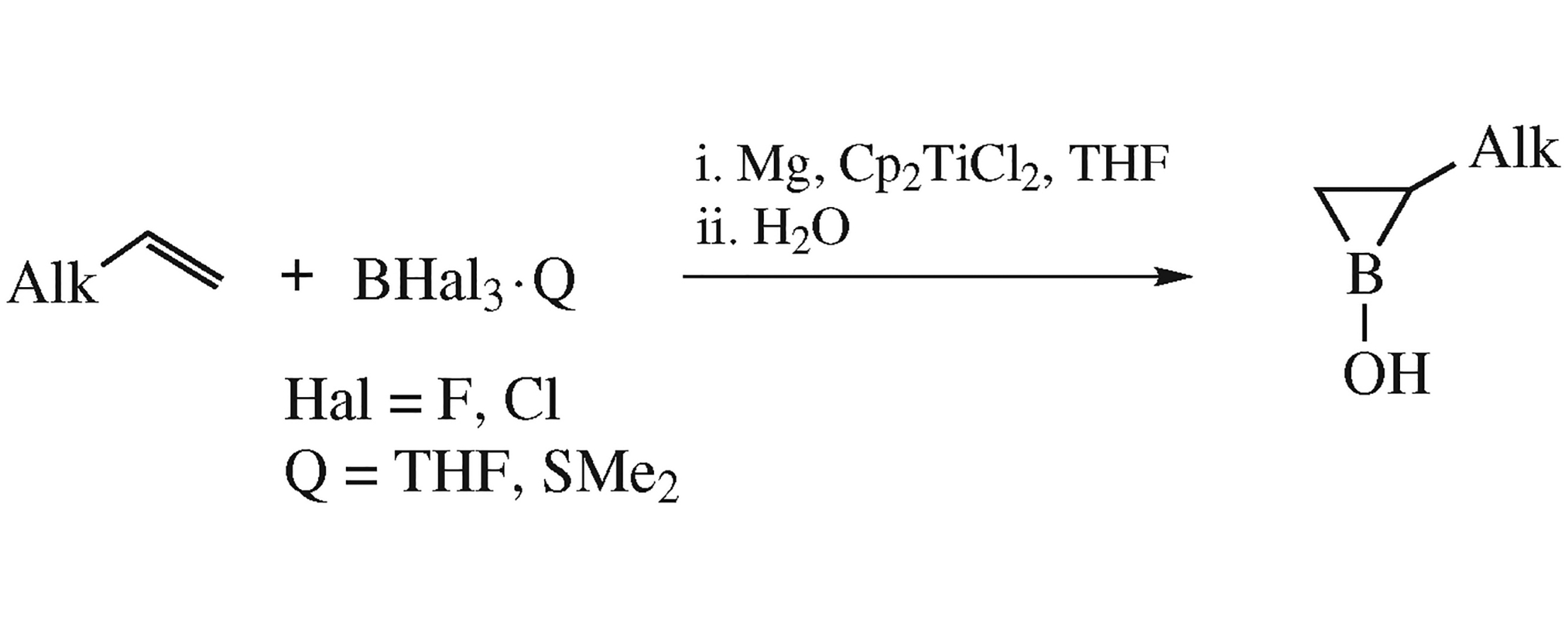Abstract
2-Alkylboriran-1-ols were obtained in a one-pot process by hydrolysis of 1-fluoro- and 1-chloroboriranes in 90–92% yield. The starting 1-haloboriranes were generated by cycloboration of α-olefins with BCl3·SMe2 or BF3·THF in the presence of Mg metal (acceptor of halogen ions) and Cp2TiCl2 catalyst.
References
1.

Khusainova L.I., Khafizova L.O., Tyumkina T.V., Dzhemilev U.M.
Russian Journal of General Chemistry,
2016
2.
10.1016/j.mencom.2018.11.003_sbref0005b
Khusainova
Russ. J. Org. Chem.,
2015
3.

Elewski B.E., Aly R., Baldwin S.L., González Soto R.F., Rich P., Weisfeld M., Wiltz H., Zane L.T., Pollak R.
Journal of the American Academy of Dermatology,
2015
4.

Gupta A.K., Daigle D.
Expert Review of Anti-Infective Therapy,
2014
5.

Baker S.J., Zhang Y., Akama T., Lau A., Zhou H., Hernandez V., Mao W., Alley M.R., Sanders V., Plattner J.J.
Journal of Medicinal Chemistry,
2006
6.

7.

Ivanov A.S., Zhalnina A.A., Shishkov S.V.
Tetrahedron,
2009
8.

Adams J., Behnke M., Chen S., Cruickshank A.A., Dick L.R., Grenier L., Klunder J.M., Ma Y., Plamondon L., Stein R.L.
Bioorganic and Medicinal Chemistry Letters,
1998
9.

Hernandez V., Crépin T., Palencia A., Cusack S., Akama T., Baker S.J., Bu W., Feng L., Freund Y.R., Liu L., Meewan M., Mohan M., Mao W., Rock F.L., Sexton H., et. al.
Antimicrobial Agents and Chemotherapy,
2013
10.
10.1016/j.mencom.2018.11.003_sbref0010h
Sonoiki
Nat. Commun.,
2017
11.

Sene S., McLane J., Schaub N., Bégu S., Hubert Mutin P., Ligon L., Gilbert R.J., Laurencin D.
Journal of Materials Chemistry B,
2016
12.

Hecker S.J., Reddy K.R., Totrov M., Hirst G.C., Lomovskaya O., Griffith D.C., King P., Tsivkovski R., Sun D., Sabet M., Tarazi Z., Clifton M.C., Atkins K., Raymond A., Potts K.T., et. al.
Journal of Medicinal Chemistry,
2015
13.

Leśnikowski Z.J.
Expert Opinion on Drug Discovery,
2016
14.

15.

Baker S.J., Ding C.Z., Akama T., Zhang Y., Hernandez V., Xia Y.
Future Medicinal Chemistry,
2009
16.

Ashe A.J., Klein W., Rousseau R.
Organometallics,
1993
17.

Brown H.C., Ravindran N.
Journal of the American Chemical Society,
1976
18.

Khusainova L.I., Khafizova L.O., Tyumkina T.V., Ryazanov K.S., Dzhemilev U.M.
Journal of Organometallic Chemistry,
2017
19.

Shao P., Shen L., Ye S.
Chinese Journal of Chemistry,
2012
20.

Prat I., Font D., Company A., Junge K., Ribas X., Beller M., Costas M.
Advanced Synthesis and Catalysis,
2013
21.

Shaikh N.S., Junge K., Beller M.
Organic Letters,
2007
22.

Szostak M., Spain M., Eberhart A.J., Procter D.J.
Journal of the American Chemical Society,
2014
23.

Kim J., Koike T., Kotani M., Yamaguchi K., Mizuno N.
Chemistry - A European Journal,
2008
24.

Fernández-Mateos E., Maciá B., Yus M.
Advanced Synthesis and Catalysis,
2013
25.
10.1016/j.mencom.2018.11.003_sbref0035a
Scau
1959
26.
10.1016/j.mencom.2018.11.003_sbref0035b
Alexander
Chemistry in the Laboratory,
1976