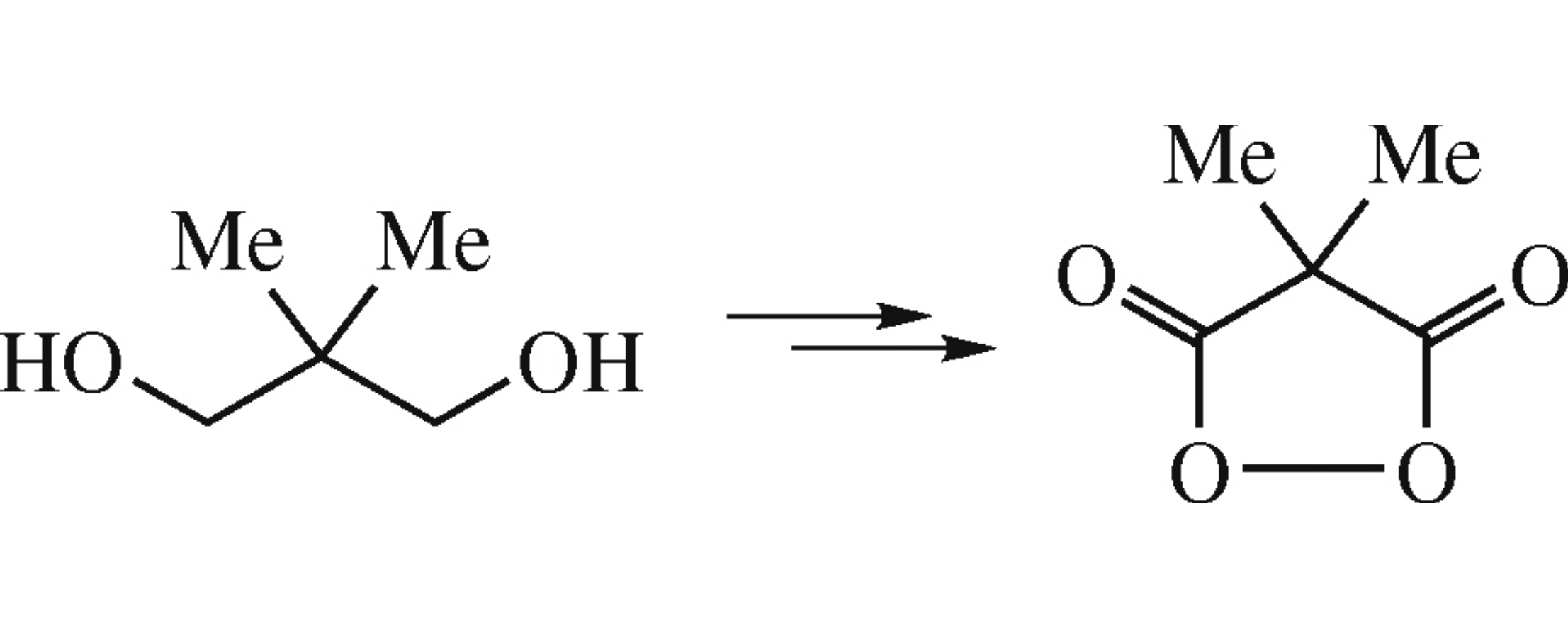
Abstract
Easily available dimethylmalonoyl peroxide is proposed as a reactive alternative to the widely known spirocyclopropyl analogue for the use in the arene acyloxylation and other oxidative reactions.
References
1.

Adam W., Rucktaeschel R.
Journal of the American Chemical Society,
1971
2.

Griffith J.C., Jones K.M., Picon S., Rawling M.J., Kariuki B.M., Campbell M., Tomkinson N.C.
Journal of the American Chemical Society,
2010
3.

Schwarz M., Reiser O.
Angewandte Chemie - International Edition,
2011
4.

Rawling M.J., Tomkinson N.C.
Organic and Biomolecular Chemistry,
2013
5.

Terent’ev A., Khodykin S., Troitskii N., Ogibin Y., Nikishin G.
Synthesis,
2004
6.

Terent’ev A.O., Khodykin S.V., Krylov I.B., Ogibin Y.N., Nikishin G.I.
Synthesis,
2006
7.

Gomes G.D., Vil' V., Terent'ev A., Alabugin I.V.
Chemical Science,
2015
8.

Alamillo-Ferrer C., Davidson S.C., Rawling M.J., Theodoulou N.H., Campbell M., Humphreys P.G., Kennedy A.R., Tomkinson N.C.
Organic Letters,
2015
9.

Alamillo-Ferrer C., Karabourniotis-Sotti M., Kennedy A.R., Campbell M., Tomkinson N.C.
Organic Letters,
2016
10.

Yuan C., Liang Y., Hernandez T., Berriochoa A., Houk K.N., Siegel D.
Nature,
2013
11.

Yuan C., Eliasen A.M., Camelio A.M., Siegel D.
Nature Protocols,
2014
12.

Dragan A., Kubczyk T.M., Rowley J.H., Sproules S., Tomkinson N.C.
Organic Letters,
2015
13.

Camelio A.M., Liang Y., Eliasen A.M., Johnson T.C., Yuan C., Schuppe A.W., Houk K.N., Siegel D.
Journal of Organic Chemistry,
2015
14.

Terent’ev A., Vil’ V., Nikishin G., Adam W.
Synlett,
2015
15.

Terent’ev A.O., Vil’ V.A., Gorlov E.S., Nikishin G.I., Pivnitsky K.K., Adam W.
Journal of Organic Chemistry,
2016
16.

Bityukov O.V., Vil’ V.A., Merkulova V.M., Nikishin G.I., Terent’ev A.O.
Pure and Applied Chemistry,
2017
17.

Eliasen A.M., Christy M., Claussen K.R., Besandre R., Thedford R.P., Siegel D.
Organic Letters,
2015
18.

Gan S., Yin J., Yao Y., Liu Y., Chang D., Zhu D., Shi L.
Organic and Biomolecular Chemistry,
2017
19.
![[3 + 2] Cycloadditions of Azides with Arynes via Photolysis of Phthaloyl Peroxide Derivatives](/storage/images/resized/iLiQsFqFaSEx6chlGQ5fbAwF6VYU3WWa08hkss0g_small_thumb.webp)
Chang D., Zhu D., Shi L.
Journal of Organic Chemistry,
2015
20.
Lapitskaya M.A., Vil’ V.A., Daeva E.D., Terent’ev A.O., Pivnitsky K.K.
Mendeleev Communications,
2016
21.
Lapitskaya M.A., Vil’ V.A., Vasil’eva L.L., Daeva E.D., Terent’ev A.O., Pivnitsky K.K.
Mendeleev Communications,
2017
22.

Jones K.M., Tomkinson N.C.
Journal of Organic Chemistry,
2012
23.

Martin M.M., Hammer F.T., Zador E.
Journal of Organic Chemistry,
1973
24.

Chapman O.L., Wojtkowski P.W., Adam W., Rodriguez O., Rucktaeschel R.
Journal of the American Chemical Society,
1972
25.
Adam W., Epe B., Schiffmann D., Vargas F., Wild D.
1988
26.

Adam W., Hückmann S., Vargas F.
Tetrahedron Letters,
1989
27.
Terent’ev A.O., Vil’ V.A., Mulina O.M., Pivnitsky K.K., Nikishin G.I.
Mendeleev Communications,
2014
28.

Evans A.R., Martin R., Taylor G.A., Yap C.H.
Journal of the Chemical Society Perkin Transactions 1,
1987