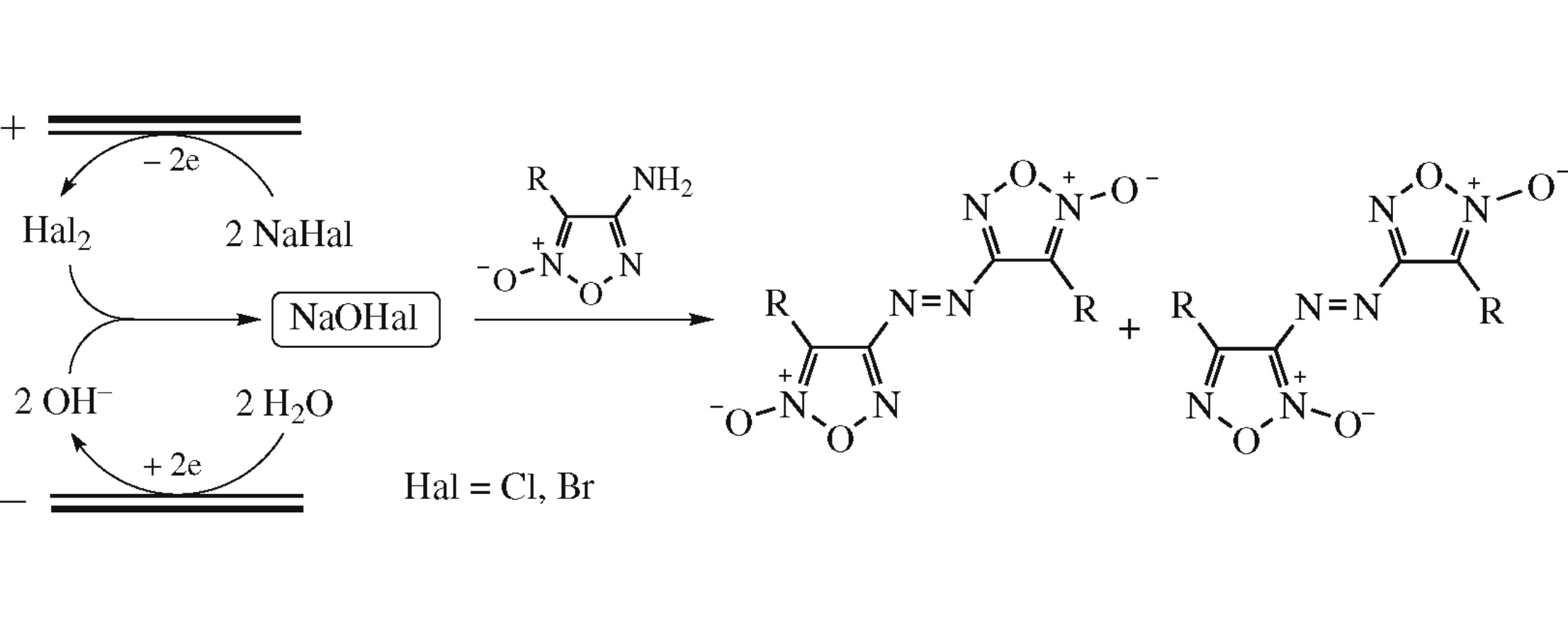
Abstract
A new eco-friendly synthesis of azofuroxans comprises the oxidative N–N coupling of aminofuroxans under the action of electrogenerated NaOCl and NaOBr. In addition, an oxidative isomerization of the aminofuroxan motif was found for the first time, which resulted in a formation of unsymmetrical azofuroxans.
References
1.
Cena C., Bertinaria M., Boschi D., Giorgis M., Gasco A.
Arkivoc,
2006
2.

Fershtat L.L., Makhova N.N.
ChemMedChem,
2017
3.

Makhova N.N., Rakitin O.A.
Chemistry of Heterocyclic Compounds,
2017
4.
Ustyuzhanina N.E., Fershtat L.L., Gening M.L., Nifantiev N.E., Makhova N.N.
Mendeleev Communications,
2016
5.
Ustyuzhanina N.E., Fershtat L.L., Gening M.L., Nifantiev N.E., Makhova N.N.
Mendeleev Communications,
2018
6.
Kulikov A.S., Larin A.A., Fershtat L.L., Anikina L.V., Pukhov S.A., Klochkov S.G., Struchkova M.I., Romanova A.A., Ananyev I.V., Makhova N.N.
Arkivoc,
2017
7.
Annaratone L., Guglielmo S., Chegaev K.Y.
Mendeleev Communications,
2017
8.
Starosotnikov A.M., Bastrakov M.A., Pavlov A.A., Fedyanin I.V., Dalinger I.L., Shevelev S.A.
Mendeleev Communications,
2016
9.

Fershtat L.L., Makhova N.N.
Russian Chemical Reviews,
2016
10.
Zlotin S.G., Churakov A.M., Dalinger I.L., Luk’yanov O.A., Makhova N.N., Sukhorukov A.Y., Tartakovsky V.A.
Mendeleev Communications,
2017
11.
![Ionic liquid-promoted [3+2]-cycloaddition reactions of nitroformonitrile oxide generated by the cycloreversion of dinitrofuroxan](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Fershtat L.L., Ovchinnikov I.V., Makhova N.N.
Tetrahedron Letters,
2014
12.

Makhova N.N., Kulikov A.S.
Russian Chemical Reviews,
2013
13.

Kuchurov I.V., Zharkov M.N., Fershtat L.L., Makhova N.N., Zlotin S.G.
ChemSusChem,
2017
14.

Fershtat L.L., Ovchinnikov I.V., Epishina M.A., Romanova A.A., Lempert D.B., Muravyev N.V., Makhova N.N.
ChemPlusChem,
2017
15.
![An efficient access to (1H-tetrazol-5-yl)furoxan ammonium salts via a two-step dehydration/[3+2]-cycloaddition strategy](/storage/images/resized/GDnYOu1UpMMfMMRV6Aqle4H0YLLsraeD9IP9qScG_small_thumb.webp)
Fershtat L.L., Epishina M.A., Kulikov A.S., Ovchinnikov I.V., Ananyev I.V., Makhova N.N.
Tetrahedron,
2015
16.

Guo T., Liu M., Huang X., Wang Z., Qiu S., Ge Z., Meng Z.
Journal of Analytical and Applied Pyrolysis,
2017
17.
10.1016/j.mencom.2018.09.023_sbref0020b
Ovchinnikov
Dokl. Chem.,
1998
18.

Sheremetev A.B., Lyalin B.V., Kozeev A.M., Palysaeva N.V., Struchkova M.I., Suponitsky K.Y.
RSC Advances,
2015
19.
Lyalin B.V., Sigacheva V.L., Kokorekin V.A., Petrosyan V.A.
Mendeleev Communications,
2015
20.
Lyalin B.V., Sigacheva V.L., Kokorekin V.A., Petrosyan V.A.
Arkivoc,
2017
21.

Ungnade H.E., Kissinger L.W.
Tetrahedron,
1963
22.

Epishina M.A., Kulikov A.S., Makhova N.N.
Russian Chemical Bulletin,
2008
23.

Ovchinnikov I.V., Kulikov A.S., Makhova N.N., Tosco P., Di Stilo A., Fruttero R., Gasco A.
Il Farmaco,
2003
24.
Blinnikov A.N., Makhova N.N., Khmel’nitskii L.I.
Mendeleev Communications,
1999
25.

Petrosyan V.A., Niyazymbetov M.E., Kozlovskaya �.V.
Russian Chemical Bulletin,
1986
26.

Salaheldin A.M., Oliveira-Campos A.M., Rodrigues L.M.
Tetrahedron Letters,
2007
27.

Kaulen J., Schäfer H.
Tetrahedron,
1982