Abstract
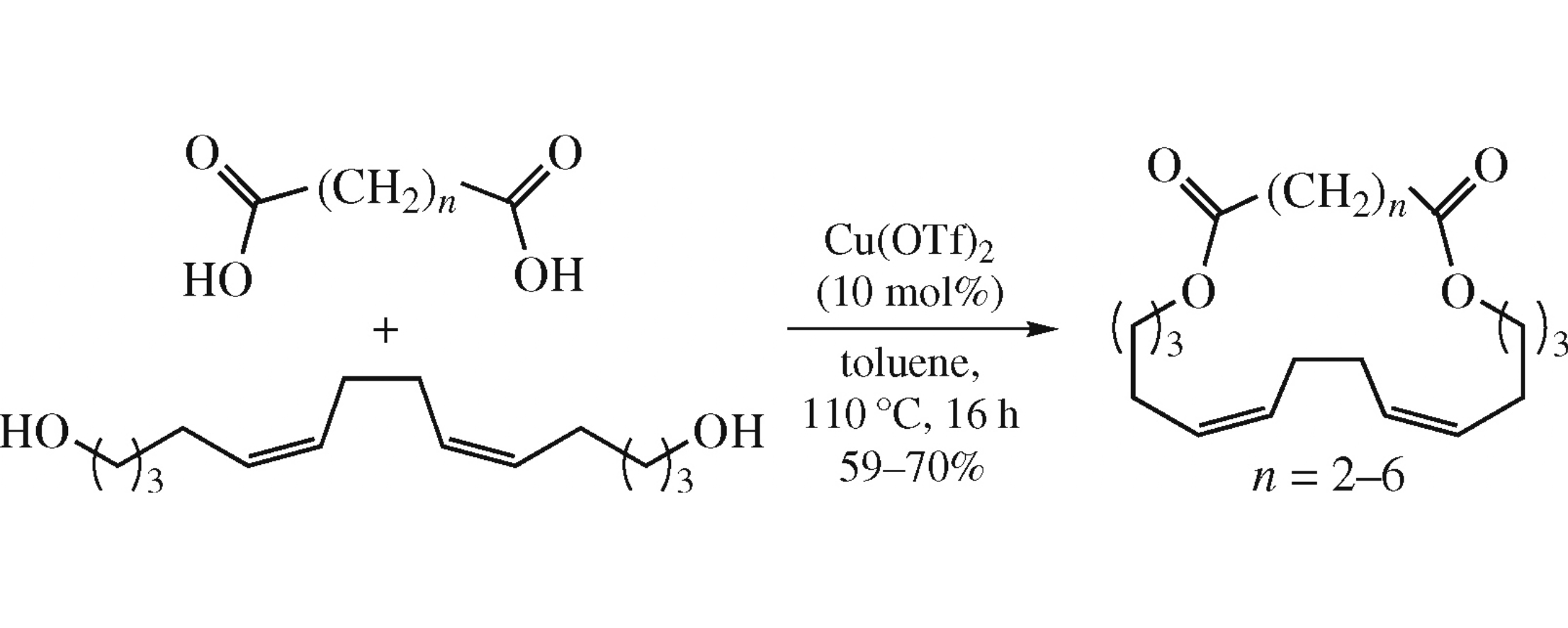
Macrodiolides containing a 1Z,5Z-diene moiety were obtained with high selectivity (>98%) and in good yields (59–70%) by the Cu(OTf)2-catalyzed intermolecular esterification of aliphatic α,ω-dicarboxylic acids with tetradeca-5Z,9Z-diene- 1,14-diol. This diol was accessed by homocyclomagnesiation of hepta-5,6-dien-1-ol tetrahydropyranyl ether using EtMgBr/Mg reagent system and Cp2TiCl2 catalyst.
References
1.

Driggers E.M., Hale S.P., Lee J., Terrett N.K.
Nature Reviews Drug Discovery,
2008
2.

Yudin A.K.
Chemical Science,
2015
3.
10.1016/j.mencom.2018.09.017_bib0015
Macrocyclic Lactones in Antiparasitic Therapy,
2002
4.
10.1016/j.mencom.2018.09.017_bib0020
Natural Lactones and Lactams: Synthesis, Occurrence and Biological Activity,
2013
5.

Shushni M.A., Singh R., Mentel R., Lindequist U.
Marine Drugs,
2011
6.

Spinella A., Caruso T., Martino M., Sessa C.
Synlett,
2001
7.

Bruno G., Caruso T., Peluso A., Spinella A.
Phytochemistry Reviews,
2004
8.

Ojika M., Kigoshi H., Suenaga K., Imamura Y., Yoshikawa K., Ishigaki T., Sakakura A., Mutou T., Yamada K.
Tetrahedron,
2012
9.

Hosoe T., Fukushima K., Takizawa K., Itabashi T., Kawahara N., Vidotto V., Kawai K.
Journal of Antibiotics,
2006
10.

Tonoi T., Kawahara R., Yoshinaga Y., Inohana T., Fujimori K., Shiina I.
Tetrahedron Letters,
2015
11.

Carballeira N.M.
Progress in Lipid Research,
2008
12.

Carballeira N.M., Betancourt J.E., Orellano E.A., González F.A.
Journal of Natural Products,
2002
13.

Carballeira N.M., Montano N., Amador L.A., Rodríguez A.D., Golovko M.Y., Golovko S.A., Reguera R.M., Álvarez-Velilla R., Balaña-Fouce R.
Lipids,
2015
14.

Nemoto T., Yoshino G., Ojika M., Sakagami Y.
Tetrahedron,
1997
15.

Chen S., Hsu C., Li C., Lu J., Chuang L.
Food Chemistry,
2011
16.

Xie K., Miles E.A., Calder P.C.
Journal of Functional Foods,
2016
17.
D’yakonov V.A., Makarov A.A., Salimova A.R., Andreev E.N., Dzhemilev U.M.
Mendeleev Communications,
2017
18.
D’yakonov V.A., Makarov A.A., Dzhemileva L.U., Andreev E.N., Dzhemilev U.M.
Mendeleev Communications,
2017
19.

D’yakonov V.A., Dzhemileva L.U., Makarov A.A., Mulyukova A.R., Baev D.S., Khusnutdinova E.K., Tolstikova T.G., Dzhemilev U.M.
Medicinal Chemistry Research,
2015
20.

D’yakonov V.A., Dzhemileva L.U., Tuktarova R.A., Makarov A.A., Islamov I.I., Mulyukova A.R., Dzhemilev U.M.
Steroids,
2015
21.

Abakumov G.A., Piskunov A.V., Cherkasov V.K., Fedushkin I.L., Ananikov V.P., Eremin D.B., Gordeev E.G., Beletskaya I.P., Averin A.D., Bochkarev M.N., Trifonov A.A., Dzhemilev U.M., D'yakonov V.A., Egorov M.P., Vereshchagin A.N., et. al.
Russian Chemical Reviews,
2018
22.

Parenty A., Moreau X., Campagne J.-.
Chemical Reviews,
2006
23.

de Léséleuc M., Collins S.K.
Chemical Communications,
2015
24.
10.1016/j.mencom.2018.09.017_bib0120
Levy
Carbon-13 Nuclear Magnetic Resonance for Organic Chemists,
1972
25.

D'yakonov V.A., Makarov A.A., Makarova E.K., Dzhemilev U.M.
Tetrahedron,
2013
26.

D’yakonov V.A., Makarov A.A., Makarova E.K., Khalilov L.M., Dzhemilev U.M.
Russian Chemical Bulletin,
2012