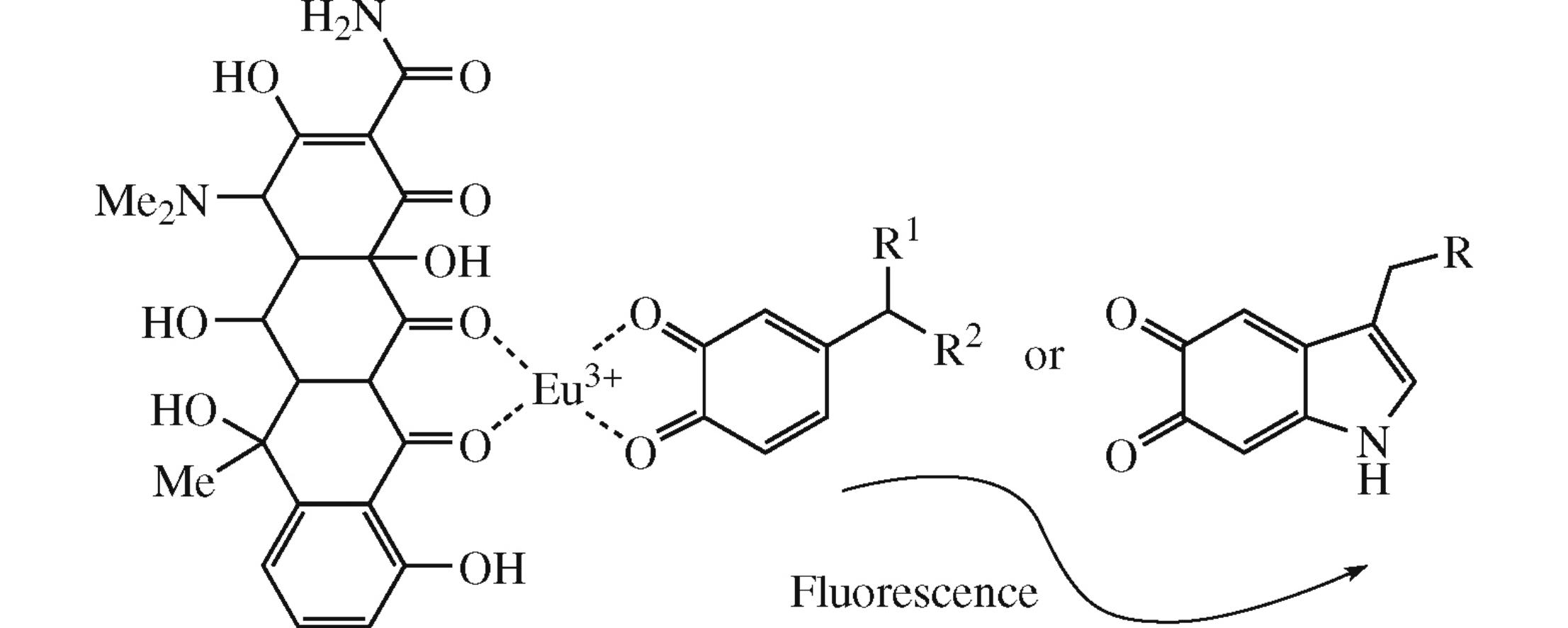
Abstract
New ternary complexes of europium with oxytetracycline possessing the intense fluorescence have been obtained for prospective applications in the high-sensitive detection of biogenic amines and their metabolites in biological fluids. Their stability constants have been determined using the Foster–Hammick–Wardley method based on the fluorescent spectroscopy data. The dependence between structure differences, stability constants of ternary complexes of europium–oxytetracycline with biogenic amines, and their metabolites, and sensitivity of their determination has been established.
References
1.
Lenders J.W., Eisenhofer G.
Endocrinology and Metabolism,
2017
2.

Verly I.R., van Kuilenburg A.B., Abeling N.G., Goorden S.M., Fiocco M., Vaz F.M., van Noesel M.M., Zwaan C.M., Kaspers G.L., Merks J.H., Caron H.N., Tytgat G.A.
European Journal of Cancer,
2017
3.

Smith S.J., Diehl N.N., Smith B.D., Mohney B.G.
Eye,
2010
4.

Factor S.
Experimental Neurology,
1995
5.

Andersen A.D., Blaabjerg M., Binzer M., Kamal A., Thagesen H., Kjaer T.W., Stenager E., Gramsbergen J.B.
Journal of Neurochemistry,
2017
6.

Umegaki H., Tamaya N., Shinkai T., Iguchi A.
Experimental Gerontology,
2000
7.
Kozin S.A., Mitkevich V.A., Makarov A.A.
Mendeleev Communications,
2016
8.

Fonseca B.M., Rodrigues M., Cristóvão A.C., Gonçalves D., Fortuna A., Bernardino L., Falcão A., Alves G.
Journal of Chromatography B: Analytical Technologies in the Biomedical and Life Sciences,
2017
9.

Veselova I.A., Sergeeva E.A., Makedonskaya M.I., Eremina O.E., Kalmykov S.N., Shekhovtsova T.N.
Journal of Analytical Chemistry,
2016
10.

Wang Y., Wang S., Tao L., Min Q., Xiang J., Wang Q., Xie J., Yue Y., Wu S., Li X., Ding H.
Biosensors and Bioelectronics,
2015
11.

Kamruzzaman M., Alam A., Lee S.H., Kim Y.H., Kim S.H.
Luminescence,
2011
12.

Wabaidur S.M., ALOthman Z.A., Naushad M.
Spectrochimica Acta - Part A: Molecular and Biomolecular Spectroscopy,
2012
13.

A simple and selective fluorometric assay for dopamine using a calcein blue–Fe2+ complex fluorophore
Seto D., Maki T., Soh N., Nakano K., Ishimatsu R., Imato T.
Talanta,
2012
14.
Kharcheva A.V., Evdokimov D.A., Reshetova M.D., Yuzhakov V.I., Patsaeva S.V., Borisova N.E.
Mendeleev Communications,
2017
15.
Kopchuk D.S., Krinochkin A.P., Kim G.A., Kozhevnikov D.N.
Mendeleev Communications,
2017
16.

Zhang X., Sweedler J.V.
Analytical Chemistry,
2001
17.

Wang H.Y., Hui Q.S., Xu L.X., Jiang J.G., Sun Y.
Analytica Chimica Acta,
2003
18.

Dehaen G., Absillis G., Driesen K., Binnemans K., Parac-Vogt T.
Helvetica Chimica Acta,
2009
19.

dos Santos Teixeira L., Grasso A.N., Monteiro A.M., Neto A.M., Vieira N.D., Gidlund M., Steffens J., Courrol L.C.
Journal of Fluorescence,
2010
20.
10.1016/j.mencom.2018.09.036_bib0100
Parker
Photoluminescence of Solutions,
1968
21.

Foster R., Hammick D.L., Wardley A.A.
Journal of the Chemical Society (Resumed),
1953
22.

Hirsch L.M., Van Geel T.F., Winefordner J.D., Kelly R.N., Schulman S.G.
Analytica Chimica Acta,
1985
23.
10.1016/j.mencom.2018.09.036_bib0115
Banwell
Fundamentals of Molecular Spectroscopy,
1983
24.

Aldrich J.
International Statistical Review,
1998
25.

Courrol L., Samad R.
Current Pharmaceutical Analysis,
2008
26.

Karaderi, S., Bilgic, D., Dölen, E., Pekin, M.
Reviews in Inorganic Chemistry,
2007
27.

Wu Z.J., Gao F., Wang J.P., Niu C.J., Niu Y.J.
Journal of Coordination Chemistry,
2005
28.
10.1016/j.mencom.2018.09.036_bib0140
Chakrawarti
J. Indian Chem. Soc.,
1983
29.

Walaas E., Walaas O., Haavaldsen S., Pedersen B.
Archives of Biochemistry and Biophysics,
1963