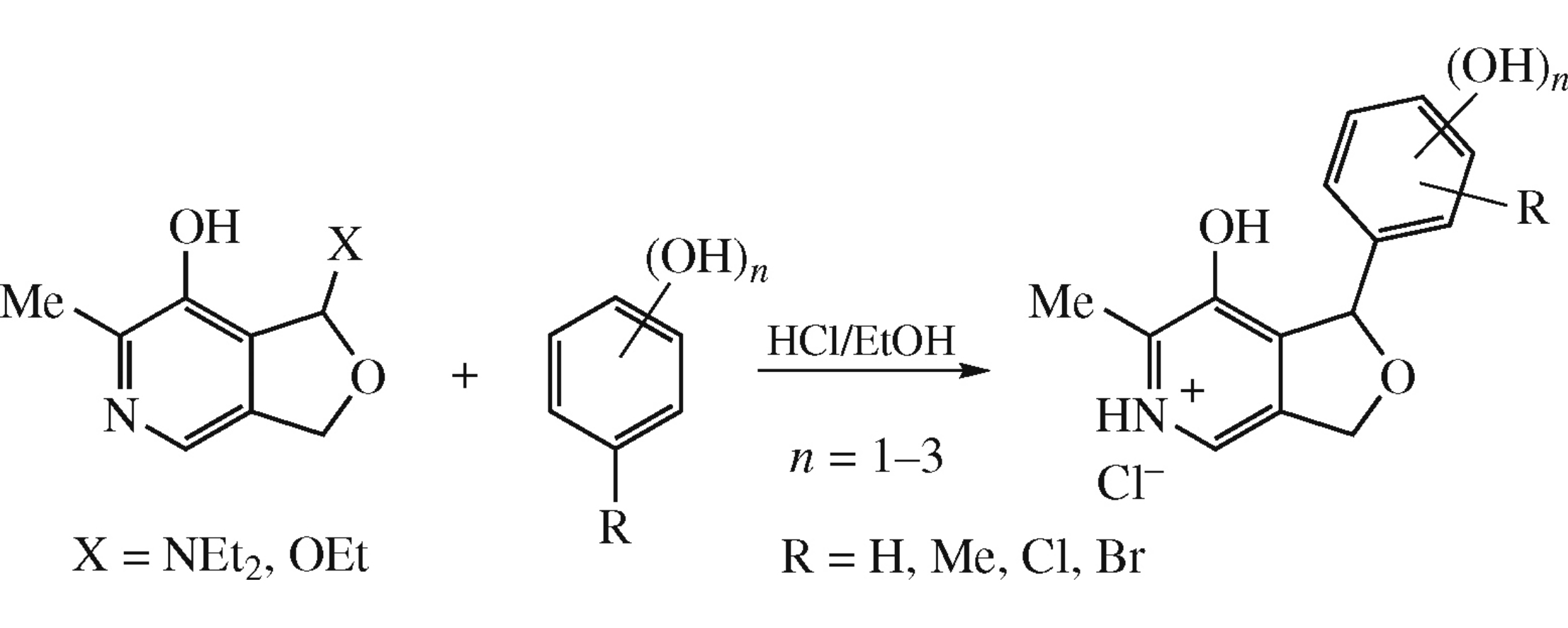
Abstract
The herein obtained 1-diethylamino-6-methyl-1,3-dihydrofuro[3,4-c]pyridin-7-ol or its 1-ethoxy analogue in their reactions with phenols or polyphenols undergo replacement of Et2N/OEt groups with (poly)hydroxyaryl moieties.
References
1.

Eliot A.C., Kirsch J.F.
Annual Review of Biochemistry,
2004
2.

Toney M.D.
Archives of Biochemistry and Biophysics,
2005
3.

Hayashi H., Mizuguchi H., Miyahara I., Islam M.M., Ikushiro H., Nakajima Y., Hirotsu K., Kagamiyama H.
Biochimica et Biophysica Acta - Proteins and Proteomics,
2003
4.

Watanabe A., Yoshimura T., Mikami B., Hayashi H., Kagamiyama H., Esaki N.
Journal of Biological Chemistry,
2002
5.

Major D.T., Gao J.
Journal of the American Chemical Society,
2006
6.

Miyashita K., Murafuji H., Iwaki H., Yoshioka E., Imanishi T.
Tetrahedron,
2003
7.

Jung K., Cho J., Lee J.S., Kim H.J., Kim Y.
Bioorganic and Medicinal Chemistry,
2013
8.

Brokamp R., Bergmann B., Müller I.B., Bienz S.
Bioorganic and Medicinal Chemistry,
2014
9.

Gazizov A.S., Kharitonova N.I., Smolobochkin A.V., Syakaev V.V., Burilov A.R., Pudovik M.A.
Monatshefte fur Chemie,
2015
10.
10.1016/j.mencom.2018.09.035_bib0050
Karjalainen
Anti-Cancer Agents Med. Chem.,
2017
11.

Smolobochkin A.V., Gazizov A.S., Burilov A.R., Pudovik M.A.
Russian Chemical Bulletin,
2016
12.

Bogdanov A.V., Zaripova I.F., Voloshina A.D., Strobykina A.S., Kulik N.V., Bukharov S.V., Mironov V.F.
Russian Journal of General Chemistry,
2018
13.
10.1016/j.mencom.2018.09.035_bib0065
Bagrov
J. Hypertens.,
2000
14.
A. Ali, I.K. Sebhat, C.L. Franklin, K.M. Rupprecht, R.K. Baker, R.P. Nargund, L. Yan, P. Huo, D. Shen, N. Almirante, S. Biondi, M. Ferrario and A. Nicotra, Patent US 8119806 B2, 2012.
15.

Garay R.P., Nazaret C., Diez J., Etienne A., Bourgain R., Braquet P., Esanu A.
Biochemical Pharmacology,
1984
16.
T. Miyata and K. Kurokawa, Patent EP 1693369 A1, 2006.
17.
Burilov A.R., Gazizov A.S., Volodina Y.M., Pudovik M.A., Habicher W.D., Bauer I., Gubaidullin A.T., Litvinova I.A., Konovalov A.I.
Mendeleev Communications,
2005
18.

Llor J., Sanchez-Ruiz J.M., Cortijo M.
Journal of the Chemical Society Perkin Transactions 2,
1988
19.
10.1016/j.mencom.2018.09.035_bib0095
Kibardina
Synthesis,
2015
20.
10.1016/j.mencom.2018.09.035_bib0100
Nakagawa
Agric. Biol. Chem.,
1977
21.

Kibardina L.K., Chumakov L.V., Trifonov A.V., Pudovik E.M., Burilov A.R., Pudovik M.A.
Russian Journal of General Chemistry,
2014
22.

NAGATA Y., YODA R., MATSUSHIMA Y.
Chemical and Pharmaceutical Bulletin,
2011
23.
10.1016/j.mencom.2018.09.035_bib0115
Sheldrick
SADABS,
1997
24.

Altomare A., Cascarano G., Giacovazzo C., Viterbo D.
Acta Crystallographica Section A Foundations of Crystallography,
1991
25.
G. M. Sheldrick, SHELX-97. Programs for Crystal Structure Analysis (Release 97-2), University of Göttingen, 1997.
26.

Farrugia L.J.
Journal of Applied Crystallography,
1999
27.
APEX2 (Version 2.1), SAINTPlus. Data Reduction and Correction Program (Version 7.31A), Bruker Advanced X-ray Solutions, Bruker AXS Inc., Madison, WI, 2006.