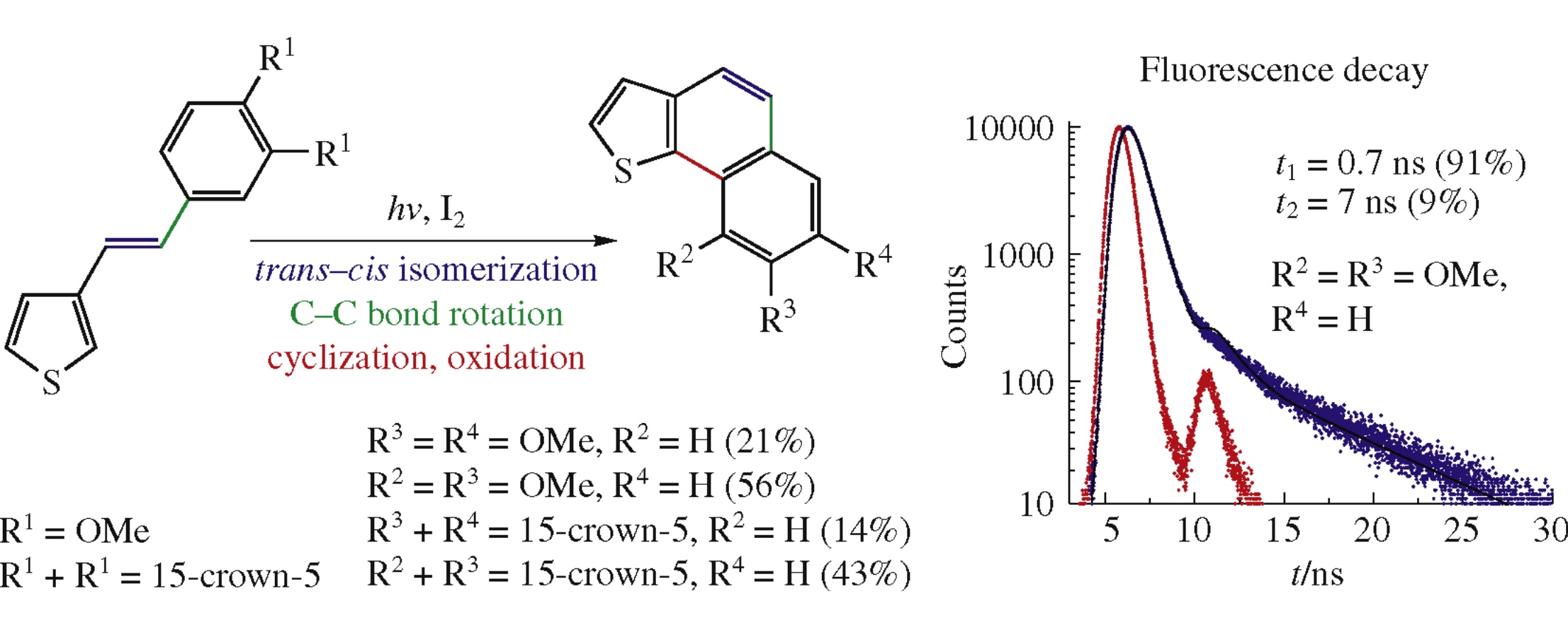
Abstract
Photochemical oxidative cyclization of 3-[(E)-2-(3,4-di-methoxyphenyl)vinyl]thiophene and its 15-crown-5-analogue (15-[(E)-2-(3-thienyl)vinyl]-2,3,5,6,8,9,11,12-octahydro-1,4,7,10,13-benzopentaoxacyclopentadecine) affords the isomeric thiophene-containing fused aromatic compounds demonstrating photophysical properties different from those of initial styryl derivatives. E-Configuration of the initial styryl dye, 3-[(E)-2-(3,4-dimethoxyphenyl)vinyl]thiophene, has been proved by X-ray analysis.
References
1.
10.1016/j.mencom.2018.09.032_bib0005
Fichou
Handbook of Oligo- and Polythiophenes,
1999
2.

Wang C., Dong H., Hu W., Liu Y., Zhu D.
Chemical Reviews,
2011
3.
Nosova E.V., Moshkina T.N., Lipunova G.N., Baklanova I.V., Kopchuk D.S., Slepukhin P.A., Charushin V.N.
Mendeleev Communications,
2018
4.

Baudoin O.
Accounts of Chemical Research,
2017
5.

Shi X., Soulé J., Doucet H.
Journal of Organic Chemistry,
2017
6.
10.1016/j.mencom.2018.09.032_bib0030
Handbook of Synthetic Photochemistry,
2010
7.

Bach T., Hehn J.P.
Angewandte Chemie - International Edition,
2011
8.

Hoffmann N.
Chemical Reviews,
2008
9.

Jørgensen K.B.
Molecules,
2010
10.

Lewis F.D., Bedell A.M., Dykstra R.E., Elbert J.E., Gould I.R., Farid S.
Journal of the American Chemical Society,
1990
11.

Tian H., Yang S.
Chemical Society Reviews,
2004
12.
10.1016/j.mencom.2018.09.032_bib0060
Irie
Phororeactive Materials for Ultrahigh-Density Optical Memory,
1993
13.

Galangau O., Nakashima T., Maurel F., Kawai T.
Chemistry - A European Journal,
2015
14.
![General Photoinduced Sequential Electrocyclization/[1,9]-Sigmatropic Rearrangement/Ring-Opening Reaction of Diarylethenes](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Lvov A.G., Shirinian V.Z., Zakharov A.V., Krayushkin M.M., Kachala V.V., Zavarzin I.V.
Journal of Organic Chemistry,
2015
15.

Ho T., Wu J., Wang S.
Angewandte Chemie - International Edition,
1999
16.

Lvov A.G., Shirinyan V.Z.
Chemistry of Heterocyclic Compounds,
2016
17.

Ho J., Ho T., Liu R.S.
Organic Letters,
2001
18.

Carruthers W., Stewart H.N.
Tetrahedron Letters,
1965
19.

Thermally irreversible photochromic systems. Reversible photocyclization of diarylethene derivatives
Irie M., Mohri M.
Journal of Organic Chemistry,
1988
20.

Song K., Peng M., Xu M., Wu L., Zhang L., Tung C.
Tetrahedron Letters,
2002
21.
![The synthesis of naphtho[1,2-b]thiophene and all of the eight isomers of monomethylnaphtho[1,2-b]thiophene](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Tedjamulia M.L., Stuart J.G., Tominaga Y., Castle R.N., Lee M.L.
Journal of Heterocyclic Chemistry,
1984
22.
Lukovskaya E.V., Sotnikova Y.A., Bobyleva A.A., Anisimov A.V., Fedorov Y.V., Moiseeva A.A., Dolganov A.V., Fedorova O.A.
Mendeleev Communications,
2016
23.

Sankar E., Raju P., Karunakaran J., Mohanakrishnan A.K.
Journal of Organic Chemistry,
2017
24.
10.1016/j.mencom.2018.09.032_bib0120
SAINT Data Reduction and Correction Program, Version 8. 34A,
2013
25.
10.1016/j.mencom.2018.09.032_bib0125
Sheldrick
SADABS-2014/5, Bruker/Siemens Area Detector Absorption Correction Program,
2014
26.

Sheldrick G.M.
Acta Crystallographica Section A Foundations of Crystallography,
2007