Abstract
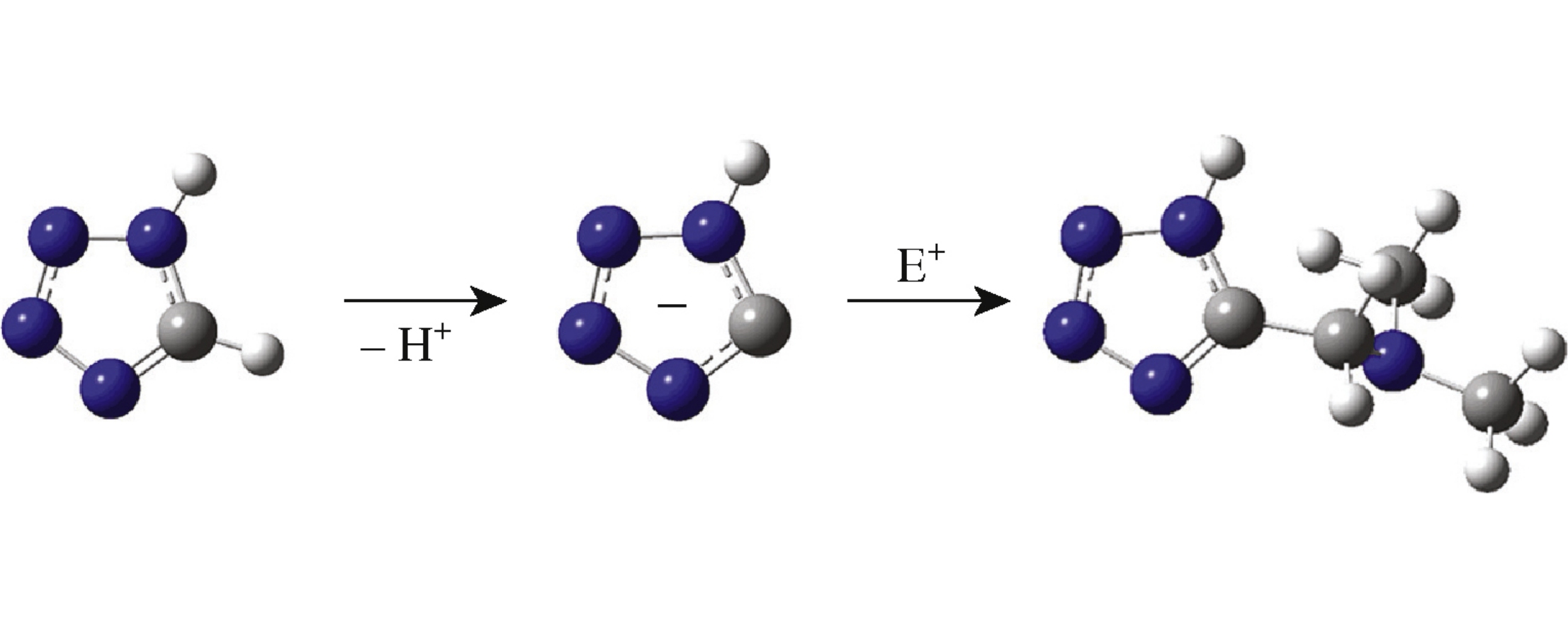
Based on analysis of quantum chemical DFT/B3LYP/6-31G(d) calculations results, thermodynamic characteristics of electrophilic dimethylaminomethylation of 1H-tetrazole and 1-methyl-tetrazole according to addition–elimination and elimination–addition schemes were correlated. The possibility of the dimethylaminomethylation without the preliminary formation of N-protonated azolium salts was demonstrated.
References
1.

Belen’kii L.I., Chuvylkin N.D.
Russian Chemical Bulletin,
2016
2.

Clementi S., Forsythe P.P., Johnson C.D., Katritzky A.R., Terem B.
Journal of the Chemical Society Perkin Transactions 2,
1974
3.

El-Anani A., Banger J., Bianchi G., Clementi S., Johnson C.D., Katritzky A.R.
Journal of the Chemical Society Perkin Transactions 2,
1973
4.

Clementi S., Forsythe P.P., Johnson C.D., Katritzky A.R.
Journal of the Chemical Society Perkin Transactions 2,
1973
5.

Katritzky A.R., Terem B., Scriven E.V., Clementi S., Tarhan H.O.
Journal of the Chemical Society Perkin Transactions 2,
1975
6.

Katritzky A.R., Clementi S., Tarhan H.O.
Journal of the Chemical Society Perkin Transactions 2,
1975
7.

Katritzky A.R., Ögretir C., Tarhan H.O., Dou H.M., Metzger J.V.
Journal of the Chemical Society Perkin Transactions 2,
1975
8.

Katritzky A.R., Tarhan H.O., Terem B.
Journal of the Chemical Society Perkin Transactions 2,
1975
9.

Belen'kii L.I.
Chemistry of Heterocyclic Compounds,
1986
10.

Belen'kii L.I., Chuvylkin N.D.
Chemistry of Heterocyclic Compounds,
1997
11.
10.1016/j.mencom.2018.09.034_bib0055
Gaponik
Chemical Problems of the Development of New Materials and Technologies,
2003
12.

Belen’kii L.I., Subbotin A.N., Chuvylkin N.D.
Russian Chemical Bulletin,
2015
13.

Belen’kii L.I., Subbotin A.N., Chuvylkin N.D.
Russian Chemical Bulletin,
2015
14.

Chuvylkin N.D., Subbotin A.N., Belen´kii L.I.
Russian Chemical Bulletin,
2016
15.

Chuvylkin N.D., Subbotin A.N., Belen’kii L.I.
Russian Chemical Bulletin,
2016
16.

Belen’kii L.I., Subbotin A.N., Chuvylkin N.D.
Russian Chemical Bulletin,
2016
17.
10.1016/j.mencom.2018.09.034_bib0085
Ostrovskii
2008
18.

Karavai V.P., Gaponik P.N.
Chemistry of Heterocyclic Compounds,
1991
19.
10.1016/j.mencom.2018.09.034_bib0095
Butler
1996
20.
10.1016/j.mencom.2018.09.034_bib0100
Frisch
Gaussian 09. Revision D. 01,
2013
21.

Belen’kii L.I., Nesterov I.D., Chuvylkin N.D.
Russian Chemical Bulletin,
2009
22.
10.1016/j.mencom.2018.09.034_bib0110
Belen’kii
Russ. Chem. Bull., Int. Ed.,
2016
23.
10.1016/j.mencom.2018.09.034_bib0115
Terentiev
1957
24.
10.1016/j.mencom.2018.09.034_bib0120
Furin
Sovremennye metody ftorirovaniya organicheskih soedinenii (Modern Methods of Fluorination of Organic Compounds),
2000
25.

Voskressensky L., Golantsov N., Maharramov A.
Synthesis,
2016
26.

Mercurierung des Anthranilsäure‐esters und seiner Alkyl‐Derivate. Ein Beitrag zur Substitutionslehre
Schoeller W., Hueter R.
Berichte der deutschen chemischen Gesellschaft,
1914
27.
10.1016/j.mencom.2018.09.034_bib0135
Schoeller
Ber.,
1919
28.
10.1016/j.mencom.2018.09.034_bib0140
Makarova
Metody elementoorganicheskoi khimii. Rtut (Methods of Organoelement Chemistry. Mercury),
1965
29.
Ananikov V.P., Galkin K.I., Egorov M.P., Sakharov A.M., Zlotin S.G., Redina E.A., Isaeva V.I., Kustov L.M., Gening M.L., Nifantiev N.E.
Mendeleev Communications,
2016