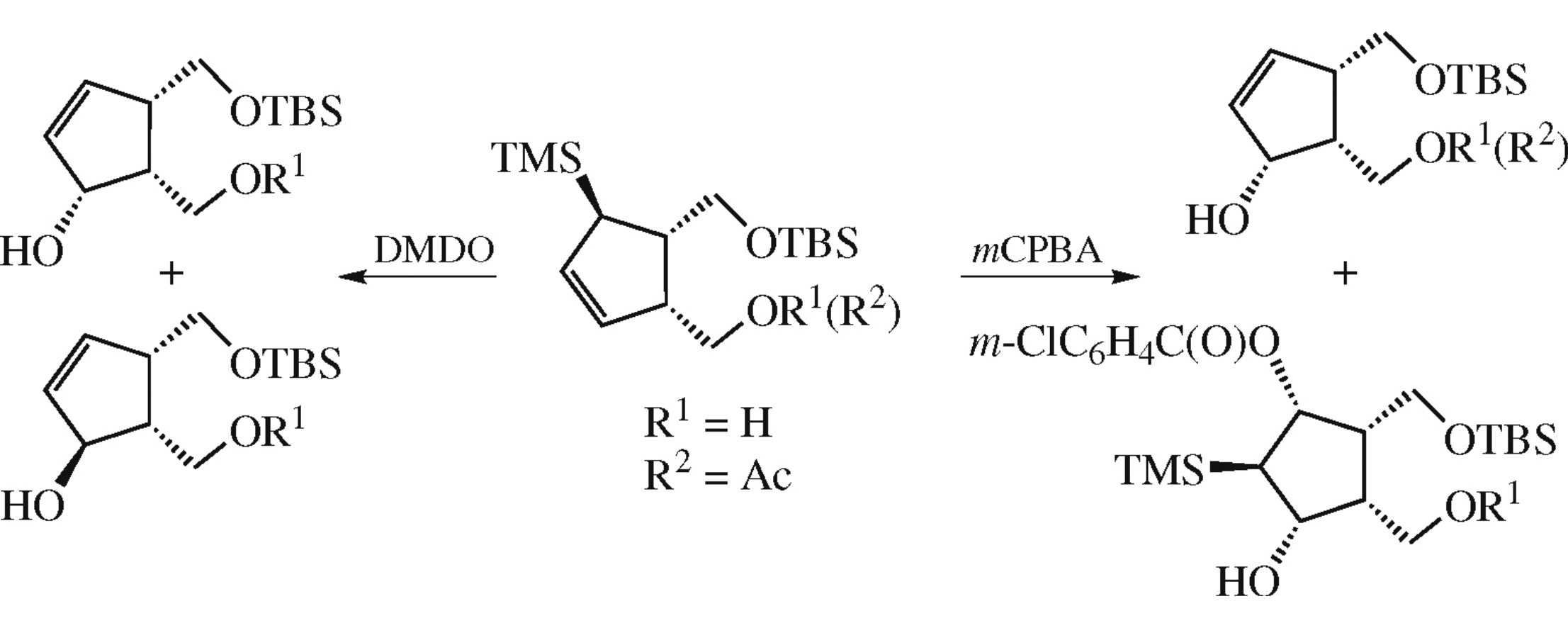
Abstract
Oxidations of {(1R,4R,5S)-2-[tert-butyl(dimethyl)silyloxy-methyl]-4-(trimethylsilyl)cyclopent-2-en-1-yl}methanol with m-chloroperoxybenzoic acid (mCPBA) and dimethyldioxirane were studied. In the case of mCPBA, an allylicic alcohol expected according to the protodesilylation mechanism and an anomalous product of 1,2-migration of the Me3Si group were obtained. The latter was formed due to the coordination and directing effects of the free OH group.
References
1.

Fleming I., Barbero A., Walter D.
Chemical Reviews,
1997
2.

Chabaud L., James P., Landais Y.
European Journal of Organic Chemistry,
2004
3.
![Some aspects of intramolecular carbocyclization of methyl (2E)-3-[(1S,2R,5R)-2-({[tert-butyl(dimethyl)-silyl]oxy}methyl)-5-(trimethylsilyl)cyclopent-3-en-1-yl]prop-2-enoate and its derivatives](/storage/images/resized/oZgeErrVFhuDksyqFURLvYS1wtVSBWczh001igGo_small_thumb.webp)
Gimazetdinov A.M., Al’mukhametov A.Z., Spirikhin L.V., Miftakhov M.S.
Russian Journal of Organic Chemistry,
2017
4.

Masse C.E., Panek J.S.
Chemical Reviews,
1995
5.

Hayashi T., Okamoto Y., Kabeta K., Hagihara T., Kumada M.
Journal of Organic Chemistry,
1984
6.

Lambert J.B., Zhao Y., Emblidge R.W., Salvador L.A., Liu X., So J., Chelius E.C.
Accounts of Chemical Research,
1998