Abstract
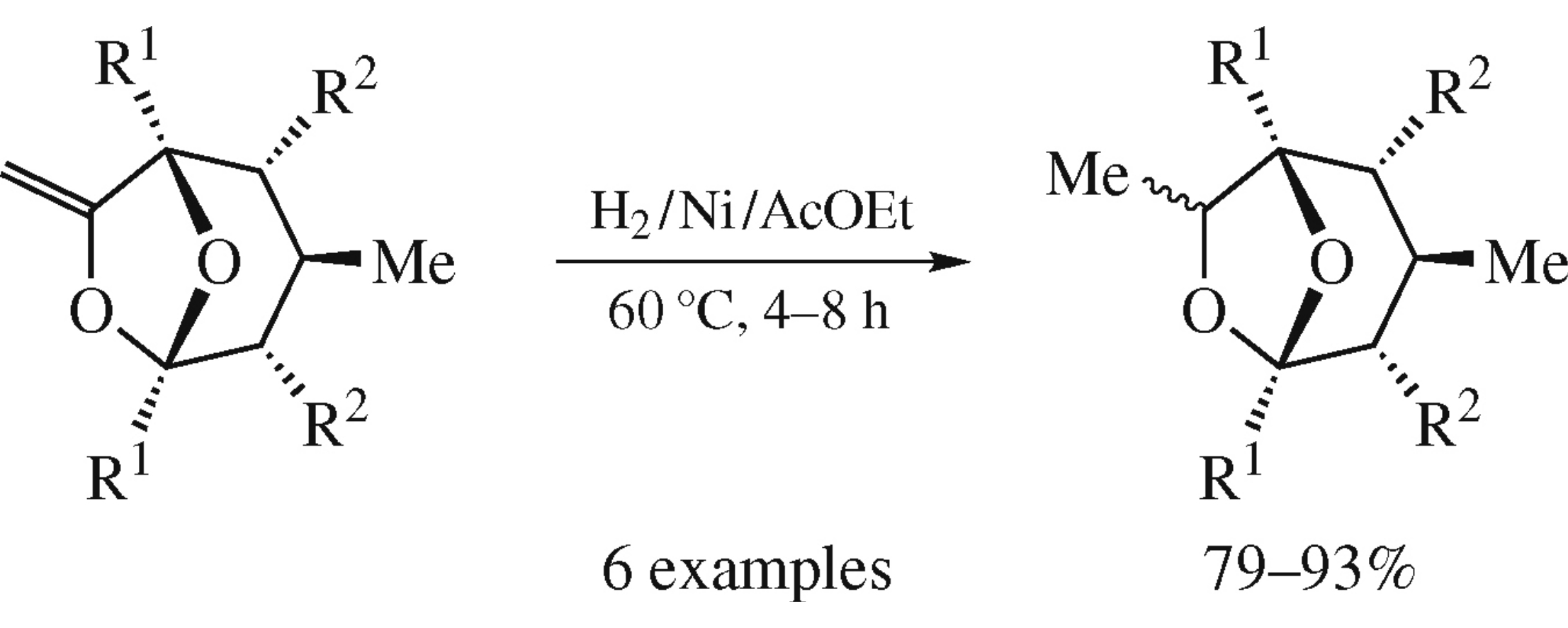
Nickel-catalyzed low pressure hydrogenation of 1,5-diaryl-7-methylidene-6,8-dioxabicyclo[3.2.1]octanes proceeds chemo-specifically at the double bond and brings about mostly 1R*,3R*,5S*,7R* diastereomers.
References
1.

2.

Greenwood D.R., Comeskey D., Hunt M.B., Rasmussen L.E.
Nature,
2005
3.

Ramos V., Vasconcelos V.
Marine Drugs,
2010
4.

Bjorklund M., Jun J.G., Mundy B.P.
Tetrahedron Letters,
1985
5.
10.1016/j.mencom.2018.09.021_bib0025
Jun
Bull. Korean Chem. Soc.,
1992
6.

Jun J., Shun Shin H.
Tetrahedron Letters,
1992
7.

Schmidt E.Y., Trofimov B.A., Zorina N.V., Mikhaleva A.I., Ushakov I.A., Skital'tseva E.V., Kazheva O.N., Alexandrov G.G., Dyachenko O.A.
European Journal of Organic Chemistry,
2010
8.

Zhang W., Tong R.
Journal of Organic Chemistry,
2016
9.
![One-Pot Assembly of 7-Methylene-6,8-dioxabicyclo[3.2.1]octanes, Congeners of Frontalin, from Ketones and Acetylene](/storage/images/resized/bRyGpdm98BkAUYiK1YFNpl5Z7hPu6Gd87gbIeuG3_small_thumb.webp)
Trofimov B.A., Schmidt E.Y., Ushakov I.A., Mikhaleva A.I., Zorina N.V., Protsuk N.I., Senotrusova E.Y., Skital'tseva E.V., Kazheva O.N., Alexandrov G.G., Dyachenko O.A.
European Journal of Organic Chemistry,
2009
10.
Schmidt E.Y., Tatarinova I.V., Ushakov I.A., Trofimov B.A.
Mendeleev Communications,
2018
11.

Schmidt E.Y., Bidusenko I.A., Ushakov I.A., Vashchenko A.V., Trofimov B.A.
Organic Letters,
2017
12.
Schmidt E.Y., Tatarinova I.V., Protsuk N.I., Ushakov I.A., Trofimov B.A.
Mendeleev Communications,
2018
13.

Gozlan C., Lafon R., Duguet N., Redl A., Lemaire M.
RSC Advances,
2014
14.

Gozlan C., Deruer E., Duclos M., Molinier V., Aubry J., Redl A., Duguet N., Lemaire M.
Green Chemistry,
2016
15.

Nakata M., Osumi T., Ueno A., Kimura T., Tamai T., Tatsuta K.
Tetrahedron Letters,
1991
16.

Vincent A., Prunet J.
Tetrahedron Letters,
2006
17.
![Hydrogenolysis of the acetal 6,8-dioxabicyclo[3.2.1]octane by aluminum chloride hydride. Evidence for the preferred direction of ring cleavage in the course of .alpha.-bromination of this acetal](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Clasper P., Brown R.K.
Journal of Organic Chemistry,
1972
18.
![A New Polymerization System for Bicyclic Acetals: Toward the Controlled/“Living” Cationic Ring-Opening Polymerization of 6,8-Dioxabicyclo[3.2.1]octane](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Torres L.F., Patten T.E.
Macromolecules,
1999
19.

Liu K., Zhou H., Wu Y., Yao Z.
Journal of Organic Chemistry,
2003