Abstract
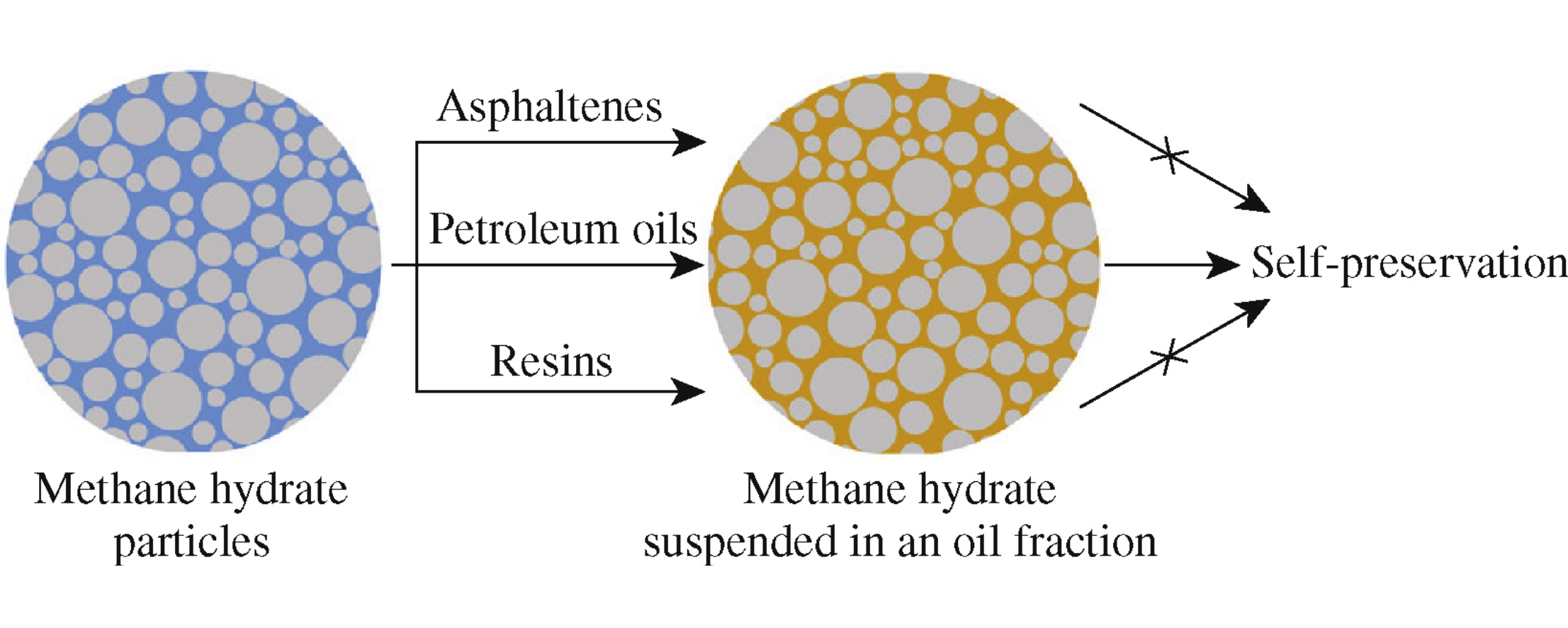
The dissociation of methane hydrate doped with a petroleum fraction (resins, asphaltenes, or oils) by its cryogenic mixing with pure hydrate particles has been studied at temperatures below 0°C. An oil fraction of petroleum (a mixture of saturated, aromatic and polar compounds) was close to crude oil in terms of efficiency with respect to the self-preservation of methane hydrate. The effect of asphaltenes on hydrate dissociation was negligible, whereas resinous substances occupied an intermediate position between the fractions of oils and asphaltenes in the degree of influence.
References
1.
10.1016/j.mencom.2018.09.028_bib0005
Yakushev
Physics and Chemistry of Ice,
1992
2.
10.1016/j.mencom.2018.09.028_bib0010
Istomin
Gas Ind. Russ.,
2006
3.

Handa Y.P.
Journal of Chemical Thermodynamics,
1986
4.
10.1016/j.mencom.2018.09.028_bib0020
Ershov
Dokl. Akad. Nauk SSSR,
1991
5.

Stern L.A., Circone S., Kirby S.H., Durham W.B.
Journal of Physical Chemistry B,
2001
6.

Melnikov V.P., Nesterov A.N., Reshetnikov A.M., Zavodovsky A.G.
Chemical Engineering Science,
2009
7.

Melnikov V.P., Nesterov A.N., Podenko L.S., Reshetnikov A.M., Shalamov V.V.
Chemical Engineering Science,
2012
8.

Takeya S., Uchida T., Nagao J., Ohmura R., Shimada W., Kamata Y., Ebinuma T., Narita H.
Chemical Engineering Science,
2005
9.

Mimachi H., Takeya S., Yoneyama A., Hyodo K., Takeda T., Gotoh Y., Murayama T.
Chemical Engineering Science,
2014
10.

Takeya S., Ripmeester J.
Angewandte Chemie - International Edition,
2008
11.

Falenty A., Kuhs W.F., Glockzin M., Rehder G.
Energy & Fuels,
2014
12.
Song M.H., Kim H.S., Kim B.M.
Journal of Mechanical Science and Technology,
2015
13.

Takeya S., Fujihisa H., Gotoh Y., Istomin V., Chuvilin E., Sakagami H., Hachikubo A.
Journal of Physical Chemistry C,
2013
14.
Stoporev A.S., Manakov A.Y., Altunina L.K., Bogoslovsky A.V.
Mendeleev Communications,
2012
15.

Stoporev A.S., Manakov A.Y., Altunina L.K., Strelets L.A.
Energy & Fuels,
2016
16.
Dvoyashkin N.K., Filippov A.
Mendeleev Communications,
2018
17.

Lv Y., Jia M., Chen J., Sun C., Gong J., Chen G., Liu B., Ren N., Guo S., Li Q.
Energy & Fuels,
2015
18.

Sato T., Takeya S., Nagashima H.D., Ohmura R.
Journal of Food Engineering,
2014
19.
10.1016/j.mencom.2018.09.028_bib0095
Bernstein
Polymorphism in Molecular Crystals,
2002
20.
Koroleva M.Y., Tokarev A.M., Yurtov E.V.
Mendeleev Communications,
2017
21.

Chen X., Song X., Huang J., Wu C., Ma D., Tian M., Jiang H., Huang P.
Energy & Fuels,
2017
22.
Talagaeva N.V., Zolotukhina E.V., Pisareva P.A., Vorotyntsev M.A.
Mendeleev Communications,
2016
23.
Urakaev F.K., Abuyeva B.B., Vorobyeva N.A., Mun G.A., Uralbekov B.M., Burkitbayev M.M.
Mendeleev Communications,
2018
24.
H.M. Weiss, A. Wilhelms, N. Mills, J. Scotchmer, P.B. Hall, K. Lind, T. Brekke, NIGOGA–The Norwegian Industry Guide to Organic Geochemical Analyses, 4th edn., Norsk Hydro, Statoil, Geolab Nor, SINTEF Petroleum Research and the Norwegian Petroleum Directorate, 2000.
25.

Ogienko A.G., Kurnosov A.V., Manakov A.Y., Larionov E.G., Ancharov A.I., Sheromov M.A., Nesterov A.N.
Journal of Physical Chemistry B,
2006